(Z)-ButylidenephthalideCAS# 72917-31-8 |
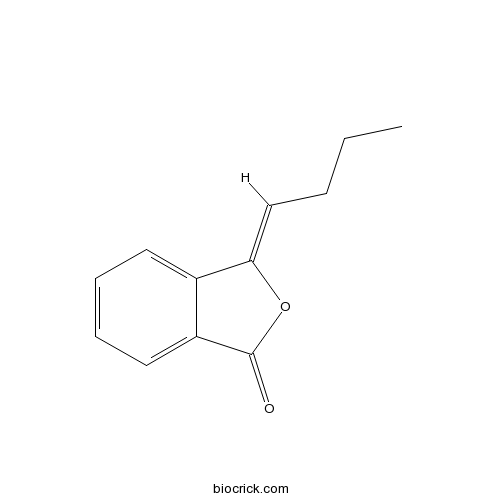
- 3-Butylidenephthalide
Catalog No.:BCN6345
CAS No.:551-08-6
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 72917-31-8 | SDF | Download SDF |
| PubChem ID | 642376 | Appearance | Oil |
| Formula | C12H12O2 | M.Wt | 188.2 |
| Type of Compound | Miscellaneous | Storage | Desiccate at -20°C |
| Synonyms | Butylidenephthalide; 3-Butylidenephthalide | ||
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | (3Z)-3-butylidene-2-benzofuran-1-one | ||
| SMILES | CCCC=C1C2=CC=CC=C2C(=O)O1 | ||
| Standard InChIKey | WMBOCUXXNSOQHM-FLIBITNWSA-N | ||
| Standard InChI | InChI=1S/C12H12O2/c1-2-3-8-11-9-6-4-5-7-10(9)12(13)14-11/h4-8H,2-3H2,1H3/b11-8- | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. (Z)-Butylidenephthalide has antitumor effect, can effectively inhibit the tumor growth in the glioma. |
| Targets | PKC |

(Z)-Butylidenephthalide Dilution Calculator

(Z)-Butylidenephthalide Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 5.3135 mL | 26.5675 mL | 53.135 mL | 106.2699 mL | 132.8374 mL |
| 5 mM | 1.0627 mL | 5.3135 mL | 10.627 mL | 21.254 mL | 26.5675 mL |
| 10 mM | 0.5313 mL | 2.6567 mL | 5.3135 mL | 10.627 mL | 13.2837 mL |
| 50 mM | 0.1063 mL | 0.5313 mL | 1.0627 mL | 2.1254 mL | 2.6567 mL |
| 100 mM | 0.0531 mL | 0.2657 mL | 0.5313 mL | 1.0627 mL | 1.3284 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- O-Methylcedrelopsin
Catalog No.:BCN3637
CAS No.:72916-61-1
- Isocrocandine
Catalog No.:BCN2071
CAS No.:72903-70-9
- 4-Propenylbrenzcatechin
Catalog No.:BCN3672
CAS No.:72898-29-4
- Benzoyloxypeoniflorin
Catalog No.:BCN2799
CAS No.:72896-40-3
- CHIR-090
Catalog No.:BCC1477
CAS No.:728865-23-4
- Agatharesinol
Catalog No.:BCN4594
CAS No.:7288-11-1
- Crocandine
Catalog No.:BCN2070
CAS No.:72855-83-5
- Echitoveniline
Catalog No.:BCN7490
CAS No.:72855-79-9
- 1-Benzyl-5-phenylbarbituric acid
Catalog No.:BCC8461
CAS No.:72846-00-5
- MIRA-1
Catalog No.:BCC2409
CAS No.:72835-26-8
- Deoxyartemisinin
Catalog No.:BCN4285
CAS No.:72826-63-2
- Atanine
Catalog No.:BCN3317
CAS No.:7282-19-1
- α,β-Methyleneadenosine 5'-triphosphate trisodium salt
Catalog No.:BCC3591
CAS No.:7292-42-4
- (RS)-4-Carboxyphenylglycine
Catalog No.:BCC6601
CAS No.:7292-81-1
- Bohemamine
Catalog No.:BCN1958
CAS No.:72926-12-6
- K858
Catalog No.:BCC7760
CAS No.:72926-24-0
- Octadecyl p-coumarate
Catalog No.:BCN7235
CAS No.:72943-88-5
- 30-Hydroxylup-20(29)-en-3-one
Catalog No.:BCN4286
CAS No.:72944-06-0
- DL-Catechin
Catalog No.:BCN6325
CAS No.:7295-85-4
- Carvedilol
Catalog No.:BCC4324
CAS No.:72956-09-3
- 2-Furoyl-LIGRLO-amide
Catalog No.:BCC3958
CAS No.:729589-58-6
- 6,7-Dihydroneridienone A
Catalog No.:BCN4020
CAS No.:72959-46-7
- Gomisin E
Catalog No.:BCN7031
CAS No.:72960-21-5
- Gomisin O
Catalog No.:BCN2875
CAS No.:72960-22-6
Local interstitial delivery of z-butylidenephthalide by polymer wafers against malignant human gliomas.[Pubmed:21565841]
Neuro Oncol. 2011 Jun;13(6):635-48.
We have shown that the natural compound z-butylidenephthalide (Bdph), isolated from the chloroform extract of Angelica sinensis, has antitumor effects. Because of the limitation of the blood-brain barrier, the Bdph dosage required for treatment of glioma is relatively high. To solve this problem, we developed a local-release system with Bdph incorporated into a biodegradable polyanhydride material, p(CPP-SA; Bdph-Wafer), and investigated its antitumor effects. On the basis of in vitro release kinetics, we demonstrated that the Bdph-Wafer released 50% of the available Bdph by the sixth day, and the release reached a plateau phase (90% of Bdph) by the 30th day. To investigate the in situ antitumor effects of the Bdph-Wafer on glioblastoma multiforme (GBM), we used 2 xenograft animal models-F344 rats (for rat GBM) and nude mice (for human GBM)-which were injected with RG2 and DBTRG-05MG cells, respectively, for tumor formation and subsequently treated subcutaneously with Bdph-Wafers. We observed a significant inhibitory effect on tumor growth, with no significant adverse effects on the rodents. Moreover, we demonstrated that the antitumor effect of Bdph on RG2 cells was via the PKC pathway, which upregulated Nurr77 and promoted its translocation from the nucleus to the cytoplasm. Finally, to study the effect of the interstitial administration of Bdph in cranial brain tumor, Bdph-Wafers were surgically placed in FGF-SV40 transgenic mice. Our Bdph-Wafer significantly reduced tumor size in a dose-dependent manner. In summary, our study showed that p(CPP-SA) containing Bdph delivered a sufficient concentration of Bdph to the tumor site and effectively inhibited the tumor growth in the glioma.
Metabolic conversion from co-existing ingredient leading to significant systemic exposure of Z-butylidenephthalide, a minor ingredient in Chuanxiong Rhizoma in rats.[Pubmed:22554277]
Curr Drug Metab. 2012 Jun 1;13(5):524-34.
Pharmacokinetic (PK) study of medicinal herbs is a great challenge, because which component(s) is(are) the bioactive ingredients is largely unknown. Most of the reported PK studies of herbs focused on the major ingredients regardless of their in vivo bioactivities, while PK of components with low content in herbs is often ignored. The present study demonstrates how PK study can reveal potential importance of a low content ingredient to the herbal bioactivities using Z-butylidenephthalide (BuPh), a bioactive phthalide present in a significantly low quantity in medicinal herb Chuanxiong Rhizoma, as an example. PK of BuPh was investigated in rats using Chuanxiong extract, fraction containing BuPh and ligustilide, and pure BuPh, respectively. The results demonstrated that remarkable blood concentrations of BuPh were observed after administration of the herbal extract and its systemic exposure was significantly different between BuPh given in pure and mixed forms. More interestingly, AUC of BuPh via intake of fraction (9.3-fold) and extract (4.5-fold) was significantly greater than that obtained from pure BuPh, which was further evidenced to be mainly due to metabolic conversion from ligustilide, a major component in Chuanxiong. Our findings revealed that although it naturally occurred in low amount, BuPh reached significant systemic concentrations via metabolic conversion from ligustilide. Moreover, our results demonstrated that PK study is one of crucial and inevitable steps for revealing in vivo bioactive ingredients of herbal medicines, and such studies should be more appropriate to focus on in vivo profile of the ingredients co-existing in herbs rather than only studying them individually.
Optimization of pressurized liquid extraction for Z-ligustilide, Z-butylidenephthalide and ferulic acid in Angelica sinensis.[Pubmed:16242882]
J Pharm Biomed Anal. 2006 Mar 18;40(5):1073-9.
Pressurized liquid extraction, one of the most promising and recent sample preparation techniques, offers the advantages of reducing solvent consumption and allowing for automated sample handling. It is being exploited in diverse areas because of its distinct advantages. However, because the extraction is performed at elevated temperatures using PLE, thermal degradation could be a concern. Z-ligustilide, one of the biologically active components in Angelica sinensis, is an unstable compound, which decomposes rapidly at high temperature. In this study, we carried out a comparative study to evaluate PLE as a possible alternative to current extraction methods like Soxhlet and sonication for simultaneous extraction of Z-ligustilide, Z-butylidenephthalide and ferulic acid in A. sinensis. The operating parameters for PLE including extraction solvent, particle size, pressure, temperature, static extraction time, flush volume and numbers of extraction were optimized by using univariate approach coupled with central composite design (CCD) in order to obtain the highest extraction efficiency. Determination of Z-ligustilide, Z-butylidenephthalide and ferulic acid were carried out by means of high performance liquid chromatography with diode-array detector. The results showed that PLE was a simple, high efficient and automated method with lower solvent consumption compared to conventional extraction methods such as Soxhlet and sonication. PLE could be used for simultaneous extraction of Z-ligustilide, Z-butylidenephthalide and ferulic acid in A. sinensis.


