Valacyclovir hydrochlorideCAS# 124832-27-5 |
- Daptomycin
Catalog No.:BCC1057
CAS No.:103060-53-3
- Nelarabine
Catalog No.:BCC1072
CAS No.:121032-29-9
- Gemcitabine HCl
Catalog No.:BCC1076
CAS No.:122111-03-9
- Clofarabine
Catalog No.:BCC1078
CAS No.:123318-82-1
- Ifosfamide
Catalog No.:BCC1164
CAS No.:3778-73-2
Quality Control & MSDS
Number of papers citing our products

Chemical structure

3D structure
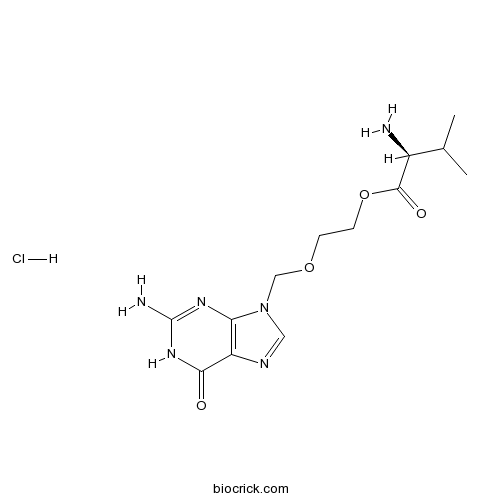
| Cas No. | 124832-27-5 | SDF | Download SDF |
| PubChem ID | 135398741 | Appearance | Powder |
| Formula | C13H21ClN6O4 | M.Wt | 360.8 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | Valaciclovir hydrochloride | ||
| Solubility | DMSO : 25 mg/mL (69.29 mM; Need ultrasonic) | ||
| Chemical Name | 2-[(2-amino-6-oxo-1~{H}-purin-9-yl)methoxy]ethyl (2~{S})-2-amino-3-methylbutanoate;hydrochloride | ||
| SMILES | [Cl-].CC(C)[C@H](N)C(=O)OCCOCn1cnc2C(=O)N=C(N)Nc12.[H+] | ||
| Standard InChIKey | ZCDDBUOENGJMLV-QRPNPIFTSA-N | ||
| Standard InChI | InChI=1S/C13H20N6O4.ClH/c1-7(2)8(14)12(21)23-4-3-22-6-19-5-16-9-10(19)17-13(15)18-11(9)20;/h5,7-8H,3-4,6,14H2,1-2H3,(H3,15,17,18,20);1H/t8-;/m0./s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Valacyclovir hydrochloride is an antiviral drug used in the management of herpes simplex, herpes zoster, and herpes B.
Target: HSV
Valacyclovir is an antiviral drug used in the management of herpes simplex, herpes zoster, and herpes B. VACV uptake was concentration dependent and saturable with a Michaelis-Menten constant and maximum velocity of 1.64 +/- 0.06 mM and 23.34 +/- 0.36 nmol/mg protein/5 min, respectively. A very similar Km value was obtained in hPEPT1/CHO cells and in rat and rabbit tissues and Caco-2 cells, suggesting that hPEPT1 dominates the intestinal transport properties of VACV in vitro .
For treatment of a first episode of genital herpes, a large comparative trial has shown that valacyclovir (1 g twice a day) is as effective as acyclovir (200 mg five times a day) when given for 10 days. For treating recurrences, two trials show that valacyclovir is as effective as acyclovir (200 mg five times a day) with a treatment period of 5 days. A daily dose of 1 g of valacyclovir is as effective as 2 g daily. Valacyclovir can be administered once a day. The concentrations of acyclovir in serum and CSF were measured at steady state after 6 days of oral treatment with 1,000 mg of valacyclovir three times a day. EC50 values of PE and AC in 3T3 cells were 0.02 and 0.01 ug/ml, while values in BHK cells were 0.2 and 0.03 ug/ml. Treatment of infected immunosuppressed mice and FA and VA (b.i.d., 5.5 days) reduced the proportion with erythema from 100% to 24% and 38%, and eliminated ear paralysis, ear lesions (vesicles, etc) and death. Virus was absent from ear and brainstem by day 6, but reappeared after discontinuation in mice treated with VA. References: | |||||

Valacyclovir hydrochloride Dilution Calculator

Valacyclovir hydrochloride Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.7716 mL | 13.8581 mL | 27.7162 mL | 55.4324 mL | 69.2905 mL |
| 5 mM | 0.5543 mL | 2.7716 mL | 5.5432 mL | 11.0865 mL | 13.8581 mL |
| 10 mM | 0.2772 mL | 1.3858 mL | 2.7716 mL | 5.5432 mL | 6.929 mL |
| 50 mM | 0.0554 mL | 0.2772 mL | 0.5543 mL | 1.1086 mL | 1.3858 mL |
| 100 mM | 0.0277 mL | 0.1386 mL | 0.2772 mL | 0.5543 mL | 0.6929 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Valacyclovir hydrochloride is an antiviral drug used in the management of herpes simplex, herpes zoster, and herpes B.
- Valaciclovir
Catalog No.:BCC2025
CAS No.:124832-26-4
- USP7-USP47 inhibitor
Catalog No.:BCC4113
CAS No.:1247825-37-1
- P 22077
Catalog No.:BCC3616
CAS No.:1247819-59-5
- Losartan Potassium (DuP 753)
Catalog No.:BCC1080
CAS No.:124750-99-8
- Quinovic acid 3-O-(6-deoxy-beta-D-glucopyranoside) 28-O-beta-D-glucopyranosyl ester
Catalog No.:BCN1595
CAS No.:124727-10-2
- Meprednisone
Catalog No.:BCC4893
CAS No.:1247-42-3
- Methyl-Dodovisate A
Catalog No.:BCN4719
CAS No.:1246937-33-6
- 5'-Prenylaliarin
Catalog No.:BCN4829
CAS No.:1246926-09-9
- 5,7,4'-Trihydroxy-3,6-dimethoxy-3',5'-diprenylflavone
Catalog No.:BCN1596
CAS No.:1246926-08-8
- VS-5584 (SB2343)
Catalog No.:BCC2047
CAS No.:1246560-33-7
- MPI-0479605
Catalog No.:BCC5347
CAS No.:1246529-32-7
- SR1078
Catalog No.:BCC1963
CAS No.:1246525-60-9
- Nadifloxacin
Catalog No.:BCC4804
CAS No.:124858-35-1
- 5-Dehydroxyparatocarpin K
Catalog No.:BCN4017
CAS No.:124858-37-3
- Isoaltholactone
Catalog No.:BCN4826
CAS No.:124868-11-7
- Benzoyl leuco methylene blue
Catalog No.:BCC8862
CAS No.:1249-97-4
- (R)-(+)-Tolterodine
Catalog No.:BCC4054
CAS No.:124937-51-5
- Tolterodine tartrate
Catalog No.:BCC4586
CAS No.:124937-52-6
- Tectoroside
Catalog No.:BCN6672
CAS No.:124960-89-0
- Raddeanoside R8
Catalog No.:BCN6555
CAS No.:124961-61-1
- Ajugacumbin A
Catalog No.:BCN3657
CAS No.:124961-66-6
- Ajugamarin L2
Catalog No.:BCN3662
CAS No.:124961-67-7
- Prednisone 21-acetate
Catalog No.:BCC9128
CAS No.:125-10-0
- Isobornyl acetate
Catalog No.:BCN8296
CAS No.:125-12-2
Acute renal injury induced by valacyclovir hydrochloride: A case report.[Pubmed:28101180]
Exp Ther Med. 2016 Dec;12(6):4025-4028.
Acyclovir has been a frequently used antiviral agent in the clinical treatment of leukemia, acute encephalitis, malignant tumor and herpes simplex. The adverse effects of this drug have been widely described in clinical practice. In the present study, a case of a 35-year-old female patient diagnosed with herpes simplex, who developed acute renal injury following treatment with Valacyclovir hydrochloride, is described. Kidney biopsy, light microscopy and laboratory examination were performed, and all findings revealed the signs of evident vacuolar degeneration of capillary endothelial and renal tubular epithelial cells, erythrocyte aggregation in partial renal tubule and microvilli exfoliation from epithelial cells. Renal interstitial edema was clearly identified. The clinical evidence observed from this female patient indicated that renal functions should be closely monitored during Valacyclovir hydrochloride administration. A variety of effective measures, such as hydration, alkalizing urine, promoting the discharge of medication and the use of antagonists are recommended following the administration of antiviral agents.
Kinetic Study of the Alkaline Degradation of Oseltamivir Phosphate and Valacyclovir Hydrochloride using Validated Stability Indicating HPLC.[Pubmed:24932100]
Anal Chem Insights. 2014 May 27;9:41-8.
Aqueous alkaline degradation was performed for oseltamivir phosphate (OP) and Valacyclovir hydrochloride (VA). Isocratic stability indicating the use of high-performance liquid chromatography (HPLC) was presented for each drug in the presence of its degradation product. The separations were performed using the Nucleosil ODS column and a mobile phase consisting of phosphate buffer (pH = 7), acetonitrile, and methanol 50:25:25 (v/v/v) for OP. For VA separation, a Nucleosil CN column using phosphate buffer (pH = 7) and methanol 85:15 (v/v) was used as a mobile phase. Ultraviolet detection at 210 nm and 254 nm was used for OP and VA, respectively. The method showed high sensitivity concerning linearity, accuracy, and precision over the range 1-250 mug mL(-1) for both drugs. The proposed method was used to determine the drug in its pharmaceutical formulation and to investigate the degradation kinetics of each drug's alkaline-stressed samples. The reactions were found to follow a first-order reaction. The activation energy could also be estimated. International Conference on Harmonisation guidelines were adopted for method validation.
Analysis of the antiviral drugs acyclovir and valacyclovir-hydrochloride in tsetse flies (Glossina pallidipes) using LC-MSMS.[Pubmed:20719583]
J Chromatogr B Analyt Technol Biomed Life Sci. 2010 Sep 15;878(26):2384-90.
A new simple, sensitive and precise liquid chromatography-tandem mass spectrometry method has been developed and validated for the determination of valacyclovir-HCl and acyclovir in tsetse flies (Glossina pallipides). Tsetse flies were extracted by ultrasonication with acidified methanol/acetonitrile, centrifuged and cleaned up by solid phase dispersion using MgSO(4) and MSPD C(18) material. Samples were analysed using a Waters Alliance 2695 series HPLC with a C(18) Gemini analytical column (150 mm x 4.6 mm x 5 microm) and a guard cartridge column connected to a Waters Quattro-Micro triple-quadrupole mass spectrometer. The isocratic mobile phase consisted of methanol:acetonitrile:water (60:30:10, v/v/v) plus formic acid (0.1%) at a flow rate of 0.25 ml/min. The precursor>product ion transition for valacyclovir (m/z 325.1>152) and acyclovir (m/z 226.1>151.9) were monitored in positive electrospray multiple reaction monitoring mode. The method was validated at fortification levels of 0.5, 1 and 2 microg/g. The range of calibration for both drugs was 0.45-4.5 microg/g. The overall accuracy of the method was 92% for valacyclovir and 95% for acyclovir with corresponding within-laboratory reproducibilities of 4.4 and 3.4%, respectively. Mean recoveries were above 80% for both drugs and repeatability ranged from 0.7 to 6.1%. For both drugs the limits of detection and quantification were 0.0625 and 0.2 microg/g, respectively. The method was applied in experiments on the mass rearing of tsetse flies for sterile insect technique (SIT) applications, in which the flies were fed with blood meals containing acyclovir or valcyclovir-HCl prior to analysis to assess effects on Glossina pallidipes Salivary Gland Hypertrophy syndrome.
Formulation development, optimization, and evaluation of sustained release tablet of valacyclovir hydrochloride by combined approach of floating and swelling for better gastric retention.[Pubmed:25787207]
Drug Deliv Transl Res. 2014 Dec;4(5-6):452-64.
The present study is intended to enhance gastric retention of sustained release tablet of Valacyclovir hydrochloride by combined approach of floating and swelling. The tablets are prepared by direct compression method. Polyethylene oxide (Polyox WSR 303) is selected as the swelling matrix agent. Sodium starch glycolate (SSG) is used as swelling enhancer, and sodium bicarbonate is used as an effervescent agent for floating. A 3(2) full factorial design is applied to systematically optimize the formulation. The concentration of Polyox WSR 303 (X 1) and concentration of SSG (X 2) are selected as independent variables. The percentage drug release at 12 h, floating lag time, and maximum percentage swelling are selected as dependent variables. Formulations are evaluated for hardness, friability, floating lag time, total floating time, percentage swelling, in vitro drug release, and in vivo floating study. The results indicated that X 1 and X 2 significantly affected the drug release properties, floating lag times, and maximum percentage swelling. Release rate decreases as the concentration of Polyox increased. Regression analysis and numerical optimization are performed to identify the best formulation. Formulation F5 prepared with Polyox WSR 303 (15 %) and SSG (10 %) is found to be the best formulation. F5 followed zero-order release mechanism. Swelling and floating gastroretentive tablets of valacyclovir HCl are successfully formulated with controlled delivery to stomach with an aim of increasing the mean residence time in the upper part of GIT where the drug has its absorption window.


