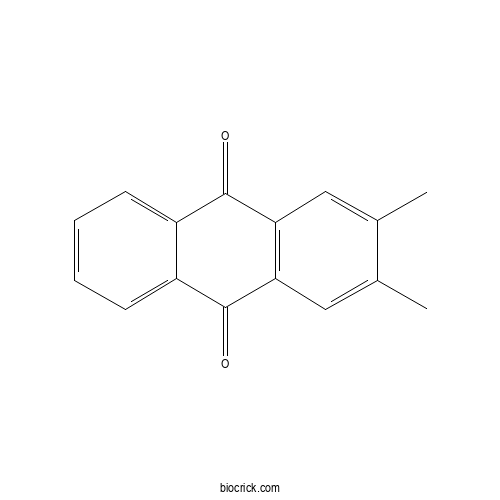
2,3-DimethylanthraquinoneCAS# 6531-35-7 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 6531-35-7 | SDF | File under preparation. |
| PubChem ID | 81019 | Appearance | Powder |
| Formula | C16H12O2 | M.Wt | 236.3 |
| Type of Compound | Anthraquinones | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | 2,3-dimethylanthracene-9,10-dione | ||
| SMILES | CC1=CC2=C(C=C1C)C(=O)C3=CC=CC=C3C2=O | ||
| Standard InChIKey | KIJPZYXCIHZVGP-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C16H12O2/c1-9-7-13-14(8-10(9)2)16(18)12-6-4-3-5-11(12)15(13)17/h3-8H,1-2H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 2,3-Dimethylanthraquinone shows carcinogenicity. | |||||

2,3-Dimethylanthraquinone Dilution Calculator

2,3-Dimethylanthraquinone Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 4.2319 mL | 21.1595 mL | 42.3191 mL | 84.6382 mL | 105.7977 mL |
| 5 mM | 0.8464 mL | 4.2319 mL | 8.4638 mL | 16.9276 mL | 21.1595 mL |
| 10 mM | 0.4232 mL | 2.116 mL | 4.2319 mL | 8.4638 mL | 10.5798 mL |
| 50 mM | 0.0846 mL | 0.4232 mL | 0.8464 mL | 1.6928 mL | 2.116 mL |
| 100 mM | 0.0423 mL | 0.2116 mL | 0.4232 mL | 0.8464 mL | 1.058 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 2,5-Dimethoxybenzoic acid
Catalog No.:BCN0160
CAS No.:2785-98-0
- Bicyclo[5.3.0]decapentaene
Catalog No.:BCN0159
CAS No.:275-51-4
- (E)-N-(2-Amino-4-fluorophenyl)-3-(1-cinnamyl-1H-pyrazol-4-yl)acrylamide
Catalog No.:BCN0158
CAS No.:1396841-57-8
- 2-Hydroxy-3-methoxybenzaldehyde
Catalog No.:BCN0157
CAS No.:148-53-8
- Lappaol B
Catalog No.:BCN0156
CAS No.:62359-60-8
- N-trans-caffeoyltyramine
Catalog No.:BCN0155
CAS No.:103188-48-3
- Helenien
Catalog No.:BCN0154
CAS No.:547-17-1
- Comanthosid B
Catalog No.:BCN0153
CAS No.:70938-60-2
- Candicine
Catalog No.:BCN0151
CAS No.:6656-13-9
- Maritimein
Catalog No.:BCN0150
CAS No.:490-54-0
- 3',4',7-Trihydroxyisoflavone
Catalog No.:BCN0149
CAS No.:485-63-2
- 2'-Hydroxyflavanone
Catalog No.:BCN0148
CAS No.:17348-76-4
- Qingdainone
Catalog No.:BCN0162
CAS No.:97457-31-3
- 7-Hydroxy-4'-methoxyflavone
Catalog No.:BCN0163
CAS No.:487-24-1
- 5-Hydroxyflavone
Catalog No.:BCN0164
CAS No.:491-78-1
- Yohimbic acid monohydrate
Catalog No.:BCN0165
CAS No.:522-87-2
- Ethyl isovalerate
Catalog No.:BCN0166
CAS No.:108-64-5
- 2,3-Dihydroxybenzoic acid
Catalog No.:BCN0167
CAS No.:303-38-8
- trans-Chrysanthemyl alcohol
Catalog No.:BCN9908
CAS No.:5617-92-5
- 6-Hydroxy-7-methoxydihydroligustilide
Catalog No.:BCN0168
CAS No.:210036-09-2
- Perlolyrine
Catalog No.:BCN0169
CAS No.:29700-20-7
- 3-Oxo-4-benzyl-3,4-dihydro-1H-pyrrolo [2,1-c] oxazine-6-methylal
Catalog No.:BCN0170
CAS No.:60026-28-0
- Eriodictyol 7-O-glucuronide
Catalog No.:BCN0171
CAS No.:125535-06-0
- Corymboside
Catalog No.:BCN0172
CAS No.:73543-87-0
An unusual reaction of the natural compound benaphthamycin B: theoretical study of a model system.[Pubmed:16438523]
J Org Chem. 2006 Feb 3;71(3):1074-9.
The surprising and complex transformation of benaphthamycin B to give quinone 2a is investigated theoretically with a model compound, 1,5-dihydroxy-4-methoxy-2,3-Dimethylanthraquinone (3). The detailed study is performed using both DFT and perturbation theory under inclusion of solvent effects. Several individual steps (reduction and hydrolysis, water elimination, ether cleavage, and oxidation) of the proposed reaction cascade calculated at the PCM-MP2/6-31G(d)//B3LYP/6-31G(d) level of theory are presented and discussed. It is shown that the key step, the ether cleavage as an S(N)2 reaction leading to the anthrone 12a, possesses a smaller activation barrier compared to the alternative process yielding 12b. Therefore, the formation of the thermodynamically preferred model quinone 13a is also the kinetically favored pathway: The results of the calculated model reaction should also be valid for benaphthamycin B (1).


