(S)-(+)-CarvoneCAS# 2244-16-8 |
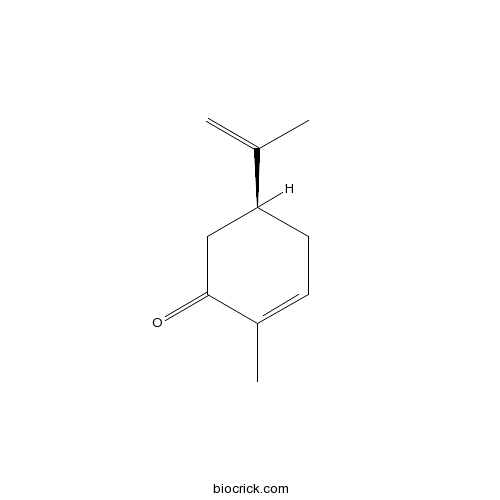
- 2-Methyl-5-Isopropenyl-2-Cyclohexenone
Catalog No.:BCC8279
CAS No.:99-49-0
- (-)-Carvone
Catalog No.:BCN8949
CAS No.:6485-40-1
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 2244-16-8 | SDF | Download SDF |
| PubChem ID | 16724 | Appearance | Light yellow clear liquid |
| Formula | C10H14O | M.Wt | 150.22 |
| Type of Compound | Monoterpenoids | Storage | Desiccate at -20°C |
| Synonyms | (+)-p-Mentha 6,8-diene 2-one | ||
| Solubility | Soluble in ethan | ||
| Chemical Name | (5S)-2-methyl-5-prop-1-en-2-ylcyclohex-2-en-1-one | ||
| SMILES | CC1=CCC(CC1=O)C(=C)C | ||
| Standard InChIKey | ULDHMXUKGWMISQ-VIFPVBQESA-N | ||
| Standard InChI | InChI=1S/C10H14O/c1-7(2)9-5-4-8(3)10(11)6-9/h4,9H,1,5-6H2,2-3H3/t9-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

(S)-(+)-Carvone Dilution Calculator

(S)-(+)-Carvone Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 6.6569 mL | 33.2845 mL | 66.569 mL | 133.1381 mL | 166.4226 mL |
| 5 mM | 1.3314 mL | 6.6569 mL | 13.3138 mL | 26.6276 mL | 33.2845 mL |
| 10 mM | 0.6657 mL | 3.3285 mL | 6.6569 mL | 13.3138 mL | 16.6423 mL |
| 50 mM | 0.1331 mL | 0.6657 mL | 1.3314 mL | 2.6628 mL | 3.3285 mL |
| 100 mM | 0.0666 mL | 0.3328 mL | 0.6657 mL | 1.3314 mL | 1.6642 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- (1S)-(-)-α-Pinene
Catalog No.:BCN9076
CAS No.:7785-26-4
- (-)-Fenchone
Catalog No.:BCN9075
CAS No.:7787-20-4
- 3-(β-D-Glucopyranosyloxy)-1,6-dihydroxy-2-methyl-9,10-anthracenedione
Catalog No.:BCN9074
CAS No.:125906-49-2
- Ferulic Acid Methyl Ester
Catalog No.:BCN9073
CAS No.:2309-07-1
- Alphalipoic acid
Catalog No.:BCN9072
CAS No.:1077-28-7
- (+)-Fenchone
Catalog No.:BCN9071
CAS No.:4695-62-9
- (-)-Menthone
Catalog No.:BCN9070
CAS No.:14073-97-3
- Benzyl alcohol
Catalog No.:BCN9069
CAS No.:100-51-6
- Citronellal
Catalog No.:BCN9068
CAS No.:106-23-0
- 10-hydroxydec-2-enoic acid
Catalog No.:BCN9067
CAS No.:14113-05-4
- Citral
Catalog No.:BCN9066
CAS No.:5392-40-5
- Eugenol acetate
Catalog No.:BCN9065
CAS No.:93-28-7
- Sodium taurocholate
Catalog No.:BCN9078
CAS No.:145-42-6
- Sodium deoxycholate
Catalog No.:BCN9079
CAS No.:302-95-4
- Lanosta-8,20(22)-dien-26-oic acid, 15-hydroxy-3,11,23-trioxo-, (15α,20Z)-
Catalog No.:BCN9080
CAS No.:1961358-01-9
- Chol-8-en-24-oic acid, 7,15-dihydroxy-4,4,14-trimethyl-3,11-dioxo-, (5α)-
Catalog No.:BCN9081
CAS No.:942936-54-1
- Calenduloside E
Catalog No.:BCN9082
CAS No.:26020-14-4
- (3β,7β,12β,20Z )- 3,7,12- trihydroxy-11,15,23-trioxo-lanost-8,20-dien-26-oic acid
Catalog No.:BCN9083
CAS No.:1961358-02-0
- (-)-Sesamin
Catalog No.:BCN9084
CAS No.:13079-95-3
- (3r)-7,2'-Dihydroxy-3',4'-dimethoxyisoflavan
Catalog No.:BCN9085
CAS No.:64474-51-7
- Cucurbitacin Q1
Catalog No.:BCN9086
CAS No.:99530-82-2
- Cyclohexanecarboxylic acid, 3-[[(2E)-3-[4-(D-glucopyranosyloxy)-3-hydroxyphenyl]-1-oxo-2-propen-1-yl...
Catalog No.:BCN9087
CAS No.:1629852-63-6
- (-)-Isobicyclogermacrenal
Catalog No.:BCN9088
CAS No.:73256-82-3
- Delphinidin 3-O-galactoside
Catalog No.:BCN9089
CAS No.:197250-28-5
Standardised comparison of limonene-derived monoterpenes identifies structural determinants of anti-inflammatory activity.[Pubmed:32350292]
Sci Rep. 2020 Apr 29;10(1):7199.
Mint species are widely used in traditional and conventional medicine as topical analgesics for osteoarthritic pain and for disorders of the gastrointestinal and respiratory tracts which are all associated with chronic inflammation. To identify the structural determinants of anti-inflammatory activity and potency which are required for chemical optimization towards development of new anti-inflammatory drugs, a selected group of monoterpenes especially abundant in mint species was screened by measuring bacterial lipopolysacharide (LPS)-induced nitric oxide (NO) production in murine macrophages. Nine compounds significantly decreased LPS-induced NO production by more than 30%. IC50 values were calculated showing that the order of potency is: (S)-(+)-Carvone > (R)-(-)-carvone > (+)-dihydrocarveol > (S)-8-hydroxycarvotanacetone > (R)-8-hydroxycarvotanacetone > (+)-dihydrocarvone > (-)-carveol > (-)-dihydrocarveol > (S)-(-)-pulegone. Considering the carbon numbering relative to the common precursor, limonene, the presence of an oxygenated group at C6 conjugated to a double bond at C1 and an isopropenyl group and S configuration at C4 are the major chemical features relevant for activity and potency. The most potent compound, (S)-(+)-Carvone, significantly decreased the expression of NOS2 and IL-1beta in macrophages and in a cell model of osteoarthritis using primary human chondrocytes. (S)-(+)-Carvone may be efficient in halting inflammation-related diseases, like osteoarthritis.
Cloning and Functional Characterization of Three Odorant Receptors From the Chinese Citrus fly Bactrocera minax (Diptera: Tephritidae).[Pubmed:32269531]
Front Physiol. 2020 Mar 25;11:246.
Insect olfactory sensing is crucial for finding food, mating, and oviposition preference. Odorant receptors (ORs) play a central role in the transmission of odorant signals into the environment by the peripheral olfactory system. Therefore, the identification and functional study of ORs are essential to better understand olfactory mechanisms in insects. OR studies on Diptera insects are primarily performed on Drosophila and mosquitoes, but few studies have been reported in Tephritidae. In this study, we examined three candidate ORs (BminOR3, BminOR12, and BminOR16) from Bactrocera minax. Our analysis of tissue expression revealed that the three BminORs were expressed in the antennae, with no difference between the male and female. In in vitro heterologous expression system of Xenopus oocytes. BminOR3/BminOrco responded strongly to 1-octen-3-ol, BminOR12/BminOrco responded to eight compounds [methyl salicylate, benzaldehyde, (Z)-3-hexenyl acetate, butyl acrylate, butyl propionate, 1-octanol, (S)-(+)-Carvone and benzyl alcohol], and BminOR16/BminOrco slightly responded to undecanol. Our results concluded that BminOR3, BimOR12, and BminOR16 could play an important role in host-finding and oviposition positioning in B. minax.


