CarboxyatractylosideCAS# 77228-71-8 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 77228-71-8 | SDF | Download SDF |
| PubChem ID | 102004836 | Appearance | Powder |
| Formula | C31H43K3O18S2 | M.Wt | 885.1 |
| Type of Compound | Diterpenoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
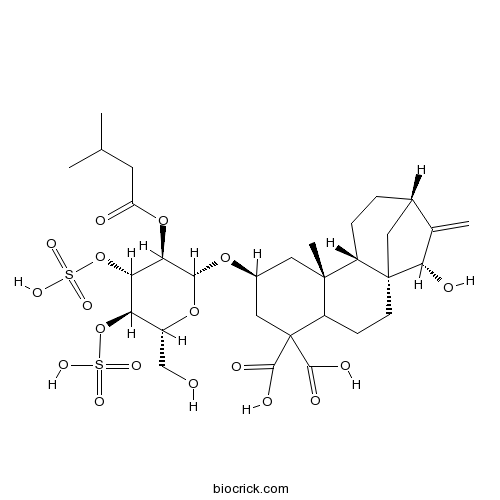
| SMILES | CC(C)CC(=O)OC1C(C(C(OC1OC2CC3(C4CCC5CC4(CCC3C(C2)(C(=O)O)C(=O)O)C(C5=C)O)C)CO)OS(=O)(=O)O)OS(=O)(=O)O | ||
| Standard InChIKey | AQFATIOBERWBDY-SRIINCLRSA-N | ||
| Standard InChI | InChI=1S/C31H46O18S2/c1-14(2)9-21(33)47-24-23(49-51(42,43)44)22(48-50(39,40)41)18(13-32)46-26(24)45-17-11-29(4)19-6-5-16-10-30(19,25(34)15(16)3)8-7-20(29)31(12-17,27(35)36)28(37)38/h14,16-20,22-26,32,34H,3,5-13H2,1-2,4H3,(H,35,36)(H,37,38)(H,39,40,41)(H,42,43,44)/t16-,17-,18+,19-,20?,22+,23-,24+,25-,26+,29+,30+/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. Carboxyatractyloside induces the inhibitory effect of tamoxifen on nonspecific membrane permeability. 2. Carboxyatractyloside can induce permeability transition, and that ageing induced mitochondrial DNA disruption and release of cytochrome c. 3. Carboxyatractyloside poisoning causes multiple organ dysfunction and can be fatal, the signs of a poor prognosis including coagulation abnormalities, hyponatraemia, marked hypoglycaemia, icterus and hepatic and renal failure, without antidote. |
| Targets | Calcium Channel | P450 (e.g. CYP17) |

Carboxyatractyloside Dilution Calculator

Carboxyatractyloside Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.1298 mL | 5.6491 mL | 11.2982 mL | 22.5963 mL | 28.2454 mL |
| 5 mM | 0.226 mL | 1.1298 mL | 2.2596 mL | 4.5193 mL | 5.6491 mL |
| 10 mM | 0.113 mL | 0.5649 mL | 1.1298 mL | 2.2596 mL | 2.8245 mL |
| 50 mM | 0.0226 mL | 0.113 mL | 0.226 mL | 0.4519 mL | 0.5649 mL |
| 100 mM | 0.0113 mL | 0.0565 mL | 0.113 mL | 0.226 mL | 0.2825 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Nefiracetam
Catalog No.:BCC4504
CAS No.:77191-36-7
- Rheediaxanthone A
Catalog No.:BCN7411
CAS No.:77181-97-6
- Acetylaconitine
Catalog No.:BCN2407
CAS No.:77181-26-1
- Delta-Caesalpin
Catalog No.:BCN6698
CAS No.:7716-14-5
- Spiraline
Catalog No.:BCN2112
CAS No.:77156-25-3
- Spiracine
Catalog No.:BCN2095
CAS No.:77156-24-2
- Spiranine
Catalog No.:BCN2094
CAS No.:77156-23-1
- 6alpha-Hydroxycleroda-3,13-dien-16,15-olid-18-oic acid
Catalog No.:BCN1359
CAS No.:771493-42-6
- SR 57227 hydrochloride
Catalog No.:BCC6967
CAS No.:77145-61-0
- Fmoc-N-Me-Phe-OH
Catalog No.:BCC2614
CAS No.:77128-73-5
- Fmoc-Phe(4-OMe)-OH,Fmoc-Tyr(Me)-OH
Catalog No.:BCC2634
CAS No.:77128-72-4
- Fmoc-Sar-OH
Catalog No.:BCC3338
CAS No.:77128-70-2
- MG 624
Catalog No.:BCC7028
CAS No.:77257-42-2
- Erythrabyssin II
Catalog No.:BCN4828
CAS No.:77263-06-0
- Abyssinone V
Catalog No.:BCN6825
CAS No.:77263-11-7
- Zederone
Catalog No.:BCN3524
CAS No.:7727-79-9
- Fmoc-Nle-OH.
Catalog No.:BCC3298
CAS No.:77284-32-3
- Edelfosine
Catalog No.:BCC7537
CAS No.:77286-66-9
- Hirsutine
Catalog No.:BCN2758
CAS No.:7729-23-9
- SL 0101-1
Catalog No.:BCC8086
CAS No.:77307-50-7
- Hyuganin D
Catalog No.:BCN7679
CAS No.:77331-76-1
- Acamprosate calcium
Catalog No.:BCC1327
CAS No.:77337-73-6
- Triacetylpseurotin A
Catalog No.:BCN6916
CAS No.:77353-57-2
- Drimiopsin C
Catalog No.:BCN4325
CAS No.:773850-90-1
Cyclosporin A is unable to inhibit carboxyatractyloside-induced permeability transition in aged mitochondria.[Pubmed:18835371]
Comp Biochem Physiol C Toxicol Pharmacol. 2009 Apr;149(3):374-81.
We studied the effect of mitochondrial ageing on membrane permeability transition. The results obtained indicate that aged mitochondria are neither able to retain Ca2+ nor to maintain a high transmembrane electric gradient. In addition, aged mitochondria undergo a large amplitude swelling. These dysfunctions were circumvented by the addition of cyclosporin A. Furthermore, it is shown that ageing-induced permeability transition causes oxidative damage on the matrix enzyme aconitase. The observed damage in aged mitochondria requires Ca2+ addition; therefore, it was not seen when Sr2+ replaced Ca2+. Two important findings in this work were the fact that despite of the presence of cyclosporin A, Carboxyatractyloside was still able to induce permeability transition, and that ageing induced mitochondrial DNA disruption and release of cytochrome c. It is likely that the membrane's increased permeability is due to the effect of fatty acids, since bovine serum albumin makes mitochondria able to retain Ca2+. However, the possibility that the damage might be the result of oxidative stress cannot be discarded.
Protective action of tamoxifen on carboxyatractyloside-induced mitochondrial permeability transition.[Pubmed:21324322]
Life Sci. 2011 Apr 11;88(15-16):681-7.
AIMS: Mitochondrial permeability transition is established after massive Ca(2+) accumulation inside the matrix, in addition to an inducer. The closure of the pore can be accomplished by adenosine diphosphate and the immunosuppressant cyclosporin A. Recently, the estrogen antagonist, tamoxifen, has been introduced as an inhibitor of the opening of the permeability transition pore. However, the mechanism by which this drug inhibits pore opening is still under discussion. This work was performed with the purpose of establishing the membrane system involved in tamoxifen-induced pore closure. For this purpose, permeability transition was induced after the addition of Carboxyatractyloside, which is a specific reagent that interacts with the adenine nucleotide translocase. MAIN METHODS: Permeability transition was assessed by analyzing matrix Ca(2+) release, transmembrane electric gradient, and mitochondrial swelling in aged, as well as in freshly prepared mitochondria. Also, cytochrome c content was analyzed in membrane mitochondria as well as in the supernatant. KEY FINDINGS: In freshly prepared mitochondria, tamoxifen, at the concentration of 10 muM, totally inhibited nonspecific membrane permeability induced by 1 muM Carboxyatractyloside. In addition, tamoxifen inhibited non-specific permeability in aged mitochondria and diminished membrane fluidity. SIGNIFICANCE: Plausibly, the inhibitory effect of tamoxifen on nonspecific membrane permeability, as induced by Carboxyatractyloside, should be ascribed to a diminution, of membrane fluidity by this drug.
Carboxyatractyloside poisoning in humans.[Pubmed:15949201]
Ann Trop Paediatr. 2005 Jun;25(2):125-34.
OBJECTIVE: Cocklebur (Xanthium strumarium) is an herbaceous annual plant with worldwide distribution. The seeds contain the glycoside Carboxyatractyloside, which is highly toxic to animals. We describe nine cases of Carboxyatractyloside poisoning in humans which, to our knowledge, has not previously been reported. The clinical, laboratory and histopathological findings and our therapeutic approach are also discussed. SUBJECTS AND METHODS: The patients presented with acute onset abdominal pain, nausea and vomiting, drowsiness, palpitations, sweating and dyspnoea. Three of them developed convulsions followed by loss of consciousness and death. RESULTS: Laboratory findings showed raised liver enzymes, indicating severe hepatocellular damage. BUN and creatinine levels were raised, especially in the fatal cases who also displayed findings of consumption coagulopathy. CPK-MB values indicative of myocardial injury were also raised, especially in the fatal cases. Three of the patients died within 48 hours of ingesting Carboxyatractyloside. Post-mortem histopathology of the liver confirmed centrilobular hepatic necrosis and renal proximal tubular necrosis, secondary changes owing to increased permeability and microvascular haemorrhage in the cerebrum and cerebellum, and leucocytic infiltrates in the muscles and various organs including pancreas, lungs and myocardium. CONCLUSIONS: Carboxyatractyloside poisoning causes multiple organ dysfunction and can be fatal. Coagulation abnormalities, hyponatraemia, marked hypoglycaemia, icterus and hepatic and renal failure are signs of a poor prognosis. No antidote is available and supportive therapy is the mainstay of treatment.
Cyclosporin A inhibits UV-radiation-induced membrane damage but is unable to inhibit carboxyatractyloside-induced permeability transition.[Pubmed:19883225]
Radiat Res. 2009 Nov;172(5):575-83.
This work was undertaken to gain further information on the chemical characteristics of the membrane entity involved in the formation of the nonspecific pore. Mitochondria were subjected to oxidative stress by exposure to UV radiation. The results indicate that ultraviolet C radiation induces structural modifications in the adenine nucleotide translocase that lead to membrane permeability transition. Membrane leakage was assessed by measuring mitochondrial Ca2+ transport, the transmembrane electric gradient, and mitochondrial swelling. UV-irradiated mitochondria were unable to retain matrix Ca2+ or to maintain a high level of membrane potential when Ca2+ was added; furthermore, UV-irradiated mitochondria underwent large amplitude swelling. Release of cytochrome c and formation of malondialdehyde, owing to lipid peroxidation, were also seen. Structural modifications of the translocase were revealed by an increase in the binding of the fluorescent probe eosin-5-maleimide to thiol residues of the ADP/ATP carrier. These modifications, taken together with findings indicating that cyclosporin resulted unable to inhibit Carboxyatractyloside-induced permeability transition, prompted us to conclude that the translocase could constitute the nonspecific pore or at least be an important modulator of it.


