SanshodiolCAS# 54854-91-0 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 54854-91-0 | SDF | Download SDF |
| PubChem ID | 14237706 | Appearance | Powder |
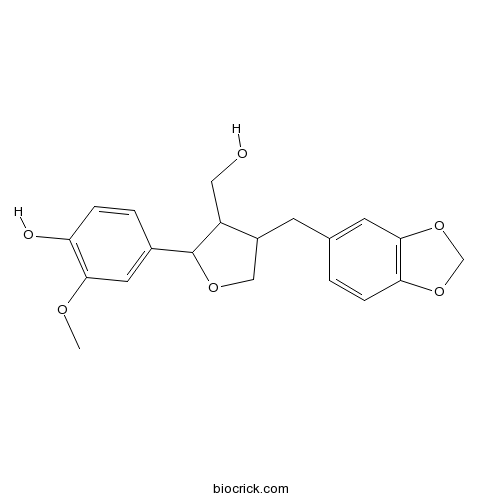
| Formula | C20H22O6 | M.Wt | 358.4 |
| Type of Compound | Lignans | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | 4-[4-(1,3-benzodioxol-5-ylmethyl)-3-(hydroxymethyl)oxolan-2-yl]-2-methoxyphenol | ||
| SMILES | COC1=C(C=CC(=C1)C2C(C(CO2)CC3=CC4=C(C=C3)OCO4)CO)O | ||
| Standard InChIKey | GRYMYKQGSSTJBA-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C20H22O6/c1-23-18-8-13(3-4-16(18)22)20-15(9-21)14(10-24-20)6-12-2-5-17-19(7-12)26-11-25-17/h2-5,7-8,14-15,20-22H,6,9-11H2,1H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Standard reference |
| Structure Identification | Chemical & Pharmaceutical Bulletin,1974,22(11) :2650-5.Studies on Xanthoxylun spp. II. Constituents of the bark of Xanthoxylum piperitum DC.[Reference: WebLink]
|

Sanshodiol Dilution Calculator

Sanshodiol Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.7902 mL | 13.9509 mL | 27.9018 mL | 55.8036 mL | 69.7545 mL |
| 5 mM | 0.558 mL | 2.7902 mL | 5.5804 mL | 11.1607 mL | 13.9509 mL |
| 10 mM | 0.279 mL | 1.3951 mL | 2.7902 mL | 5.5804 mL | 6.9754 mL |
| 50 mM | 0.0558 mL | 0.279 mL | 0.558 mL | 1.1161 mL | 1.3951 mL |
| 100 mM | 0.0279 mL | 0.1395 mL | 0.279 mL | 0.558 mL | 0.6975 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Protogracillin(P)
Catalog No.:BCC8352
CAS No.:54848-30-5
- Roseoside
Catalog No.:BCN5728
CAS No.:54835-70-0
- Daphneolone
Catalog No.:BCN3230
CAS No.:54835-64-2
- Protoveratrine B
Catalog No.:BCN2435
CAS No.:124-97-0
- Trichodesmine
Catalog No.:BCN2145
CAS No.:548-90-3
- Gyrophoric acid
Catalog No.:BCN5731
CAS No.:548-89-0
- Galangin
Catalog No.:BCN5730
CAS No.:548-83-4
- Pinobanksin
Catalog No.:BCN5729
CAS No.:548-82-3
- Tectorigenin
Catalog No.:BCN1019
CAS No.:548-77-6
- Irigenin
Catalog No.:BCN3849
CAS No.:548-76-5
- Quercetagetin-7-O-glucoside
Catalog No.:BCN6480
CAS No.:548-75-4
- Crystal Violet
Catalog No.:BCC4772
CAS No.:548-62-9
- PD 334581
Catalog No.:BCC6300
CAS No.:548756-68-9
- Arborinine
Catalog No.:BCN7438
CAS No.:5489-57-6
- Amitriptyline HCl
Catalog No.:BCC5033
CAS No.:549-18-8
- 8-Oxyberberine
Catalog No.:BCN3135
CAS No.:549-21-3
- Quercetin 3-O-beta-D-xylopyranoside
Catalog No.:BCN2851
CAS No.:549-32-6
- beta-Yohimbine
Catalog No.:BCN5733
CAS No.:549-84-8
- Phytolaccagenic acid
Catalog No.:BCN8090
CAS No.:54928-05-1
- Shikalkin
Catalog No.:BCC8359
CAS No.:54952-43-1
- 2-Oxopomolic acid
Catalog No.:BCN5732
CAS No.:54963-52-9
- Florilenalin
Catalog No.:BCN6422
CAS No.:54964-49-7
- Albendazole
Catalog No.:BCC3718
CAS No.:54965-21-8
- Tamoxifen Citrate
Catalog No.:BCC4382
CAS No.:54965-24-1
Short and stereoselective total synthesis of furano lignans (+/-)-dihydrosesamin, (+/-)-lariciresinol dimethyl ether, (+/-)-acuminatin methyl ether, (+/-)-sanshodiol methyl ether, (+/-)-lariciresinol, (+/-)-acuminatin, and (+/-)-lariciresinol monomethyl ether and furofuran lignans (+/-)-sesamin, (+/-)-eudesmin, (+/-)-piperitol methyl ether, (+/-)-pinoresinol, (+/-)-piperitol, and (+/-)-pinoresinol monomethyl ether by radical cyclization of epoxides using a transition-metal radical source.[Pubmed:12003531]
J Org Chem. 2002 May 17;67(10):3242-8.
Intramolecular radical cyclization of suitably substituted epoxy ethers 4a-g using bis(cyclopentadienyl)titanium(III) chloride as the radical source resulted in trisubstituted tetrahydrofurano lignans and 2,6-diaryl-3,7-dioxabicyclo[3.3.0]octane lignans depending on the reaction conditions. The titanium(III) species was prepared in situ from commercially available titanocene dichloride and activated zinc dust in THF. Upon radical cyclization followed by acidic workup, epoxy olefinic ethers 4a-g afforded furano lignans dihydrosesamin 1a, lariciresinol dimethyl ether 1b, acuminatin methyl ether 1e, and Sanshodiol methyl ether 1g directly and lariciresinol 1h, acuminatin 1i, and lariciresinol monomethyl ether 1j after removal of the benzyl protecting group by controlled hydrogenolysis of the corresponding cyclized products. The furofuran lignans sesamin 2a, eudesmin 2b, and piperitol methyl ether 2e were also prepared directly by using the same precursors 4a-f on radical cyclization followed by treatment with iodine and pinoresinol 2h, piperitol 2i, and pinoresinol monomethyl ether 2j after controlled hydrogenolysis of the benzyl protecting group of the corresponding cyclized products. Two naturally occurring acyclic lignans, secoisolariciresinol 5h and secoisolariciresinol dimethyl ether 5b, have also been prepared by exhaustive hydrogenolysis of 2h and 2b, respectively.


