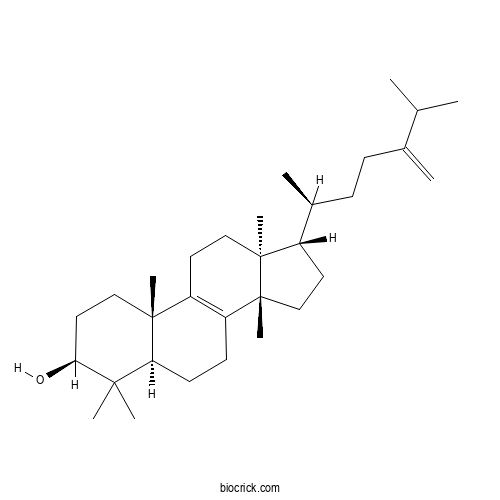
EuphorbolCAS# 566-14-3 |
- Eburicol
Catalog No.:BCN4252
CAS No.:6890-88-6
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 566-14-3 | SDF | Download SDF |
| PubChem ID | 10863111 | Appearance | White powder |
| Formula | C31H52O | M.Wt | 440.7 |
| Type of Compound | Isoprenoids | Storage | Desiccate at -20°C |
| Synonyms | Euphorbadienol; 24-Methyltirucalla 8,24-dien 3β-ol | ||
| Solubility | soluble in chloroform, methanol and DMSO; insoluble in water | ||
| Chemical Name | (3S,5R,10S,13S,14S,17S)-4,4,10,13,14-pentamethyl-17-[(2S)-6-methyl-5-methylideneheptan-2-yl]-2,3,5,6,7,11,12,15,16,17-decahydro-1H-cyclopenta[a]phenanthren-3-ol | ||
| SMILES | CC(C)C(=C)CCC(C)C1CCC2(C1(CCC3=C2CCC4C3(CCC(C4(C)C)O)C)C)C | ||
| Standard InChIKey | XJLZCPIILZRCPS-CKCUNPICSA-N | ||
| Standard InChI | InChI=1S/C31H52O/c1-20(2)21(3)10-11-22(4)23-14-18-31(9)25-12-13-26-28(5,6)27(32)16-17-29(26,7)24(25)15-19-30(23,31)8/h20,22-23,26-27,32H,3,10-19H2,1-2,4-9H3/t22-,23-,26-,27-,29+,30-,31+/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Euphorbol Dilution Calculator

Euphorbol Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.2691 mL | 11.3456 mL | 22.6912 mL | 45.3823 mL | 56.7279 mL |
| 5 mM | 0.4538 mL | 2.2691 mL | 4.5382 mL | 9.0765 mL | 11.3456 mL |
| 10 mM | 0.2269 mL | 1.1346 mL | 2.2691 mL | 4.5382 mL | 5.6728 mL |
| 50 mM | 0.0454 mL | 0.2269 mL | 0.4538 mL | 0.9076 mL | 1.1346 mL |
| 100 mM | 0.0227 mL | 0.1135 mL | 0.2269 mL | 0.4538 mL | 0.5673 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Ethyl trans-caffeate
Catalog No.:BCN0320
CAS No.:66648-50-8
- (+/-)-Eriodictyol
Catalog No.:BCN0319
CAS No.:4049-38-1
- Echinatine N-oxide
Catalog No.:BCN0318
CAS No.:20267-93-0
- Diosmetin 7-glucuronide
Catalog No.:BCN0317
CAS No.:35110-20-4
- 2',6'-Dihydroxy 4',4-dimethoxychalcone
Catalog No.:BCN0316
CAS No.:94441-99-3
- Dihydroavenanthramide D
Catalog No.:BCN0315
CAS No.:697235-49-7
- 23-epi-26-Deoxyactein
Catalog No.:BCN0314
CAS No.:501938-01-8
- 5,6-Dehydro 7,8-dihydrokavain
Catalog No.:BCN0313
CAS No.:3155-51-9
- Cycloartenol
Catalog No.:BCN0312
CAS No.:469-38-5
- trans-Coutaric acid
Catalog No.:BCN0311
CAS No.:27174-07-8
- trans-p-Coumaric acid
Catalog No.:BCN0310
CAS No.:501-98-4
- Convolidine
Catalog No.:BCN0309
CAS No.:63911-32-0
- trans-Ferulic acid
Catalog No.:BCN0322
CAS No.:537-98-4
- Fukinolic acid
Catalog No.:BCN0323
CAS No.:50982-40-6
- Grindelic acid
Catalog No.:BCN0324
CAS No.:1438-57-9
- (Rac)-Hesperetin
Catalog No.:BCN0325
CAS No.:69097-99-0
- 3'-Hydroxyflavone
Catalog No.:BCN0326
CAS No.:70460-18-3
- Hyperforin (stable Dicyclohexylammonium salt)
Catalog No.:BCN0327
CAS No.:238074-03-8
- Isomenthone
Catalog No.:BCN0328
CAS No.:491-07-6
- Isoxanthohumol
Catalog No.:BCN0329
CAS No.:521-48-2
- Lactucin
Catalog No.:BCN0330
CAS No.:1891-29-8
- Lavandulol
Catalog No.:BCN0331
CAS No.:58461-27-1
- Lavandulyl acetate
Catalog No.:BCN0332
CAS No.:25905-14-0
- (+)-Lupanine hydrochloride
Catalog No.:BCN0333
CAS No.:1025-39-4
Triterpene derivatives from Euphorbia enhance resistance against Verticillium wilt of tomato.[Pubmed:28027775]
Phytochemistry. 2017 Mar;135:169-180.
Oxidation of alpha-Euphorbol and 31-norlanostenol, two triterpenic compounds isolated from the latex of Euphorbia resinifera and Euphorbia officinarum respectively, yielded four products named 3beta-tosyloxy-4alpha,14alpha-dimethyl-5alpha-cholesta-7,9-diene; 4alpha,14alpha-dimethyl-5alpha-cholesta-7,9-dien-3beta-ol; 24-methylen-elemo-lanosta-8,24-dien-3-one and elemo-lanost-8-en-3,11,24-trione. They were evaluated for protection of tomato plants against Verticillium dahliae in a greenhouse. The four semisynthesized products were phytotoxic at higher concentrations as they completely inhibited tomato germination at 100 and 500 mug/ml. However at lower concentrations (10 and 50 mug/ml) germination and root length were not affected. Disease resistance against Verticillium wilt was assessed in tomato plants derived from seeds that germinated in the presence of 10 and 50 mug/ml of the four products. All of them were able to reduce significantly disease severity, with 10 mug/ml being more effective than 50 mug/ml. Reduction of leaf alteration index and of stunting index ranged from 52 to 68% and from 43 to 67%, respectively, while vessel discoloration was reduced by at least 95%. The compounds were also able to elicit H2O2 accumulation before and after fungal inoculation and to significantly enhance peroxidase and polyphenol oxidase activities. These results suggest that the hemisynthetized triterpenes can be used as elicitors of disease resistance.
Two new lanostane triterpenoids from the branches and leaves of Polyalthia oblique.[Pubmed:24914904]
Molecules. 2014 Jun 6;19(6):7621-8.
Two new lanostane triterpenoids, 20-hydroxyEuphorbol-7-one (1) and 15alpha-hydroxyEuphorbol-7,11-dione (2), together with four known triterpenoids, Euphorbol-7-one (3), friedelin (4), stigmast-4-ene-6alpha-ol-3-one (5), stigmasta-4-en-3,6-dione (6), were isolated from ethanol extract of the branches and leaves of Polyalthia obliqua. The structures of 1 and 2 were elucidated on the basis of extensive spectroscopic analysis and comparisons with related known compounds. Antibacterial activities of two new compounds and four known compounds were tested.
Antileishmanial and antitrypanosomal activity of triterpene derivatives from latex of two Euphorbia species.[Pubmed:21950160]
Z Naturforsch C J Biosci. 2011 Jul-Aug;66(7-8):360-6.
The in vitro activity on Leishmania infantum promastigotes and Trypanosoma cruzi epimastigotes of 25 semisynthetic terpenoid derivatives has been evaluated. These compounds were obtained through chemical modifications of the major components of Euphorbia resinifera (alpha-euphol and alpha-Euphorbol) and Euphorbia officinarum (obtusifoliol and 31-norlanosterol). Leishmaniasis and Chagas' disease are major worldwide health problems. The drugs of choice for their treatment are still problematic in both cases, and therefore there is an urgent need to discover new drugs with high activity and low side effects. Natural products have become a key source of new drugs in the last years. The genus Euphorbia has been the subject of abundant phytochemical and pharmacological research because of its potential medical applications, but the antiparasitic effects of derivatives from plants of this genus are still unknown. Our results showed that 76% and 64% of the test compounds had antiparasitic effects on L. infantum and T cruzi, respectively. The different activities on both parasites, especially their moderate effects on mammalian cells, indicate an interesting selective toxicity.
[Chemical constitutents in fermental mycelium of Xylaria nigripes].[Pubmed:18831203]
Zhongguo Zhong Yao Za Zhi. 2008 Jun;33(11):1269-72.
Nine known compounds, 5-hydroxy-7-methoxy-2-methyl-4-chromanone (1), 5, 7-dihydroxy-2-methyl-4-chromanone (2), 5-hydroxyl-2-methyl-4-chromanone (3), 1-(2, 6-dihydroxyphenyl)-3-hydroxybutanone(4), 5alpha, 8alpha-epidioxyergosta-6, 22-dien-3beta-ol (5), (22E,24R)-ergost-7, 22-dien-3beta, 5alpha, 6alpha-triol (6), Euphorbol (7), beta-sitosterol (8) and 2-(4-hydroxyphenyl) -ethanol (9) were isolated and identified in Xylaria nigripes for the first time, and their structures were mainly determined by MS and NMR methods. Compounds 1 and 2 were isolated as the natural product for the first time.
Bioactive triterpene derivatives from latex of two Euphorbia species.[Pubmed:18304594]
Phytochemistry. 2008 Apr;69(6):1328-38.
We have investigated the antifeedant and toxic effects of 23 semisynthetic terpenoid derivatives obtained through chemical modifications of the major components of Euphorbia resinifera (alpha-euphol and alpha-Euphorbol) and E. officinarum (obtusifoliol and 31-norlanostenol) latex on several insect species (Spodoptera littoralis, Myzus persicae and Rhopalosiphum padi), their selective cytotoxicity on insect Sf9 and mammalian CHO cells and their phytotoxic effects on Lactuca sativa. The conversions focused mainly on positions 3,7,11, and 24 with several oxidizing agents. A total of 18 compounds affected S. littoralis growth (IGR). Our results support the importance of the C-3 substituent, suggest the involvement of the C-7 substituent and indicate that the C-3 hydroxyl is not essential for the IGR effect. Overall, Sf9 cells were more sensitive to the active compounds than CHO cells. All of these compounds had non selective moderate phytotoxic effects on radicle elongation of L. sativa.
Eupha-7,9(11),24-trien-3beta-ol ("antiquol C") and other triterpenes from Euphorbia antiquorum latex and their inhibitory effects on Epstein-Barr virus activation.[Pubmed:11858748]
J Nat Prod. 2002 Feb;65(2):158-62.
The structures of three triterpene alcohols isolated from the latex of Euphorbia antiquorum were established to be eupha-7,9(11),24-trien-3beta-ol (2; antiquol C), 19(10-->9)abeo-8alpha,9beta,10alpha-eupha-5,24-dien-3beta-ol (3; antiquol B), and 24-methyltirucalla-8,24(24(1))-dien-3beta-ol (4; Euphorbol) on the basis of spectroscopic methods. Compounds 3 and 4 have previously been assigned the erroneous structures of 10alpha-cucurbita-5,24-dien-3alpha-ol and 24-methyleupha-8,24(24(1))-dien-3beta-ol, respectively. Compounds 2-4 and four other known compounds isolated from the latex, euphol (1), lemmaphylla-7,21-dien-3beta-ol (5), isohelianol (6), and camelliol C (7), showed potent inhibitory effects on Epstein-Barr virus early antigen (EBV-EA) activation induced by the tumor promoter 12-O-tetradecanoylphorbol-13-acetate (TPA).
Pneumocysterol [(24Z)-ethylidenelanost-8-en-3beta-ol], a rare sterol detected in the opportunistic pathogen Pneumocystis carinii hominis: structural identity and chemical synthesis.[Pubmed:9874778]
Proc Natl Acad Sci U S A. 1999 Jan 5;96(1):97-102.
Pneumocystis carinii pneumonia (PcP) remains among the most prevalent opportunistic infections among AIDS patients. Currently, drugs used clinically for deep mycosis act by binding ergosterol or disrupting its biosynthesis. Although classified as a fungus, P. carinii lacks ergosterol. Instead, the pathogen synthesizes a number of distinct Delta7, 24-alkylsterols, despite the abundance of cholesterol, which it can scavenge from the lung alveolus. Thus, the pathogen-specific sterols appear vital for organism survival and proliferation. In the present study, high concentrations of a C32 sterol were found in human-derived P. carinii hominis. The definitive structural identities of two C-24 alkylated lanosterol compounds, previously not reported for rat-derived P. carinii carinii, were determined by using GLC, MS, and NMR spectroscopy together with the chemical syntheses of authentic standards. The C31 and C32 sterols were identified as Euphorbol (24-methylenelanost-8-en-3beta-ol) and pneumocysterol [(24Z)-ethylidenelanost-8-en-3beta-ol], respectively. The identification of these and other 24-alkylsterols in P. carinii hominis suggests that (i) sterol C-24 methyltransferase activities are extraordinarily high in this organism, (ii) 24-alkylsterols are important components of the pathogen's membranes, because the addition of these side groups onto the sterol side chain requires substantial ATP equivalents, and (iii) the inefficacy of azole drugs against P. carinii can be explained by the ability of this organism to form 24-alkysterols before demethylation of the lanosterol nucleus. Because mammals cannot form 24-alkylsterols, their biosyntheses in P. carinii are attractive targets for the development of chemotherapeutic strategies against this opportunistic infection.


