Poria cocos
Poria cocos
1. The products in our compound library are selected from thousands of unique natural products; 2. It has the characteristics of diverse structure, diverse sources and wide coverage of activities; 3. Provide information on the activity of products from major journals, patents and research reports around the world, providing theoretical direction and research basis for further research and screening; 4. Free combination according to the type, source, target and disease of natural product; 5. The compound powder is placed in a covered tube and then discharged into a 10 x 10 cryostat; 6. Transport in ice pack or dry ice pack. Please store it at -20 °C as soon as possible after receiving the product, and use it as soon as possible after opening.

Natural products/compounds from Poria cocos
- Cat.No. Product Name CAS Number COA
-
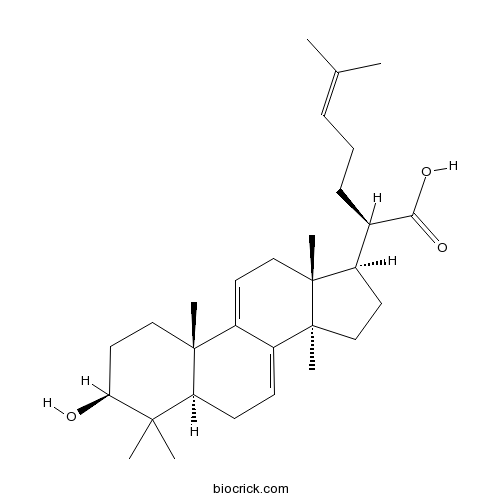
BCN3741
Poricoic acid A(F)137551-38-3
Instructions

-
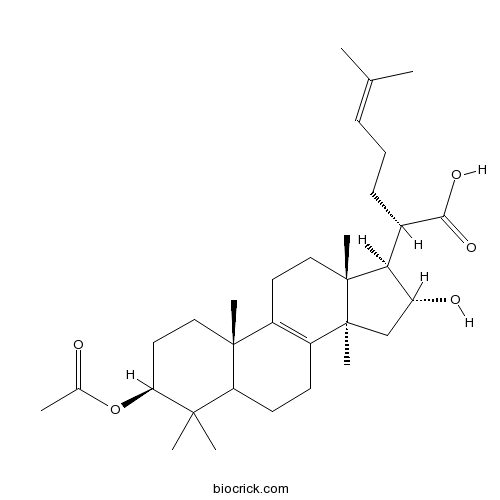
BCN1532
3-O-Acetyl-16 alpha-hydroxytrametenolic acid168293-13-8
Instructions
-
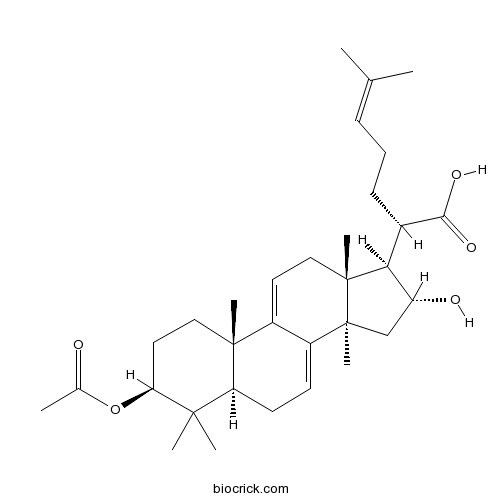
BCN1531
3-O-Acetyl-16 alpha-hydroxydehydrotrametenolic acid168293-14-9
Instructions
-
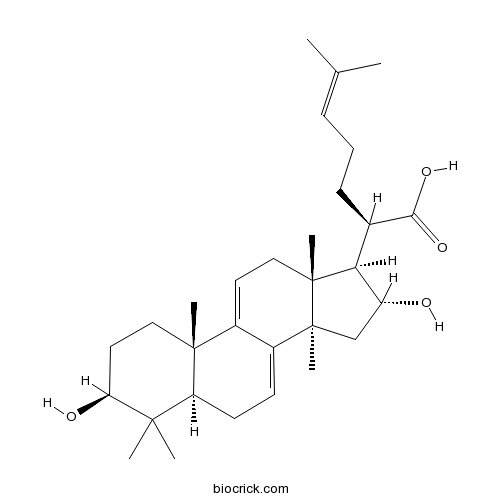
BCN1523
16alpha-Hydroxydehydrotrametenolic acid176390-66-2
Instructions
-
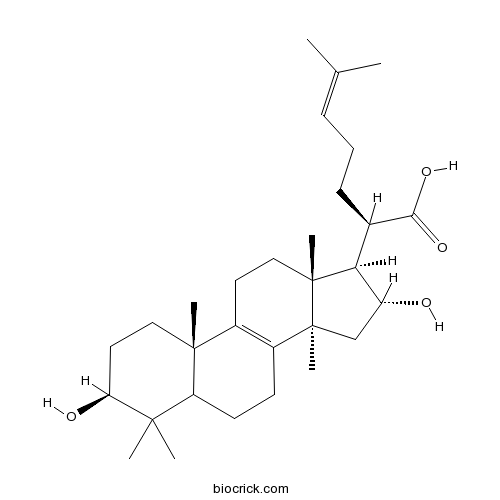
BCN2917
16 alpha-Hydroxytrametenolic acid176390-68-4
Instructions
-
BCN6347
Pachymic acid29070-92-6
Instructions

-
BCN2718
Dehydrotrametenolic acid29220-16-4
Instructions
-
BCN3645
Polyporenic acid C465-18-9
Instructions

-
BCN5654
Hesperidin520-26-3
Instructions

-
BCN3740
Dehydrotumulosic acid6754-16-1
Instructions

-
BCN3646
Dehydroeburicoic acid6879-05-6
Instructions

-
BCN5459
Nerolidol7212-44-4
Instructions

-
BCN3648
Dehydropachymic acid77012-31-8
Instructions

Bioactive compounds from sclerotia extract of Poria cocos that control adipocyte and osteoblast differentiation.[Pubmed: 30092384]
Poria cocos Wolf confers edible sclerotia also known as 'Indian bread' in North America, that have been used for the treatment of various diseases in Asian countries. As part of our ongoing aim to identify biologically new metabolites from Korean edible mushrooms, we investigated the ethanol (EtOH) extract of the sclerotia of P. cocos by applying a comparative LC/MS- and bioassay-based analysis approach, since the EtOH extract reciprocally regulated adipocyte and osteoblast differentiation in mouse mesenchymal stem cells (MSCs). Bioassay-based analysis of the EtOH extract led to the successful isolation of two sterols, ergosterol peroxide (1) and 9,11-dehydroergosterol peroxide (2); three diterpenes, dehydroabietic acid (3), 7-oxocallitrisic acid, (4) and pimaric acid (5); and two triterpenes, dehydroeburicoic acid monoacetate (6) and eburicoic acid acetate (7) from the active hexane-soluble fraction. The isolated compounds (1-7) were examined for their effects on the regulation of MSC differentiation. The two sterols (1 and 2) were able to suppress MSC differentiation toward adipocytes. In contrast, the three diterpenes (3-5) showed activity to promote osteogenic differentiation of MSC. These findings demonstrate that the EtOH extract of P. cocos sclerotia is worth consideration as a new potential source of bioactive compounds effective in the treatment of osteoporosis in the elderly, since the extract contains sterols that inhibit adipogenic differentiation as well as diterpenes that promote osteogenic differentiation from MSCs.
Transcriptome analysis of three cultivars of Poria cocos reveals genes related to the biosynthesis of polysaccharides.[Pubmed: 30010416]
Poria cocos (P. cocos) polysaccharides (PCPs) are used to improve immunity and possess antitumor activities. We compared three cultivars of P. cocos (5.78, XJ 28 and JHYH) PCP contents. Then we determined that malZ, galA, SORD, gnl and bglX are key enzymes within the PCP biosynthetic pathway by using HiSeq2500 transcriptome and qRT-PCR validation. Our results provide more detailed information about the PCP biosynthesis pathway at the molecular level in P. cocos and establish the functions for the molecular breeding to produce polysaccharides in general for therapeutic use in Chinese medicinal plants.
Targeted Screening Approach to Systematically Identify the Absorbed Effect Substances of Poria cocos in Vivo Using Ultrahigh Performance Liquid Chromatography Tandem Mass Spectrometry.[Pubmed: 29985616]
Poria cocos are extensively used as nutritious food, dietary supplements, and oriental medicine in Asia. However, the effect substances are still not very clear. In this study, a targeted screening approach was developed to systematically identify absorbed constituents of Poria cocos in vivo using ultrahigh performance liquid chromatography tandem mass spectrometry combined with UNIFI software. First, incubation reactions in vitro with rat intestinal microflora and rat liver microsomes were conducted to sum up metabolic rules of main constituents. Second, the absorbed constituents in vivo were picked out and identified based on the results of metabolic study in vitro. Finally, the absorbed active constituents in the treatment of Alzheimer's disease were screened by targeted network pharmacology analysis. A total of 62 absorbed prototypes and 59 metabolites were identified and characterized in dosed plasma. Thirty potential active constituents were screened, and 86 drug-targets shared by absorbed constituents and Alzheimer's disease were discovered by targeted network pharmacology analysis. In general, this proposed targeted strategy comprehensively provides new insight for active ingredients of Poria cocos.
Dangguishaoyao-San attenuates LPS-induced neuroinflammation via the TLRs/NF-κB signaling pathway.[Pubmed: 29857298]
Dangguishaoyao-San (DSS) is composed of six traditional Chinese medicines, including Angelica sinensis, Paeoniae radix, Rhizoma Ligusticum, Poria cocos, Rhizoma Atractylodis Macrocephalae, and Rhizoma Alismatis. DSS has been reported to be effective in alleviating the symptoms of Alzheimer's disease (AD). The aim of this study was to investigate the mechanism of action of DSS in vitro using lipopolysaccharide (LPS)-stimulated BV-2 microglia cells.
Carboxymethyl pachyman (CMP) reduces intestinal mucositis and regulates the intestinal microflora in 5-fluorouracil-treated CT26 tumour-bearing mice.[Pubmed: 29756138]
The compound 5-fluorouracil (5-FU) is the first choice chemotherapeutic agent for the treatment of colorectal cancer (CRC), but intestinal mucositis is a primary limiting factor in anticancer therapy. There is currently no broadly effective targeted treatment to cure this side effect. Carboxymethylated pachyman (CMP) is a polysaccharide that is modified from the structure of pachyman isolated from Poria cocos (Chinese name: Fu Ling). Meanwhile, recent studies have shown that CMP exhibits immune regulatory, anti-inflammatory and antioxidant activities. Therefore, the purpose of this study was to evaluate the intestinal protective effect of CMP in 5-FU-treated CT26 tumour-bearing mice and to further explore its underlying mechanism(s) of action. Initially, a CT26 colon carcinoma xenograft mice model was established. The colon length, colon tissue injury, intestinal flora, short-chain fatty acids (SCFAs) and indicators linked to inflammation, antioxidation and apoptosis were then measured. Our results showed that CMP in combination with 5-FU reversed intestinal shortening (p < 0.01) and alleviated 5-FU-induced colon injury (p < 0.001) via suppression of ROS production; increasing the levels of CAT, GSH-Px and GSH; decreasing expression of NF-κB, p-p38 and Bax; and elevating the levels of Nrf2 and Bcl-2. More importantly, CMP had a significant impact and counteracted the intestinal microflora disorders produced by 5-FU by increasing the proportion of Bacteroidetes, lactobacilli, and butyric acid-producing and acetic acid-producing bacteria and restoring the intestinal flora diversity. Overall, this work suggested that CMP could regulate the ecological balance of the intestinal flora and reduce colon injuries induced by 5-FU in CT26 tumour-bearing mice, and the mechanism involved may be associated with the regulation of the NF-κB, Nrf2-ARE and MAPK/P38 pathways.


