Gyrophoric acidCAS# 548-89-0 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 548-89-0 | SDF | Download SDF |
| PubChem ID | 135728 | Appearance | Powder |
| Formula | C24H20O10 | M.Wt | 468.4 |
| Type of Compound | Phenols | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
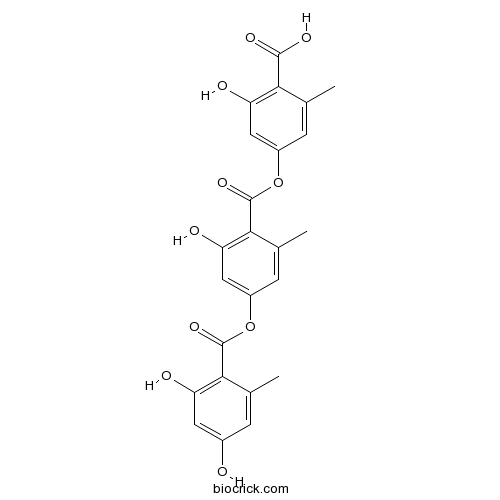
| Chemical Name | 4-[4-(2,4-dihydroxy-6-methylbenzoyl)oxy-2-hydroxy-6-methylbenzoyl]oxy-2-hydroxy-6-methylbenzoic acid | ||
| SMILES | CC1=CC(=CC(=C1C(=O)OC2=CC(=C(C(=C2)C)C(=O)OC3=CC(=C(C(=C3)C)C(=O)O)O)O)O)O | ||
| Standard InChIKey | ATQPZSQVWCPVGV-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C24H20O10/c1-10-4-13(25)7-16(26)20(10)23(31)34-15-6-12(3)21(18(28)9-15)24(32)33-14-5-11(2)19(22(29)30)17(27)8-14/h4-9,25-28H,1-3H3,(H,29,30) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. Gyrophoric acid has anti-proliferative/cytotoxic effect. 2. Vulpinic and gyrophoric acids are known as ultraviolet filters for natural lichen populations, they can effectively prevent cytotoxic, apoptotic and cytoskeleton alterative activities of 2.5 J/cm(2) UVB in a dose-dependent manner, suggest that vulpinic and gyrophoric acids can be promising cosmetic ingredients to photo-protect human skin cells. |
| Targets | cAMP |

Gyrophoric acid Dilution Calculator

Gyrophoric acid Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.1349 mL | 10.6746 mL | 21.3493 mL | 42.6985 mL | 53.3732 mL |
| 5 mM | 0.427 mL | 2.1349 mL | 4.2699 mL | 8.5397 mL | 10.6746 mL |
| 10 mM | 0.2135 mL | 1.0675 mL | 2.1349 mL | 4.2699 mL | 5.3373 mL |
| 50 mM | 0.0427 mL | 0.2135 mL | 0.427 mL | 0.854 mL | 1.0675 mL |
| 100 mM | 0.0213 mL | 0.1067 mL | 0.2135 mL | 0.427 mL | 0.5337 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Galangin
Catalog No.:BCN5730
CAS No.:548-83-4
- Pinobanksin
Catalog No.:BCN5729
CAS No.:548-82-3
- Tectorigenin
Catalog No.:BCN1019
CAS No.:548-77-6
- Irigenin
Catalog No.:BCN3849
CAS No.:548-76-5
- Quercetagetin-7-O-glucoside
Catalog No.:BCN6480
CAS No.:548-75-4
- Crystal Violet
Catalog No.:BCC4772
CAS No.:548-62-9
- Cornin
Catalog No.:BCN5008
CAS No.:548-37-8
- Oxysanguinarine
Catalog No.:BCN8100
CAS No.:548-30-1
- Isolariciresinol
Catalog No.:BCN5727
CAS No.:548-29-8
- Isoginkgetin
Catalog No.:BCN2320
CAS No.:548-19-6
- Roemerine
Catalog No.:BCN8236
CAS No.:548-08-3
- Hypericin
Catalog No.:BCN5977
CAS No.:548-04-9
- Trichodesmine
Catalog No.:BCN2145
CAS No.:548-90-3
- Protoveratrine B
Catalog No.:BCN2435
CAS No.:124-97-0
- Daphneolone
Catalog No.:BCN3230
CAS No.:54835-64-2
- Roseoside
Catalog No.:BCN5728
CAS No.:54835-70-0
- Protogracillin(P)
Catalog No.:BCC8352
CAS No.:54848-30-5
- Sanshodiol
Catalog No.:BCN6577
CAS No.:54854-91-0
- PD 334581
Catalog No.:BCC6300
CAS No.:548756-68-9
- Arborinine
Catalog No.:BCN7438
CAS No.:5489-57-6
- Amitriptyline HCl
Catalog No.:BCC5033
CAS No.:549-18-8
- 8-Oxyberberine
Catalog No.:BCN3135
CAS No.:549-21-3
- Quercetin 3-O-beta-D-xylopyranoside
Catalog No.:BCN2851
CAS No.:549-32-6
- beta-Yohimbine
Catalog No.:BCN5733
CAS No.:549-84-8
Photoprotective Activity of Vulpinic and Gyrophoric Acids Toward Ultraviolet B-Induced Damage in Human Keratinocytes.[Pubmed:26463741]
Phytother Res. 2016 Jan;30(1):9-15.
Vulpinic and Gyrophoric acids are known as ultraviolet filters for natural lichen populations because of their chemical structures. However, to the best of our knowledge, there has been no reference to their cosmetic potential for skin protection against ultraviolet B (UVB)-induced damage and, consequently, we propose to highlight their photoprotective profiles in human keratinocytes (HaCaT). Therefore, vulpinic acid and Gyrophoric acid were isolated from acetone extracts of Letharia vulpina and Xanthoparmelia pokornyi, respectively. Their photoprotective activities on irradiated HaCaT cells and destructive effects on non-irradiated HaCaT cells were compared through in vitro experimentation: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide and lactate dehydrogenase assays, 4',6-diamino-2-phenylindole and tetramethylrhodamine B isothiocyanate-phalloidin staining protocols. Both of the lichen substances effectively prevented cytotoxic, apoptotic and cytoskeleton alterative activities of 2.5 J/cm(2) UVB in a dose-dependent manner. Moreover, vulpinic and Gyrophoric acids showed no toxic, apoptotic or cytoskeleton alterative effects on non-irradiated HaCaT cells, except at high doses (>/=400 muM) of Gyrophoric acid. The findings suggest that vulpinic and Gyrophoric acids can be promising cosmetic ingredients to photo-protect human skin cells and should therefore be further investigated by in vitro and in vivo multiple bioassays.
Variable responses of different human cancer cells to the lichen compounds parietin, atranorin, usnic acid and gyrophoric acid.[Pubmed:20837130]
Toxicol In Vitro. 2011 Feb;25(1):37-44.
One of the ways for searching for potentially new anti-cancer drugs is the testing of various naturally synthesized compounds. Lichens are a source of unique chemical agents of which some have already been proved to be effective against various cancer in vitro models. Our study reports on the sensitivity of up to nine human cancer cell lines (A2780, HeLa, MCF-7, SK-BR-3, HT-29, HCT-116 p53(+/+), HCT-116 p53(-/-), HL-60 and Jurkat) to the anti-proliferative/cytotoxic effects of four typical secondary metabolites of lichens (parietin, atranorin, usnic acid and Gyrophoric acid). Variations in the dynamics of tumour cell line populations were evaluated by the MTT, clonogenic and viability assays, cell proliferation and detachment, cell cycle transition and apoptotic nuclear morphology, thereby confirming their concentration- and time-dependent cytotoxicity. However, in comparison with parietin and Gyrophoric acid, the suppression of viability and cell proliferation by usnic acid or atranorin was found to be more efficient at equitoxic doses and correlated more strongly with an increased number of floating cells or a higher apoptotic index. Moreover, the analysis of cell cycle distribution also revealed an accumulation of cells in S-phase. This study has confirmed a differential sensitivity of cancer cell lines to lichen secondary metabolites.


