Isodon serra
Isodon serra
1. The products in our compound library are selected from thousands of unique natural products; 2. It has the characteristics of diverse structure, diverse sources and wide coverage of activities; 3. Provide information on the activity of products from major journals, patents and research reports around the world, providing theoretical direction and research basis for further research and screening; 4. Free combination according to the type, source, target and disease of natural product; 5. The compound powder is placed in a covered tube and then discharged into a 10 x 10 cryostat; 6. Transport in ice pack or dry ice pack. Please store it at -20 °C as soon as possible after receiving the product, and use it as soon as possible after opening.
Natural products/compounds from Isodon serra
- Cat.No. Product Name CAS Number COA
-
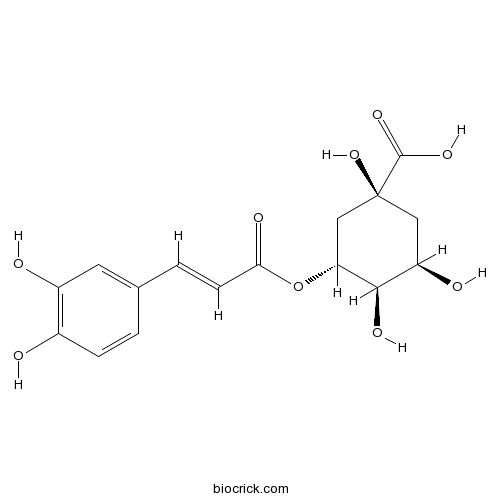
BCN5906
Chlorogenic acid327-97-9
Instructions
Ent-Abietanoids Isolated from Isodon serra.[Pubmed: 28218684]
None
Bioactive ent-kaurane diterpenoids from Isodon serra.[Pubmed: 27298277]
Nine 7,20-epoxy-ent-kaurane diterpenoids (15-acetylmegathyrin B, serrin E, 14β-hydroxyrabdocoestin A, serrin F, serrin G, 11-epi-rabdocoestin A, serrin H, serrin I, and 15-acetylenanderianin N), along with seven known ones, were isolated from the aerial parts of Isodon serra. Their structures were elucidated by extensive spectroscopic analysis, and the absolute configuration of 15-acetylmegathyrin B was determined by signal-crystal X-ray diffraction. All of these compounds were evaluated for their cytotoxic activities against five human tumor cell lines (HL-60, SMMC-7721, A-549, MCF-7, SW480). Serrin F, rabdocoestin B and 1α,11β-dihydroxy-1α,11β-acetonide-7α,20-epoxy-ent-kaur-16-en-15-one showed cytotoxic activities against all cell lines, with IC50 values ranging from 0.7 to 4.6 μM; serrin F also strongly inhibited NO production in LPS-stimulated RAW264.7 cells. Otherwise, 14β-hydroxyrabdocoestin A, serrins H and I, as well as enanderianin N and megathyrin B, also exhibited inhibitory effects towards NO production, while no cytotoxicity against five cell lines was detected.
Investigation of free amino acid, total phenolics, antioxidant activity and purine alkaloids to assess the health properties of non-Camellia tea.[Pubmed: 27006902]
To find novel functional beverages from folk teas, 33 species of frequently used non-Camellia tea (plants other than Camellia) were collected and compared with Camellia tea (green tea, pu-erh tea and black tea) for the first time. Data are reported here on the quantities of 20 free amino acids (FAAs) and three purine alkaloids (measured by UHPLC), total polyphenols (measured by Folin-Ciocalteu assay), and antioxidant activity (DPPH). The total amounts of FAAs in non-Camellia tea (0.62-18.99 mg/g) are generally less than that of Camellia tea (16.55-24.99 mg/g). However, for certain FAAs, the quantities were much higher in some non-Camellia teas, such as γ-aminobutyric acid in teas from Ampelopsis grossedentata, Isodon serra and Hibiscus sabdariffa. Interestingly, theanine was detected in tea from Potentilla fruticosa (1.16±0.81 mg/g). Furthermore, the content of polyphenols in teas from A. grossedentata, Acer tataricum subsp. ginnala are significantly higher than those from Camellia tea; teas from I. serra, Pistacia chinensis and A. tataricum subsp. ginnala have remarkable antioxidant activities similar to the activities from green tea (44.23 μg/mL). Purine alkaloids (caffeine, theobromine and theophylline) were not detected in non-Camellia teas. The investigation suggest some non-Camellia teas may be great functional natural products with potential for prevention of chronic diseases and aging, by providing with abundant polyphenols, antioxidants and specific FAAs.


