Reseda odorata
Reseda odorata
1. The products in our compound library are selected from thousands of unique natural products; 2. It has the characteristics of diverse structure, diverse sources and wide coverage of activities; 3. Provide information on the activity of products from major journals, patents and research reports around the world, providing theoretical direction and research basis for further research and screening; 4. Free combination according to the type, source, target and disease of natural product; 5. The compound powder is placed in a covered tube and then discharged into a 10 x 10 cryostat; 6. Transport in ice pack or dry ice pack. Please store it at -20 °C as soon as possible after receiving the product, and use it as soon as possible after opening.
Natural products/compounds from Reseda odorata
- Cat.No. Product Name CAS Number COA
-
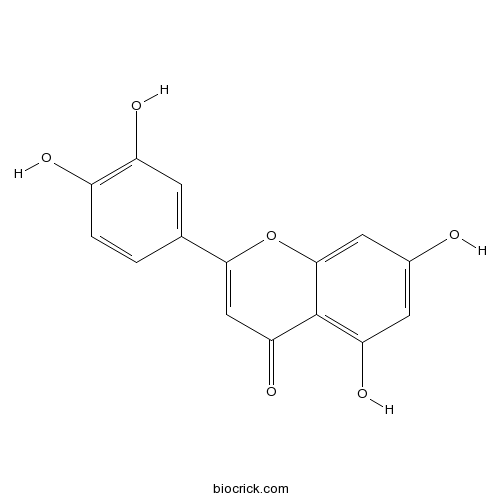
BCN5600
Luteolin491-70-3
Instructions
The crystal structure of 3-hydroxy-3-isobutyl-2-pyrrolidone-5-carboxylic acid, lactam of 4-hydroxy-4-isobutylglutamic acid from Reseda odorata L.[Pubmed: 883458]
The relative configuration of 3-hydroxy-3-isobutyl-2-pyrrolidone-5-carboxylic acid has been determined by an X-ray crystal structure analysis. The compound crystallized in space group P212121 with two molecules in the asymmetric unit. a=16.023(3) A, b=19.349(7) A, c=6.9053(16) A. The structure was solved by direct methods using MULTAN and refined by full-matrix least-squares technique to an R of 0.137 for 645 diffractometer-collected intensities. The absolute configurations of the title compounds were deduced; the configuration of the amino acid and the lactam are 2(S), 4(S) and 3(S), 5(S), respectively.
Biosynthesis of phenylalanine, tyrosine, 3-(3-carbocyphenyl) alanine and 3-(3-carbocy-4-hydroxyphenyl) alanine in higher plants. Examples of the transformation possibilities for chorismic acid.[Pubmed: 1120151]
14C-labelled shikimic acid and double labelled shikimic acid tritiated stereospicifically at C-6 are incorporated into 3-(3-carboxyphenyl) alanine, 3-(3-carboxy-4-hydroxyphenyl) alanine, phenylalanine, and tyrosine in Reseda lutea L., Reseda odorata L., Iris x Hollandica cv. Prof. Blauw, and Iris x hollandica cv. Wedgwood. The experiments with 14C-labelled shikimic acid confirm that the aromatic carboxyl groups and rings in 3-(3-carboxyphenyl) alanine and 3-(3-carbocy-4-hydroxyphenyl) alanine derive from the carbocyl group and ring in shikimic acid whereas the experiments with double labelled shikimic acid demonstrate that the pro-6S-hydrogen atom is retained and the pro-6R-hydrogen atom lost in the biosynthesis of 3-(3-carboxyphenyl) alanine, phenylalanine, and tyrosine in the plants used. 3H was located in the ortho-position in the aromatic rings of phenylalanine and tyrosine but in a position para to the alanine side chain of 3- (3-carboxyphenly) alanine. No 3H was found in 3- (3-carboxy-4-hydroxyphenyl) alanine. This supports a derivation of the last two compounds from chorismic acid via isochorismic acid, isoprephenic acid, and 3'-carboxyphenylpyruvic acid and 3'-carbocy-4'-hydroxyphenylphruvic acid. The 3H/14C ratio in 3-(3-carboxyphenyl) alanine was found higher than in the precursor used. This isotope effect must operate by competition between the pathways from isoprephenic acid to 3'-carbocyphenylpyruvic acid and to 3'-carbocy-4'- hydroxyphenylpyruvic acic. The proposed biosynthetic pathways for the two carboxy-substituted amino acids are in agreement with their distribution patterns in the plant kingdom and suggest that they may derive from minor changes of enzymes involved in the general pathways of aromatic biosynthesis.


