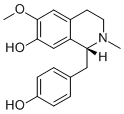
(+/-)-N-methylcoclaurineCAS# 1472-62-4 |
Quality Control & MSDS
Package In Stock
Number of papers citing our products

| Cas No. | 1472-62-4 | SDF | File under preparation. |
| PubChem ID | N/A | Appearance | Powder |
| Formula | C18H21NO3 | M.Wt | 299.4 |
| Type of Compound | Alkaloids | Storage | Desiccate at -20°C |
| Synonyms | (±)-N-Methylcoclaurine | ||
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

(+/-)-N-methylcoclaurine Dilution Calculator

(+/-)-N-methylcoclaurine Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.34 mL | 16.7001 mL | 33.4001 mL | 66.8003 mL | 83.5003 mL |
| 5 mM | 0.668 mL | 3.34 mL | 6.68 mL | 13.3601 mL | 16.7001 mL |
| 10 mM | 0.334 mL | 1.67 mL | 3.34 mL | 6.68 mL | 8.35 mL |
| 50 mM | 0.0668 mL | 0.334 mL | 0.668 mL | 1.336 mL | 1.67 mL |
| 100 mM | 0.0334 mL | 0.167 mL | 0.334 mL | 0.668 mL | 0.835 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Bis(2-ethylhexyl) terephthalate
Catalog No.:BCN0457
CAS No.:6422-86-2
- Toxyloxanthone B
Catalog No.:BCN0456
CAS No.:50875-24-6
- V1 iridoid
Catalog No.:BCN0455
CAS No.:87441-71-2
- Acronyculatin Q
Catalog No.:BCN0454
CAS No.:2287183-54-2
- Mecambridine
Catalog No.:BCN0453
CAS No.:31098-60-9
- Acropyrone
Catalog No.:BCN0452
CAS No.:1381875-14-4
- Furanofukinin
Catalog No.:BCN0451
CAS No.:34335-93-8
- 6-O-Ethyltetradymodiol
Catalog No.:BCN0450
CAS No.:1028530-06-4
- 6β-Ethoxyfuranoeremophilane
Catalog No.:BCN0449
CAS No.:82462-36-0
- Phenylacetylglycine
Catalog No.:BCN0448
CAS No.:500-98-1
- N1-Methyl-2-pyridone-5-carboxamide
Catalog No.:BCN0447
CAS No.:701-44-0
- Petasalbin
Catalog No.:BCN0446
CAS No.:4176-11-8
- 5-exo-Hydroxyborneol
Catalog No.:BCN0459
CAS No.:32751-73-8
- 3,8-Di-O-methylellagic acid 2-O-(5'-O-acetyl)arabinofuranoside
Catalog No.:BCN0460
CAS No.:365540-09-6
- 4,5-Dioxodehydroasimilobine
Catalog No.:BCN0461
CAS No.:82644-81-3
- 2,4-Dihydroxy-6-methoxy-3,5-diprenylacetophenone
Catalog No.:BCN0462
CAS No.:123999-38-2
- Alpinigenine
Catalog No.:BCN0463
CAS No.:14028-91-2
- Tabernaecorymbosine A
Catalog No.:BCN0464
CAS No.:1262306-81-9
- Flindulatin
Catalog No.:BCN0465
CAS No.:521-44-8
- (±)-Cleroindicin F
Catalog No.:BCN0466
CAS No.:94535-01-0
- p-O-Farnesylcoumaric acid
Catalog No.:BCN0467
CAS No.:939769-47-8
- 6,4'-Dihydroxy-5,7-dimethoxyflavanone
Catalog No.:BCN0468
CAS No.:6951-57-1
- 5-endo-Hydroxyborneol
Catalog No.:BCN0469
CAS No.:68738-10-3
- 1-O-p-Coumaroylglucose
Catalog No.:BCN0470
CAS No.:7139-64-2
Isolation and characterization of two O-methyltransferases involved in benzylisoquinoline alkaloid biosynthesis in sacred lotus (Nelumbo nucifera).[Pubmed:31914404]
J Biol Chem. 2020 Feb 7;295(6):1598-1612.
Benzylisoquinoline alkaloids (BIAs) are a major class of plant metabolites with many pharmacological benefits. Sacred lotus (Nelumbo nucifera) is an ancient aquatic plant of medicinal value because of antiviral and immunomodulatory activities linked to its constituent BIAs. Although more than 30 BIAs belonging to the 1-benzylisoquinoline, aporphine, and bisbenzylisoquinoline structural subclasses and displaying a predominant R-enantiomeric conformation have been isolated from N. nucifera, its BIA biosynthetic genes and enzymes remain unknown. Herein, we report the isolation and biochemical characterization of two O-methyltransferases (OMTs) involved in BIA biosynthesis in sacred lotus. Five homologous genes, designated NnOMT1-5 and encoding polypeptides sharing >40% amino acid sequence identity, were expressed in Escherichia coli Functional characterization of the purified recombinant proteins revealed that NnOMT1 is a regiospecific 1-benzylisoquinoline 6-O-methyltransferase (6OMT) accepting both R- and S-substrates, whereas NnOMT5 is mainly a 7-O-methyltransferase (7OMT), with relatively minor 6OMT activity and a strong stereospecific preference for S-enantiomers. Available aporphines were not accepted as substrates by either enzyme, suggesting that O-methylation precedes BIA formation from 1-benzylisoquinoline intermediates. Km values for NnOMT1 and NnOMT5 were 20 and 13 mum for (R,S)-norcoclaurine and (S)-N-methylcoclaurine, respectively, similar to those for OMTs from other BIA-producing plants. Organ-based correlations of alkaloid content, OMT activity in crude extracts, and OMT gene expression supported physiological roles for NnOMT1 and NnOMT5 in BIA metabolism, occurring primarily in young leaves and embryos of sacred lotus. In summary, our work identifies two OMTs involved in BIA metabolism in the medicinal plant N. nucifera.
Identification and characterization of methyltransferases involved in benzylisoquinoline alkaloids biosynthesis from Stephania intermedia.[Pubmed:31865477]
Biotechnol Lett. 2020 Mar;42(3):461-469.
OBJECTIVES: To characterize methyltransferases involved in the biosynthesis of benzylisoquinoline alkaloids in Stephania intermedia. RESULTS: Three N-methyltransferases, SiCNMT1, SiCNMT2, SiCNMT3, and O-methyltransferase SiSOMT were identified in Stephania intermedia. Then, four methyltransferase genes were cloned into the pGEX-6P-1 vector. The recombinant vectors were transformed into Escherichia coli BL21(DE3) for expression and were functionally tested. SiCNMT1, SiCNMT2, and SiCNMT3 could methylate (R)-coclaurine to produce (R)-N-methylcoclaurine. SiCNMT2 further methylated the product of (R)-N-methylcoclaurine to produce (R)-magnocurarine. Similarly, (R)-norcoclaurine was continuously catalyzed to yield (R)-N-methylnorcoclaurine and (R)-N, N-dimethylnorcoclaurine by SiCNMT2. Furthermore, SiSOMT was shown to catalyze the conversion of (S)-scoulerine to (S)-tetrahydropalmatine. CONCLUSIONS: The key methyltransferases, which were in the last step biosynthesis of (R)-magnocurarine, (R)-N, N-dimethylnorcoclaurine and (S)-tetrahydropalmatine were revealed and their activities were verified in vitro. Four novel methyltransferases will be promising candidates for methylation of benzylisoquinoline alkaloids.
A single residue determines substrate preference in benzylisoquinoline alkaloid N-methyltransferases.[Pubmed:31765874]
Phytochemistry. 2020 Feb;170:112193.
N-methylation is a recurring feature in the biosynthesis of many plant specialized metabolites, including alkaloids. A crucial step in the conserved central pathway that provides intermediates for the biosynthesis of benzylisoquinoline alkaloids (BIAs) involves conversion of the secondary amine (S)-coclaurine into the tertiary amine (S)-N-methylcoclaurine by coclaurine N-methyltransferase (CNMT). Subsequent enzymatic steps yield the core intermediate (S)-reticuline, from which various branch pathways for the biosynthesis of major BIAs such as morphine, noscapine and sanguinarine diverge. An additional N-methylation yielding quaternary BIAs is catalyzed by reticuline N-methyltransferase (RNMT), such as in the branch pathway leading to the taxonomically widespread and ecologically significant alkaloid magnoflorine. Despite their functional differences, analysis of primary sequence information has been unable to accurately distinguish between CNMT-like and RNMT-like enzymes, necessitating laborious in vitro screening. Furthermore, despite a recent emphasis on structural characterization of BIA NMTs, the features and mechanisms underlying the CNMT-RNMT functional dichotomy were unknown. We report the identification of structural variants tightly correlated with function in known BIA NMTs and show through reciprocal mutagenesis that a single residue acts as a switch between CNMT- and RNMT-like functions. We use yeast in vivo screening to show that this discovery allows for accurate prediction of activity strictly from primary sequence information and, on this basis, improve the annotation of previously reported putative BIA NMTs. Our results highlight the unusually short mutational distance separating ancestral CNMT-like enzymes from more evolutionarily advanced RNMT-like enzymes, and thus help explain the widespread yet sporadic occurrence of quaternary BIAs in plants. While this is the first report of structural variants controlling mono-versus di-methylation activity among plant NMT enzymes, comparison with bacterial MT enzymes also suggests possible convergent evolution.
Investigation of benzylisoquinoline alkaloid biosynthetic pathway and its transcriptional regulation in lotus.[Pubmed:29872534]
Hortic Res. 2018 Jun 1;5:29.
Lotus predominantly accumulates benzylisoquinoline alkaloids (BIAs), but their biosynthesis and regulation remain unclear. Here, we investigated structural and regulatory genes involved in BIA accumulation in lotus. Two clustered CYP80 genes were identified to be responsible for the biosynthesis of bis-BIAs and aporphine-type BIAs, respectively, and their tissue-specific expression causes divergence in alkaloid component between leaf and embryo. In contrast with the common (S)-reticuline precursor for most BIAs, aporphine alkaloids in lotus leaf may result from the (S)-N-methylcoclaurine precursor. Structural diversity of BIA alkaloids in the leaf is attributed to enzymatic modifications, including intramolecular C-C phenol coupling on ring A and methylation and demethylation at certain positions. Additionally, most BIA biosynthetic pathway genes show higher levels of expression in the leaf of high-BIA cultivar compared with low-BIA cultivar, suggesting transcriptional regulation of BIA accumulation in lotus. Five transcription factors, including three MYBs, one ethylene-responsive factor, and one basic helix-loop-helix (bHLH), were identified to be candidate regulators of BIA biosynthesis in lotus. Our study reveals a BIA biosynthetic pathway and its transcriptional regulation in lotus, which will enable a deeper understanding of BIA biosynthesis in plants.
[Studies on the chemical components of Nelumbinis Plumula and the inhibitory activity on protein disulfide isomerase].[Pubmed:29139271]
Zhongguo Zhong Yao Za Zhi. 2017 Aug;42(15):3004-3010.
Increasing evidence suggested that protein disulfide isomerase supported the survival and progression of several cancers. Nelumbinis Plumula is a Chinese traditional herb which showed antitumor activity. To find if the Nelumbinis Plumula affect protein disulfide isomerase activity, we studied its chemical constituents, and 12 monomeric compounds were isolated by means of solvent extraction, silica gel column chromatography, preparative HPLC and recrystallization. Among them, N-methylcoclaurine, kaempferol, chrysoeriol-7-O-neohesperidoside and mannitol were obtained for the first time. Following, we tested the compounds inhibitory activity on protein disulfide isomerase. The results showed that N-methylcoclaurine, neferine, liensinine and isoliensinine could inhibit the activity of protein disulfide isomerase in vitro, their IC(5)(0) values were 1.4, 2.9, 4.0 and 5.4 mumol*L(-)(1), respectively.
Computer-Aided (13)C NMR Chemical Profiling of Crude Natural Extracts without Fractionation.[Pubmed:28414230]
J Nat Prod. 2017 May 26;80(5):1387-1396.
A computer-aided, (13)C NMR-based dereplication method is presented for the chemical profiling of natural extracts without any fractionation. An algorithm was developed in order to compare the (13)C NMR chemical shifts obtained from a single routine spectrum with a set of predicted NMR data stored in a natural metabolite database. The algorithm evaluates the quality of the matching between experimental and predicted data by calculating a score function and returns the list of metabolites that are most likely to be present in the studied extract. The proof of principle of the method is demonstrated on a crude alkaloid extract obtained from the leaves of Peumus boldus, resulting in the identification of eight alkaloids, including isocorydine, rogersine, boldine, reticuline, coclaurine, laurotetanine, N-methylcoclaurine, and norisocorydine, as well as three monoterpenes, namely, p-cymene, eucalyptol, and alpha-terpinene. The results were compared to those obtained with other methods, either involving a fractionation step before the chemical profiling process or using mass spectrometry detection in the infusion mode or coupled to gas chromatography.
Digital Gene Expression Analysis Provides Insight into the Transcript Profile of the Genes Involved in Aporphine Alkaloid Biosynthesis in Lotus (Nelumbo nucifera).[Pubmed:28197160]
Front Plant Sci. 2017 Jan 31;8:80.
The predominant alkaloids in lotus leaves are aporphine alkaloids. These are the most important active components and have many pharmacological properties, but little is known about their biosynthesis. We used digital gene expression (DGE) technology to identify differentially-expressed genes (DEGs) between two lotus cultivars with different alkaloid contents at four leaf development stages. We also predicted potential genes involved in aporphine alkaloid biosynthesis by weighted gene co-expression network analysis (WGCNA). Approximately 335 billion nucleotides were generated; and 94% of which were aligned against the reference genome. Of 22 thousand expressed genes, 19,000 were differentially expressed between the two cultivars at the four stages. Gene Ontology (GO) enrichment analysis revealed that catalytic activity and oxidoreductase activity were enriched significantly in most pairwise comparisons. In Kyoto Encyclopedia of Genes and Genomes (KEGG) analysis, dozens of DEGs were assigned to the categories of biosynthesis of secondary metabolites, isoquinoline alkaloid biosynthesis, and flavonoid biosynthesis. The genes encoding norcoclaurine synthase (NCS), norcoclaurine 6-O-methyltransferase (6OMT), coclaurine N-methyltransferase (CNMT), N-methylcoclaurine 3'-hydroxylase (NMCH), and 3'-hydroxy-N-methylcoclaurine 4'-O-methyltransferase (4'OMT) in the common pathways of benzylisoquinoline alkaloid biosynthesis and the ones encoding corytuberine synthase (CTS) in aporphine alkaloid biosynthetic pathway, which have been characterized in other plants, were identified in lotus. These genes had positive effects on alkaloid content, albeit with phenotypic lag. The WGCNA of DEGs revealed that one network module was associated with the dynamic change of alkaloid content. Eleven genes encoding proteins with methyltransferase, oxidoreductase and CYP450 activities were identified. These were surmised to be genes involved in aporphine alkaloid biosynthesis. This transcriptomic database provides new directions for future studies on clarifying the aporphine alkaloid pathway.
Quantitative Determination of Alkaloids in Lotus Flower (Flower Buds of Nelumbo nucifera) and Their Melanogenesis Inhibitory Activity.[Pubmed:27447599]
Molecules. 2016 Jul 19;21(7). pii: molecules21070930.
A quantitative analytical method for five aporphine alkaloids, nuciferine (1), nornuciferine (2), N-methylasimilobine (3), asimilobine (4), and pronuciferine (5), and five benzylisoquinoline alkaloids, armepavine (6), norarmepavine (7), N-methylcoclaurine (8), coclaurine (9), and norjuziphine (10), identified as the constituents responsible for the melanogenesis inhibitory activity of the extracts of lotus flowers (the flower buds of Nelumbo nucifera), has been developed using liquid chromatography-mass spectrometry. The optimum conditions for separation and detection of these 10 alkaloids were achieved on a piNAP column, a reversed-phase column with naphthylethyl group-bonded silica packing material, with CH(3)CN-0.2% aqueous acetic acid as the mobile phase and using mass spectrometry equipped with a positive-mode electrospray ionization source. According to the protocol established, distributions of these 10 alkaloids in the petal, receptacle, and stamen parts, which were separated from the whole flower, were examined. As expected, excellent correlations were observed between the total alkaloid content and melanogenesis inhibitory activity. Among the active alkaloids, nornuciferine (2) was found to give a carbamate salt (2'') via formation of an unstable carbamic acid (2') by absorption of carbon dioxide from the air.
Functional Characterization of 4'OMT and 7OMT Genes in BIA Biosynthesis.[Pubmed:26909086]
Front Plant Sci. 2016 Feb 16;7:98.
Alkaloids are diverse group of secondary metabolites generally found in plants. Opium poppy (Papaver somniferum L.), the only commercial source of morphinan alkaloids, has been used as a medicinal plant since ancient times. It produces benzylisoquinoline alkaloids (BIA) including the narcotic analgesic morphine, the muscle relaxant papaverine, and the anti-cancer agent noscapine. Though BIAs play crucial roles in many biological mechanisms their steps in biosynthesis and the responsible genes remain to be revealed. In this study, expressions of 3-hydroxy-N-methylcoclaurine 4'-methyltransferase (4'OMT) and reticuline 7-O-methyltransferase (7OMT) genes were subjected to manipulation to functionally characterize their roles in BIA biosynthesis. Measurements of alkaloid accumulation were performed in leaf, stem, and capsule tissues accordingly. Suppression of 4'OMT expression caused reduction in the total alkaloid content in stem tissue whereas total alkaloid content was significantly induced in the capsule. Silencing of the 7OMT gene also caused repression in total alkaloid content in the stem. On the other hand, over-expression of 4'OMT and 7OMT resulted in higher morphine accumulation in the stem but suppressed amount in the capsule. Moreover, differential expression in several BIA synthesis genes (CNMT, TYDC, 6OMT, SAT, COR, 4'OMT, and 7OMT) were observed upon manipulation of 4'OMT and 7OMT expression. Upon silencing and overexpression applications, tissue specific effects of these genes were identified. Manipulation of 4'OMT and 7OMT genes caused differentiated accumulation of BIAs including morphine and noscapine in capsule and stem tissues.
In vitro opioid receptor affinity and in vivo behavioral studies of Nelumbo nucifera flower.[Pubmed:26260436]
J Ethnopharmacol. 2015 Nov 4;174:57-65.
ETHNOPHARMACOLOGICAL RELEVANCE: Nelumbo nucifera Geartn., known as sacred lotus, has been used traditionally in South East Asia as a traditional medicine for various CNS disorders including stress, fever, depression, insomnia, and cognitive conditions. AIM OF THE STUDY: To investigate the in vitro cannabinoid and opioid receptor binding affinities, and in vivo behavioral actions of Nelumbo flower extracts and to isolate the potential compounds to treat CNS associated disorders. MATERIALS AND METHODS: The white and pink flowers of N. nucifera were extracted with 95% EtOH, followed by acid-base partitioning using CHCl3 to give acidic and basic partitions. These partitions were subjected to Centrifugal Preparative TLC (CPTLC) to yield benzyltetrahydroisoquinoline (BTIQ) alkaloids and long chain fatty acids, identified by physical and spectroscopic methods. In addition, EtOH extracts and partitions were analyzed for chemical markers by UHPLC/MS and GC/MS. In vitro neuropharmacological effects were evaluated by cannabinoid (CB1 and CB2) and opioid [delta (delta), kappa (k), and mu (micro)] competitive radioligand binding and GTPgammaS functional assays. The in vivo behavioral effect was studied through the use of the mouse tetrad assay at 10, 30, 75 and 100mg/kg/ip doses that revealed the effect on locomotion, catalepsy, body temperature, and nociception of acidic and basic CHCl3 partitions, fractions, and compounds. RESULTS: Three aporphines, nuciferine (1), N-nor-nuciferine (2), asimilobine (3), and five BTIQs, armepavine (4), O-methylcoclaurine (5), N-methylcoclaurine (6), coclaurine (7), neferine (10), and a mixture of linoleic and palmitic acids (LA and PA), were identified and evaluated for cannabinoid and opioid receptor displacement activities. Compounds 5-7 showed binding affinities for the k opioid receptor with equilibrium dissociation constant (Ki) values of 3.5 +/- 0.3, 0.9 +/- 0.1, 2.2 +/- 0.2 muM, respectively. Compound 10 displayed affinities for delta-and micro- opioid receptors with Ki values of 0.7 +/- 0.1 and 1.8 +/- 0.2 muM, respectively, and was determined to be a weak delta agonist by GTPgammaS functional assay. The mixture of LA and PA (1:1) showed an affinity for delta opioid receptor with a Ki value of 9.2 +/- 1.1 muM. The acidic and basic CHCl3 partitions, compounds 1 and 7, and 5-7 mixture were subjected to the tetrad assay, of which the acidic partition displayed decreased locomotion and increased catalepsy, antinociception, and hypothermia in animal at doses of 75-100 mg/kg/ip, and also showed clonic-tonic seizures upon touch at 100mg/kg. CONCLUSION: Bioassay-guided isolation revealed compounds 5-7, 10, and the mixture of LA and PA displayed various degrees of opioid receptor radioligand displacement affinities. The in vivo tetrad assay of acidic CHCl3 partition, enriched with aporphines 1 and 2, displayed actions on all four points of behavioral parameters. It can be concluded that the in vivo mild canabimimetic-type effect observed for the CHCl3 partition is likely mediated through other CNS mechanisms since the extracts, partitions, and isolated compounds had no affinity for the in vitro CB1 and CB2 receptors. This work, along with traditional use and the reported bioactivities of the BTIQ alkaloids, suggested further studies on N. nucifera are needed to understand the roles that the extracts and/or individual compounds might contribute to the behavioral effects.
Alkaloids from Peumus boldus and their acetylcholinesterase, butyrylcholinesterase and prolyl oligopeptidase inhibition activity.[Pubmed:25973480]
Nat Prod Commun. 2015 Apr;10(4):577-80.
Eleven isoquinoline alkaloids (1-11) were isolated from dried leaves of Peumus boldus Mol. by standard chromatographic methods. The chemical structures were elucidated by MS, and 1D and 2D NMR spectroscopic analysis, and by comparison with literature data. Compounds isolated in sufficient amount were evaluated for their acetylcholinesterase, and butyrylcholinesterase inhibition activity using Ellman's method. In the prolyl oligopeptidase assay, Z-Gly-Pro-p-nitroanilide was used as substrate. Promising butyrylcholinesterase inhibition activities were demonstrated by two benzylisoquinoline alkaloids, reticuline (8) and N-methylcoclaurine (9), with IC50 values of 33.6 +/- 3.0 microM and 15.0 +/- 1.4 microM, respectively. Important prolyl oligopeptidase inhibition activities were shown by N-methyllaurotetanine (6) and sinoacutine (4) with IC50 values of 135.4 +/- 23.2 microM and 143.1 +/- 25.4 microM, respectively. Other tested compounds were considered inactive.
[Cytotoxic alkaloids from stems of Nelumbo nucifera].[Pubmed:24791498]
Zhongguo Zhong Yao Za Zhi. 2013 Dec;38(23):4104-8.
Chemical investigation was carried out to study the alkaloids from stems of Nelumbo nucifera and their cytotoxic activities. The constituents were separated by column chromatography, and their structures were elucidated by spectroscopic data analyses. The isolated compounds were evaluated for their cytotoxic activities by MTr method. Fifteen compounds were isolated from the total alkaloids extract and identified as asimilobine (1), isococlaurine (2), N-acetylnorarmepavine (3), crykonisine (4), velucryptine (5), pycnarrhine (6), liriodenine (7), nuciferine (8), nornuciferine (9), armepavine (10), N-methylasimilobine (11), coclaurine (12), N-norarmepavine (13), N-methylcoclaurine (14) and lysicamine (15). Compounds 1-7 and 12-15 were isolated from stems of this plant for the first time, and compounds 2-6 were firstly isolated from the family Nelumbonaceae. Compounds 7-10, 13 and 14 showed significant cytotoxic activities against HL-60 carcinoma cell line with inhibitory ratios of 51.36%, 59.09%, 52.51%, 53.93%, 51.43%, and 64.31% at concentration of 1 x 10(-5) mol x L(-1), respectively.
Comparative transcriptome analysis using high papaverine mutant of Papaver somniferum reveals pathway and uncharacterized steps of papaverine biosynthesis.[Pubmed:23738019]
PLoS One. 2013 May 30;8(5):e65622.
The benzylisoquinoline alkaloid papaverine, synthesized in low amount in most of the opium poppy varieties of Papaver somniferum, is used as a vasodilator muscle relaxant and antispasmodic. Papaverine biosynthesis remains controversial as two different routes utilizing either (S)-coclaurine or (S)-reticuline have been proposed with uncharacterized intermediate steps. In an attempt to elucidate papaverine biosynthesis and identify putative genes involved in uncharacterized steps, we carried out comparative transcriptome analysis of high papaverine mutant (pap1) and normal cultivar (BR086) of P. somniferum. This natural mutant synthesizes more than 12-fold papaverine in comparison to BR086. We established more than 238 Mb transcriptome data separately for pap1 and BR086. Assembly of reads generated 127,342 and 106,128 unigenes in pap1 and BR086, respectively. Digital gene expression analysis of transcriptomes revealed 3,336 differentially expressing unigenes. Enhanced expression of (S)-norcoclaurine-6-O-methyltransferase (6OMT), (S)-3'-hydroxy-N-methylcoclaurine 4'-O-methyltransferase (4'OMT), norreticuline 7-O-methyltransferase (N7OMT) and down-regulation of reticuline 7-O-methyltransferase (7OMT) in pap1 in comparison to BR086 suggest (S)-coclaurine as the route for papaverine biosynthesis. We also identified several methyltransferases and dehydrogenases with enhanced expression in pap1 in comparison to BR086. Our analysis using natural mutant, pap1, concludes that (S)-coclaurine is the branch-point intermediate and preferred route for papaverine biosynthesis. Differentially expressing methyltransferases and dehydrogenases identified in this study will help in elucidating complete biosynthetic pathway of papaverine. The information generated will be helpful in developing strategies for enhanced biosynthesis of papaverine through biotechnological approaches.
Genetic and chemical components analysis of Papaver setigerum naturalized in Korea.[Pubmed:22921420]
Forensic Sci Int. 2012 Oct 10;222(1-3):387-93.
Of the 110 species of genus Papaver, only Papaver somniferum and P. setigerum are controlled poppies in Korea. All poppy samples share similar morphology therefore it is important to check if they contain controlled substances such as morphine and codeine for forensic purpose. Since the alkaloid content of Papaver plants varies according to their growing stage, chemical components analysis alone is not enough to identify exact species. In 2010, hundreds of poppy plants suspected to be P. somniferum were found in Jeju Island, South Korea. They had a slightly different but overall similar appearance to P. somniferum. Using GC-MS analysis, codeine, rhoeadine, papaverine, protopine, noscapine, setigeridine and trace amounts of morphine were detected in these samples. Although their chemical components were different from what has been described in literatures for P. setigerum, they could be assumed to be P. setigerum based on their morphological features and GC-MS results. Also, chromosome numbers using their seeds showed 2n=44 and the numbers were in accordance with those of P. setigerum. Nucleotide substitution or insertion/deletion of ITS (internal transcribed spacer), 18S rRNA (ribosomal RNA), rbcL (large subunit of ribulose 1,5-bisphosphate carboxylase), trnL-trnF IGS (intergenic spacer), trnL intron and psbA-trnH were assessed as universal genetic markers for P. setigerum. Also, genetic analysis using six target genes involved in the biosynthesis of benzylisoquinoline alkaloids, including TYDC (tyrosine/dopa decarboxylase), SAT (salutaridinol-7-O-acetyltransferase), BBE (berberine bridge enzyme), COR (codeinone reductase), CYP80B1 ((S)-N-methylcoclaurine 3'-hydroxylase) and NCS (norcoclaurine synthase) were tested as Papaver-specific genetic markers by the existence of their PCR products. From the results, the sequences of the 6 universal genetic markers and 6 Papaver-specific genetic markers for P. setigerum were identified and then Genbank accession numbers of them were registered in NCBI. Also, the trnL intron and psbA-trnH nucleic acid sequences of the 7 Papaver species were identified and registered.


