GrandiflorosideCAS# 61186-24-1 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 61186-24-1 | SDF | Download SDF |
| PubChem ID | 20056012 | Appearance | Powder |
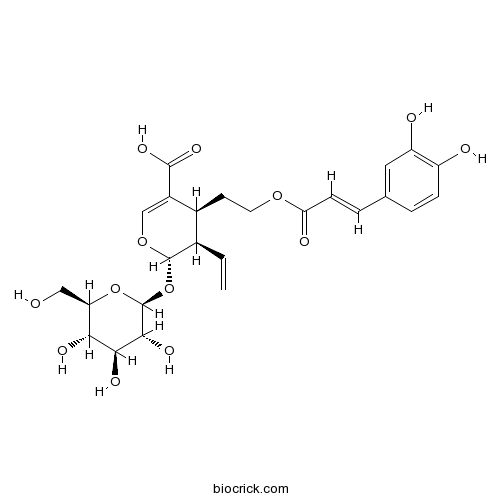
| Formula | C25H30O13 | M.Wt | 538.5 |
| Type of Compound | Iridoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | (2S,3R,4S)-4-[2-[(E)-3-(3,4-dihydroxyphenyl)prop-2-enoyl]oxyethyl]-3-ethenyl-2-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-3,4-dihydro-2H-pyran-5-carboxylic acid | ||
| SMILES | C=CC1C(C(=COC1OC2C(C(C(C(O2)CO)O)O)O)C(=O)O)CCOC(=O)C=CC3=CC(=C(C=C3)O)O | ||
| Standard InChIKey | ZPEFYJBGAZLAKK-IBKSRVHKSA-N | ||
| Standard InChI | InChI=1S/C25H30O13/c1-2-13-14(7-8-35-19(29)6-4-12-3-5-16(27)17(28)9-12)15(23(33)34)11-36-24(13)38-25-22(32)21(31)20(30)18(10-26)37-25/h2-6,9,11,13-14,18,20-22,24-28,30-32H,1,7-8,10H2,(H,33,34)/b6-4+/t13-,14+,18-,20-,21+,22-,24+,25+/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. Grandifloroside shows good antioxidative and potent anti-inflammatory activities, it also exhibits potent inhibitory activity against TNF-α and 5α-reductase, suggests that it might be developed as a source of potent anti-oxidative and anti-inflammatory agents and therapeutic agent for benign prostatic hypertrophy (BPH). |
| Targets | TNF-α | 5-alpha Reductase |

Grandifloroside Dilution Calculator

Grandifloroside Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.857 mL | 9.2851 mL | 18.5701 mL | 37.1402 mL | 46.4253 mL |
| 5 mM | 0.3714 mL | 1.857 mL | 3.714 mL | 7.428 mL | 9.2851 mL |
| 10 mM | 0.1857 mL | 0.9285 mL | 1.857 mL | 3.714 mL | 4.6425 mL |
| 50 mM | 0.0371 mL | 0.1857 mL | 0.3714 mL | 0.7428 mL | 0.9285 mL |
| 100 mM | 0.0186 mL | 0.0929 mL | 0.1857 mL | 0.3714 mL | 0.4643 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 4-(3,4-Dihydroxyphenyl)-2-butanone
Catalog No.:BCN4132
CAS No.:61152-62-3
- 6alpha-Hydroxymedicarpin
Catalog No.:BCN3939
CAS No.:61135-92-0
- 3,9-Dihydroxypterocarpan
Catalog No.:BCN4131
CAS No.:61135-91-9
- Epipterosin L 2'-O-glucoside
Catalog No.:BCN4614
CAS No.:61117-89-3
- Bromhexine hydrochloride
Catalog No.:BCC8898
CAS No.:611-75-6
- (R)-Mandelic acid
Catalog No.:BCN8532
CAS No.:611-71-2
- Tectoridin
Catalog No.:BCN1020
CAS No.:611-40-5
- 4-Phenyl-1,2,3,4-tetrahydroisoquinoline hydrochloride
Catalog No.:BCC6769
CAS No.:6109-35-9
- Icotinib
Catalog No.:BCC4473
CAS No.:610798-31-7
- Isobonducellin
Catalog No.:BCN4130
CAS No.:610778-85-3
- Boc-His(Nτ-Me)-OH
Catalog No.:BCC2684
CAS No.:61070-22-2
- L-Thyroxine sodium salt pentahydrate
Catalog No.:BCC4283
CAS No.:6106-07-6
- Quinine HCl Dihydrate
Catalog No.:BCC4933
CAS No.:6119-47-7
- Uzarigenin digitaloside
Catalog No.:BCN4613
CAS No.:61217-80-9
- 6alpha-Hydroxymaackiain
Catalog No.:BCN3947
CAS No.:61218-44-8
- 11-Hydroxybisabola-1,3,5-trien-9-one
Catalog No.:BCN7530
CAS No.:61235-23-2
- Denudadione C
Catalog No.:BCN6608
CAS No.:61240-34-4
- AZD1080
Catalog No.:BCC4508
CAS No.:612487-72-6
- PTP1B-IN-1
Catalog No.:BCC5506
CAS No.:612530-44-6
- Cannabispiran
Catalog No.:BCN4134
CAS No.:61262-81-5
- Vitexilactone
Catalog No.:BCN4135
CAS No.:61263-49-8
- Acteoside
Catalog No.:BCN4136
CAS No.:61276-17-3
- Boc-D-Phe(4-NO2)-OH
Catalog No.:BCC3276
CAS No.:61280-75-9
- Schizandrin B
Catalog No.:BCN1022
CAS No.:61281-37-6
Triterpene glycosides with stimulatory activity on melanogenesis from the aerial parts of Weigela subsessilis.[Pubmed:25630794]
Arch Pharm Res. 2015 Aug;38(8):1541-51.
Three new triterpene glycosides (Lonicerosides K, L and M) and 11 known compounds were isolated from the aerial parts of Weigela subsessilis. Among the known isolated compounds, loniceroside A, sweroside, kaempferol-3-O-glucopyranoside 6''-(3-hydroxy-3-methylglutarate), kaempferol-3-O-acetylglucoside and Grandifloroside were reported for the first time in a Weigela genus plant. Their chemical structures were identified using extensive spectroscopic analysis including two-dimensional (2D)-NMR experiments, HR-ESI-QTOF-MS and comparison with reported data. Among these compounds, lonicerosides A and L had potent melanogenesis stimulatory activity in murine B16F0 melanoma cells. The structural relationship of active compounds was discussed.
Inhibitory Activities of Phenolic Compounds Isolated from Adina rubella Leaves Against 5alpha-Reductase Associated with Benign Prostatic Hypertrophy.[Pubmed:27399661]
Molecules. 2016 Jul 7;21(7). pii: molecules21070887.
Adina rubella Hance (AR), a plant native to Korea, has been used as traditional medicine for dysentery, eczema, intoxication, and external hemorrhages. Previous phytochemical studies of AR have reported several components, including terpenoids, phenolics, and alkaloids. The current study evaluated the anti-oxidative and anti-inflammatory activities and 5alpha-reductase inhibition of isolated compounds of AR leaves to find a potential therapeutic agent for benign prostatic hypertrophy (BPH). Repeated chromatographic isolation of an 80% acetone extract of AR leaves yielded seven phenolic compounds: caffeic acid (1), chlorogenic acid (2), methyl chlorogenate (3), quercetin-3-rutinoside (4), kaempferol-3-O-alpha-l-rhamnopyranosyl-(1-->6)-beta-d-glucopyranoside (5), hyperoside (6), and Grandifloroside (7). Compound 7 is a novel compound in AR. Caffeoyl derivatives 1-3 and 7 showed good anti-oxidative activities. In particular, caffeic acid (1) and Grandifloroside (7) showed potent anti-inflammatory activities, and 7 also exhibited potent inhibitory activity against TNF-alpha and 5alpha-reductase. Our results show that the extract and Grandifloroside (7) from leaves of AR might be developed as a source of potent anti-oxidative and anti-inflammatory agents and therapeutic agent for BPH.


