Gynura japonica
Gynura japonica
1. The products in our compound library are selected from thousands of unique natural products; 2. It has the characteristics of diverse structure, diverse sources and wide coverage of activities; 3. Provide information on the activity of products from major journals, patents and research reports around the world, providing theoretical direction and research basis for further research and screening; 4. Free combination according to the type, source, target and disease of natural product; 5. The compound powder is placed in a covered tube and then discharged into a 10 x 10 cryostat; 6. Transport in ice pack or dry ice pack. Please store it at -20 °C as soon as possible after receiving the product, and use it as soon as possible after opening.
Natural products/compounds from Gynura japonica
- Cat.No. Product Name CAS Number COA
-
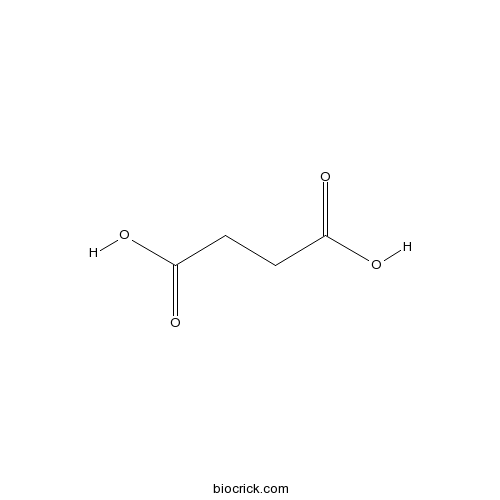
BCN5890
Succinic acid110-15-6
Instructions
-
BCN5563
Seneciphylline480-81-9
Instructions

-
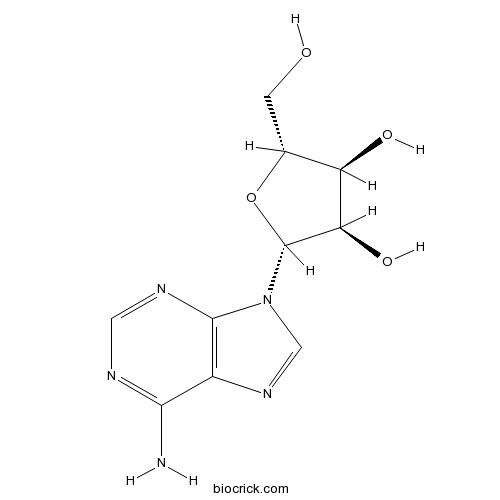
BCN5796
Adenosine58-61-7
Instructions
Mass-spectrometry-directed analysis and purification of pyrrolizidine alkaloid cis/trans isomers in Gynura japonica.[Pubmed: 24840731]
Pyrrolizidine alkaloids are highly hepatotoxic natural chemicals that produce irreversible chronic and acute hepatotoxic effects on human beings. Purification of large amounts of pyrrolizidine alkaloids is necessary for toxicity studies. In this study, an efficient method for targeted analysis and purification of pyrrolizidine alkaloid cis/trans isomers from herbal materials was developed for the first time. Targeted analysis of the hepatotoxic pyrrolizidine alkaloids was performed by liquid chromatography with tandem mass spectrometry (precursor ion scan and daughter ion scan), and the purification of pyrrolizidine alkaloids was achieved with a mass-directed auto purification system. The extraction and preparative liquid chromatography conditions were optimized. The developed method was applied to analysis of Gynura japonica (Thunb.) Juel., a herbal medicine traditionally used for detumescence and relieving pain but is potentially hepatotoxic as it contains pyrrolizidine alkaloids. Twelve pyrrolizidine alkaloids (six cis/trans isomer pairs) were identified with reference compounds or characterized by liquid chromatography with tandem mass spectrometry, and five individual pyrrolizidine alkaloids, including (E)-seneciphylline, seneciphylline, integerrimine, senecionine, and seneciphyllinine, were prepared from G. japonica roots with high efficiency. The results of this work provide a new technique for the preparation of large amounts of pyrrolizidine alkaloid reference substances, which will also benefit toxicological studies of pyrrolizidine alkaloids and treatments for pyrrolizidine alkaloid-induced toxicity.
Anti-platelet aggregation and chemical constituents from the rhizome of Gynura japonica.[Pubmed: 14531028]
A novel quinonoid terpenoid, (-)- alpha-tocospirone, a new chromanone, (-)-gynuraone, as well as three new steroids, (22 E,24 S)-7 alpha-hydroperoxystigmasta-5,22-dien-3 beta-ol, (22 E,24 S)-stigmasta-1,4,22-trien-3-one, and (24 R)-stigmasta-1,4-dien-3-one, together with 15 known components, were isolated from the rhizome of Formosan Gynura japonica. Structures of the new compounds were determined through spectral analyses and chemical evidence. Several of the isolated compounds, caryophyllene oxide, 6-acetyl-2,2-dimethylchroman-4-one, vanillin, 2,6-dimethoxy-1,4-benzoquinone, and benzoic acid exhibited significant anti-platelet aggregation activity in vitro.


