Nemonapride5-HT1A agonist. Also highly potent D2-like dopamine antagonist CAS# 75272-39-8 |
- GSK744 (S/GSK1265744)
Catalog No.:BCC3888
CAS No.:1051375-10-0
- Atazanavir
Catalog No.:BCC3622
CAS No.:198904-31-3
- BMS-707035
Catalog No.:BCC2133
CAS No.:729607-74-3
- MK-2048
Catalog No.:BCC2136
CAS No.:869901-69-9
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 75272-39-8 | SDF | Download SDF |
| PubChem ID | 156333 | Appearance | Powder |
| Formula | C21H26ClN3O2 | M.Wt | 387.91 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | YM 09151, YM 09151-2 | ||
| Solubility | Soluble to 20 mM in ethanol and to 25 mM in DMSO | ||
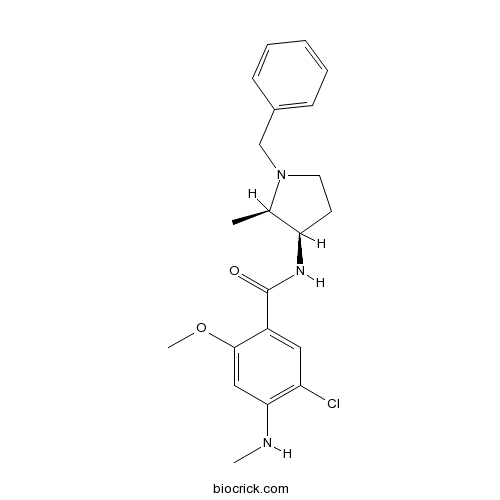
| Chemical Name | N-[(2R,3R)-1-benzyl-2-methylpyrrolidin-3-yl]-5-chloro-2-methoxy-4-(methylamino)benzamide | ||
| SMILES | CC1C(CCN1CC2=CC=CC=C2)NC(=O)C3=CC(=C(C=C3OC)NC)Cl | ||
| Standard InChIKey | KRVOJOCLBAAKSJ-RDTXWAMCSA-N | ||
| Standard InChI | InChI=1S/C21H26ClN3O2/c1-14-18(9-10-25(14)13-15-7-5-4-6-8-15)24-21(26)16-11-17(22)19(23-2)12-20(16)27-3/h4-8,11-12,14,18,23H,9-10,13H2,1-3H3,(H,24,26)/t14-,18-/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Highly potent dopamine D2-like receptor antagonist; selective over D1-like receptors (Ki values are 0.1 and 740 nM for D2-like and D1-like receptors respectively). Also potent 5-HT1A receptor agonist (IC50 = 34 nM) and has affinity for sigma receptors. |

Nemonapride Dilution Calculator

Nemonapride Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.5779 mL | 12.8896 mL | 25.7792 mL | 51.5584 mL | 64.4479 mL |
| 5 mM | 0.5156 mL | 2.5779 mL | 5.1558 mL | 10.3117 mL | 12.8896 mL |
| 10 mM | 0.2578 mL | 1.289 mL | 2.5779 mL | 5.1558 mL | 6.4448 mL |
| 50 mM | 0.0516 mL | 0.2578 mL | 0.5156 mL | 1.0312 mL | 1.289 mL |
| 100 mM | 0.0258 mL | 0.1289 mL | 0.2578 mL | 0.5156 mL | 0.6445 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- H-Trp-OMe.HCl
Catalog No.:BCC3114
CAS No.:7524-52-9
- H-Phe-OMe.HCl
Catalog No.:BCC3009
CAS No.:7524-50-7
- Ophiopogonanone A
Catalog No.:BCN6630
CAS No.:75239-63-3
- TAK-700 R-form
Catalog No.:BCC4203
CAS No.:752243-39-3
- 3,6-Bis(hydroxymethyl)durene
Catalog No.:BCC8598
CAS No.:7522-62-5
- Chikusetsusaponin IV
Catalog No.:BCN2683
CAS No.:7518-22-1
- H-Leu-OMe.HCl
Catalog No.:BCC2973
CAS No.:7517-19-3
- PRE-084 hydrochloride
Catalog No.:BCC6708
CAS No.:75136-54-8
- 2-Acetamido-2-deoxy-D-glucose
Catalog No.:BCC8508
CAS No.:7512-17-6
- Obacunone
Catalog No.:BCN4303
CAS No.:751-03-1
- 2',6'-Dihydroxy-4'-methoxyacetophenone
Catalog No.:BCN6891
CAS No.:7507-89-3
- 20,24-Dihydroxydammar-25-en-3-one
Catalog No.:BCN4302
CAS No.:75069-59-9
- HEPES Sodium salt
Catalog No.:BCC7591
CAS No.:75277-39-3
- Kukoamine A
Catalog No.:BCN3835
CAS No.:75288-96-9
- H-Pro-NH2
Catalog No.:BCC3018
CAS No.:7531-52-4
- H-Leucinol
Catalog No.:BCC2725
CAS No.:7533-40-6
- Lovastatin
Catalog No.:BCN1060
CAS No.:75330-75-5
- Indacaterol Maleate
Catalog No.:BCC4358
CAS No.:753498-25-8
- Boc-Asn-OH
Catalog No.:BCC3071
CAS No.:7536-55-2
- Boc-Asp(OBzl)-OH
Catalog No.:BCC2608
CAS No.:7536-58-5
- EHT 1864
Catalog No.:BCC6075
CAS No.:754240-09-0
- Moxonidine
Catalog No.:BCC2142
CAS No.:75438-57-2
- CGP 37157
Catalog No.:BCC6943
CAS No.:75450-34-9
- N-Methylnuciferine
Catalog No.:BCN3971
CAS No.:754919-24-9
Positron emission tomography and ex vivo and in vitro autoradiography studies on dopamine D2-like receptor degeneration in the quinolinic acid-lesioned rat striatum: comparison of [11C]raclopride, [11C]nemonapride and [11C]N-methylspiperone.[Pubmed:11929700]
Nucl Med Biol. 2002 Apr;29(3):307-16.
With [11C]raclopride,[11C]Nemonapride and [11C]N-methylspiperone, degeneration of dopamine D2-like receptors in the unilaterally quinolinic acid-lesioned rats was evaluated by positron emission tomography (PET) and ex vivo and in vitro autoradiography. PET showed a decreased uptake of [11C]raclopride in the lesioned striatum, but an increased uptake of [11C]Nemonapride and [11C]N-methylspiperone despite a decreased binding in vitro. Ex vivo autoradiography showed an increased accumulation of the three ligands in the cortical region overlying the injured striatum, probably enlarging PET signals. PET has the limited potential for evaluating the receptor degeneration in the present animal model.
3-Aminopyrrolidines from alpha-aminoacids: total synthesis of (+)-nemonapride from D-alanine.[Pubmed:18177051]
J Org Chem. 2008 Feb 1;73(3):1162-4.
The antipsychotic compound Nemonapride 1 was synthesized in nine steps from d-alanine 2. The key steps for the synthesis of the 3-aminopyrrolidine moiety include a Birch reduction of a cyclic enaminoester and the reduction of a pyrrolidinone to the pyrrolidine 7. Final coupling with the benzoic acid derivative 9 gave 1 as a single enantio- and diastereomer.
Differential in vivo inhibition of [3H]nemonapride binding by atypical antipsychotics in rat striatum, olfactory lobes, and frontal cortex.[Pubmed:16020947]
Pharmacology. 2005 Oct;75(2):63-8.
Dopamine D2 receptor blockade is thought to be mandatory for antipsychotic action because most of the currently used antipsychotics have high affinity at these receptors. Here, we examined the in vivo binding characteristics of the D2-like receptor antagonist [3H]Nemonapride in rat brain areas including the striatum, olfactory lobes and frontal cortex and its inhibition by a series of D2 antagonist antipsychotics. In vivo affinity of [3H]Nemonapride was similar (apparent Kd value: 0.05 micromol/kg) in all brain regions examined. The estimated number of binding sites was higher in the striatum (66 fmol/mg wet weight) than in the olfactory lobes (28 fmol/mg wet weight) and the frontal cortex (21 fmol/mg wet weight). In the striatum, [3H]Nemonapride binding was inhibited in a dose-dependent manner with the following order of potency (ED50, mg/kg): Nemonapride (0.04), raclopride (0.13), spiperone and risperidone (0.14), haloperidol (0.21), clozapine (7.2) and thioridazine (9.4); in the olfactory lobes: Nemonapride (0.03), raclopride and spiperone (0.09), haloperidol (0.10), risperidone (0.15), thioridazine and clozapine (11); in the frontal cortex, only the high affinity dopamine D2 antagonist compounds Nemonapride (0.05), haloperidol (0.09), and raclopride (0.12) significantly decreased the binding of [3H]Nemonapride. The present data suggest that conventional and atypical antipsychotics may be distinguished by their differential occupancy of striatal versus frontocortical D2-like receptors in vivo.
Concise, Protecting-Group-Free Synthesis of (+)-Nemonapride via Eu(OTf)3-Catalyzed Aminolysis of 3,4-Epoxy Alcohol.[Pubmed:28049911]
Chem Pharm Bull (Tokyo). 2017;65(1):22-24.
A concise, protecting-group-free synthesis of the antipsychotic agent (+)-Nemonapride has been achieved featuring a europium(III) trifluoromethanesulfonate (Eu(OTf)3)-catalyzed C4 selective aminolysis of a 3,4-epoxy alcohol by benzylamine and an expedient use of the resulting 4-benzylamino-1,3-diol product for constructing the pyrrolidine skeleton.
5-HT1A receptor agonist properties of the antipsychotic, nemonapride: comparison with bromerguride and clozapine.[Pubmed:9369342]
Eur J Pharmacol. 1997 Sep 10;334(2-3):141-7.
5-HT1A receptor agonists are thought to enhance the antipsychotic-like effects of dopamine D2 receptor antagonists while reducing their potential to produce extrapyramidal side effects. Thus, 5-HT1A receptor agonist properties of mixed 5-HT1A receptor agonists/D2 receptor antagonists might be of clinical importance. The antipsychotics, clozapine and Nemonapride, and the putative antipsychotic, bromerguride, have intermediate to high affinity for 5-HT1A receptors. The present study examined the 5-HT1A receptor agonist activity of Nemonapride and bromerguride, in comparison with clozapine, which has partial 5-HT1A receptor agonist properties in vitro. Here, 5-HT1A receptor activation was examined in vitro, by measuring forskolin-stimulated cAMP accumulation in HeLa cells expressing human 5-HT1A receptors, and in vivo, by using microdialysis to measure the extracellular concentration of hippocampal 5-hydroxytryptamine (5-HT) in rats. Nemonapride markedly decreased both forskolin-stimulated cAMP accumulation and the extracellular concentration of 5-HT; both effects were antagonized by the 5-HT1A receptor antagonist, N-[2-[4-(2-methoxyphenyl)-1-piperazinyl]ethyl]-N-(2-pyridinyl) cyclohexanecarboxamide (WAY100635). In contrast, clozapine only partially decreased forskolin-stimulated cAMP accumulation and extracellular 5-HT, and only its effects on cAMP accumulation were attenuated by WAY100635. Bromerguride decreased neither forskolin-stimulated cAMP accumulation nor extracellular 5-HT; instead, it antagonized the decrease of cAMP accumulation produced by 5-HT and the decrease of extracellular 5-HT produced by the 5-HT1A agonist (+/-)-8-hydroxy-2-(di-n-propylamino)tetralin (8-OH-DPAT). The selective D2 receptor antagonist, raclopride, affected neither forskolin-stimulated cAMP in vitro nor extracellular 5-HT in vivo. Thus, in contrast with clozapine and bromerguride, only the novel antipsychotic, Nemonapride, exhibited marked 5-HT1A receptor agonist properties both in vitro and in vivo; conceivably, these properties may play a role in its preclinical and clinical effects.
[3H]YM-09151-2 (nemonapride), a potent radioligand for both sigma 1 and sigma 2 receptor subtypes.[Pubmed:8804051]
Neuroreport. 1996 Apr 10;7(5):1057-61.
Using K+ phosphate buffer with 25 nM spiperone, [3H]YM-09151-2 binding showed a high affinity for sigma receptors but no affinity for D2 dopamine or 5-HT1A receptors in rat brain. The order of pKi values of various sigma compounds at [3H]YM-09151-2 binding sites and stereoisomer selectivity were consistent with previous studies using other sigma ligands such as (+)-[3H]SKF-10047, [3H]DTG and (+)-[3H]3-PPP. Although Scatchard analysis fitted a one-site model, competition between [3H]YM-09151-2 and (+)-pentazocine revealed two sites, sigma 1 and sigma 2 receptors, at which the Ki values of YM-09151-2 were 8.4 nM and 9.6nM, respectively. Autoradiography using [3H]YM-09151-2 also showed a characteristic distribution of sigma receptors in rat brain. [3H]YM-09151-2 is, therefore, a potent and useful radioligand for sigma 1/sigma 2 receptor subtypes.
Selective binding of YM-09151-2, a new potent neuroleptic, to D2-dopaminergic receptors.[Pubmed:6138453]
Jpn J Pharmacol. 1983 Aug;33(4):749-55.
Effects of YM-09151-2 and five other neuroleptics (haloperidol, spiperone, chlorpromazine, sulpiride and clozapine) on the binding of [3H]-ligands to nine different receptors (alpha 1-adrenergic, alpha 2-adrenergic, beta-adrenergic, muscarinic, D2-dopaminergic, H1-histaminergic, 5HT1-serotonergic, 5HT2-serotonergic and opiate receptors) and on dopamine-sensitive adenylate cyclase were determined using brain membranes in the rat, guinea-pig and dog. The affinity of YM-09151-2 for D2-receptors with a Ki value of 0.1 nM was more than 1000-times higher than that for the other receptors and dopamine-sensitive adenylate cyclase, and it was the greatest among the neuroleptics tested.


