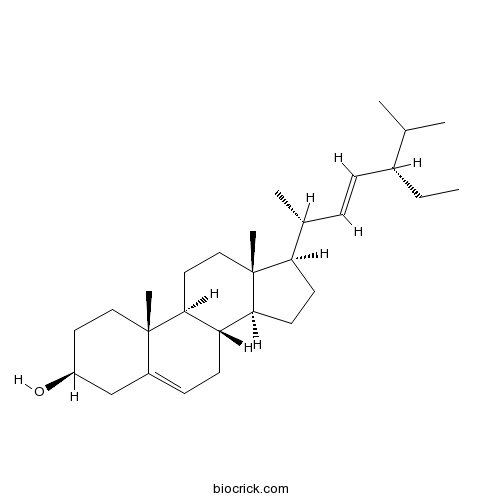
A 3β-sterol that consists of 3β-hydroxystigmastane having double bonds at the 5,6- and 22,23-positions.
InChI=1S/C29H48O/c1-7-21(19(2)3)9-8-20(4)25-12-13-26-24-11-10-22-18-23(30)14-16-28(22,5)27(24)15-17-29(25,26)6/h8-10,19-21,23-27,30H,7,11-18H2,1-6H3/b9-8+/t20-,21-,23+,24+,25-,26+,27+,28+,29-/m1/s1
Stigmasterol, a soy lipid-derived phytosterol, is an antagonist of the bile acid nuclear receptor FXR.[1]
Stigmasterol, isolated from the bark of Butea monosperma, has thyroid inhibitory, anti-peroxidative and hypoglycemic effects .[2]
Stigmasterol has potential anti-osteoarthritic properties, can inhibit several pro-inflammatory and matrix degradation mediators typically involved in OA-induced cartilage degradation, at least in part through the inhibition of the NF-kappaB pathway.[3]
Stigmasterol can inhibit tumour promotion in mouse skin two-stage carcinogenesis.[4]
Stigmasterol can induce cognitive ameliorative effects, mediated by the enhancement of cholinergic neurotransmission system via the activation of estrogen or NMDA receptors.[5]
Stigmasterol, when fed, lowers plasma cholesterol levels, inhibits intestinal cholesterol and plant sterol absorption, and suppresses hepatic cholesterol and classic bile acid synthesis in Wistar as well as WKY rats. However, plasma and hepatic incorporation of stigmasterol is low.[6]
English website: Stigmasterol
Japanese website: Stigmasterol
Chinese website: Stigmasterol
[1] Carter B A, Taylor O A, Prendergast D R, et al. Pediatric Res, 2007, 62(3):301-6.
[2] Panda S, Jafri M, Kar A, et al. Fitoterapia, 2009, 80(2):123-6.
[3] Gabay O, Sanchez C, Salvat C, et al. Osteoarthr Cartilage, 2010, 18(1):106-16.
[4] Kasahara Y, Kumaki K, Katagiri S, et al. Phytother Res 1994, 8(6):327-31.
[5] Park S J, Dong H K, Jung J M, et al. Eur J Pharmacol, 2012, 676(1-3):64-70.
[6] Batta A K, Xu G, Honda A, et al. Metabolism , 2006, 55(3):292-9.
[7] Liu S Y, Sun L J, Guo X. Adv Materials Res, 2011, 233-235:1206-9.