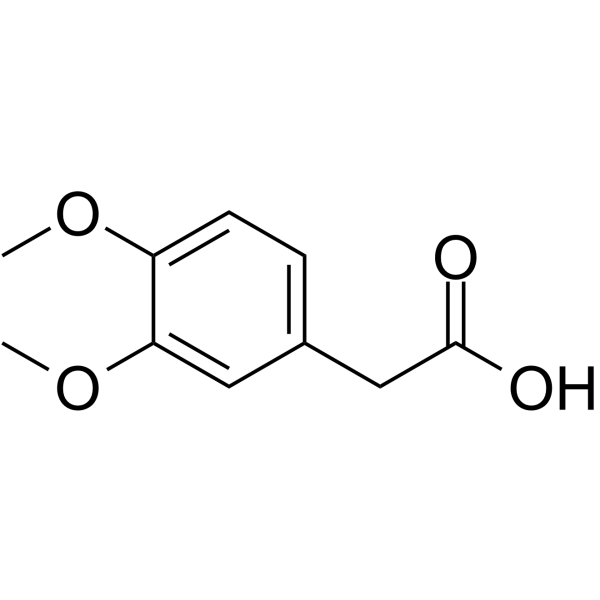
Homoveratric acidCAS# 93-40-3 |
Quality Control & MSDS
Package In Stock
Number of papers citing our products

| Cas No. | 93-40-3 | SDF | File under preparation. |
| PubChem ID | N/A | Appearance | Powder |
| Formula | C10H12O4 | M.Wt | 196.2 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | 3,4-Dimethoxybenzeneacetic acid,2-(3,4-Dimethoxyphenyl)acetate | ||
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Homoveratric acid Dilution Calculator

Homoveratric acid Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 5.0968 mL | 25.4842 mL | 50.9684 mL | 101.9368 mL | 127.421 mL |
| 5 mM | 1.0194 mL | 5.0968 mL | 10.1937 mL | 20.3874 mL | 25.4842 mL |
| 10 mM | 0.5097 mL | 2.5484 mL | 5.0968 mL | 10.1937 mL | 12.7421 mL |
| 50 mM | 0.1019 mL | 0.5097 mL | 1.0194 mL | 2.0387 mL | 2.5484 mL |
| 100 mM | 0.051 mL | 0.2548 mL | 0.5097 mL | 1.0194 mL | 1.2742 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Ladanetin-6-O-β-D-glucopyranoside
Catalog No.:BCX1699
CAS No.:89647-63-2
- Pedaliin 6''-acetate
Catalog No.:BCX1698
CAS No.:160789-41-3
- Cirsiliol 4'-glucoside
Catalog No.:BCX1697
CAS No.:41087-98-3
- Ladanetin-6-O-β-(6''-O- acetyl)glucoside
Catalog No.:BCX1696
CAS No.:1256959-27-9
- Complanatin II
Catalog No.:BCX1695
CAS No.:142449-94-3
- Apigenin-7-O-diglucuronoside
Catalog No.:BCX1694
CAS No.:119738-57-7
- Arborescosidic acid
Catalog No.:BCX1693
CAS No.:325798-51-4
- (+)-(3’S,4’R)-3’-acetyl-4’-isobutyrylkhellactone
Catalog No.:BCX1692
CAS No.:1562996-11-5
- Bruceantarin
Catalog No.:BCX1691
CAS No.:41451-76-7
- 2-Mialine
Catalog No.:BCX1690
CAS No.:634-97-9
- Glycoside St-J
Catalog No.:BCX1689
CAS No.:203513-88-6
- Protoapigenone
Catalog No.:BCX1688
CAS No.:862884-32-0
- Scutellarein-7-O-diglucuronide
Catalog No.:BCX1701
CAS No.:150641-65-9
- Alismanol M
Catalog No.:BCX1702
CAS No.:2408810-59-1
- Celangulin V
Catalog No.:BCX1703
CAS No.:139979-81-0
- Caffeic acid tetramer
Catalog No.:BCX1704
CAS No.:130286-75-8
- (1α,2α,6β,8α,9α)-1,2,6,8,12-pentakis(acetyloxy)-9-(benzoyloxy)dihydro-β-agarofuran
Catalog No.:BCX1705
CAS No.:128443-60-7
- Epinodosinol
Catalog No.:BCX1706
CAS No.:27548-88-5
- Apigenin 4'-O-β-D-glucopyranoside
Catalog No.:BCX1707
CAS No.:20486-34-4
- Celangulatin C
Catalog No.:BCX1708
CAS No.:1000784-44-0
- Angulatin G
Catalog No.:BCX1709
CAS No.:1000784-46-2
- Angulatin E
Catalog No.:BCX1710
CAS No.:403613-20-7
- Protoapigenin 4'-O-β-D-glucoside
Catalog No.:BCX1711
CAS No.:163559-04-4
- 2-(trans-1,4-dihydroxy-cyclohexyl)-5,7-dihydroxy-chromone
Catalog No.:BCX1712
CAS No.:1270013-29-0
Smart chemometrics-assisted spectrophotometric methods for efficient resolution and simultaneous determination of paracetamol, caffeine, drotaverine HCl along with three of their corresponding related impurities.[Pubmed:37798793]
BMC Chem. 2023 Oct 5;17(1):133.
Three novel, simple and accurate multivariate spectrophotometric assisted mathematical techniques were developed for determination of paracetamol, caffeine, drotaverine HCl and their related impurities. The used multivariate algorithms are principal component regression (PCR), partial least squares (PLS), and synergy intervals partial least squares (siPLS). Linearity of the suggested methods was found to be (1.00-14.60, 1.40-7.00, 1.40-3.80, 1.00-3.00, 1.50-3.50 and 2.50-4.50 microg/mL) for paracetamol, caffeine, drotaverine HCl, and their related impurities; p-aminophenol, theophylline and Homoveratric acid, correspondingly. The presented methods were effectively implemented in the determination of the cited compounds in their laboratory prepared mixtures. Commercially available tablet preparation was also analyzed using the applied methods where no impurities were detected and without interference from tablet additives. Moreover, statistical analysis did not reveal any noticeable differences between the obtained results and those acquired from the reported method in terms of accuracy and precision. The developed multivariate algorithms were validated by means of internal and external validation sets. The obtained results showed the siPLS algorithm's superiority to PCR and PLS according to the values of correlation coefficient values (r) and the lowest root mean square error of prediction (RMSEP). The combination of four subintervals [10, 12, 14, and 17] produced the highest efficiency model. Furthermore, these methods may be an applicable substitute to HPLC ones in quality control laboratories during rush of analyses where several samples have to be analyzed in a short time.
HPLC-DAD-MS and Antioxidant Profile of Fractions from Amontillado Sherry Wine Obtained Using High-Speed Counter-Current Chromatography.[Pubmed:33435411]
Foods. 2021 Jan 9;10(1):131.
In the present work, the polyphenolic profile of a complex matrix such as Amontillado sherry has been processed by means of high-speed counter-current chromatography (HSCCC) and characterized by HPLC-DAD-MS. An Amberlite XAD-7 column was used to obtain the wine extract, and three different biphasic solvent systems were applied for HSCCC separation: MTBE (methyl tert-butyl ether)/n-butanol/acetonitrile/water (1.1/3/1.1/5+0.1% trifluoroacetic acid), MTBE/n-butanol/acetonitrile/water (2/2/1/5), and hexane/ethyl acetate/ethanol/water (1/5/1/5). As a result, 42 phenolic compounds and furanic derivatives have been identified by means of HPLC-DAD-MS, with 11 of them being identified for the first time in Sherry wines: 3-feruloylquinic acid, isovanillin, ethyl vanillate, furoic acid, dihydro-p-coumaric acid, 6-O-feruloylglucose, ethyl gallate, hydroxytyrosol, methyl protocatechuate, Homoveratric acid and veratraldehyde. In addition, the antioxidant capacity (ABTS) of the obtained fractions was determined, revealing higher values in those fractions in which compounds such as gallic acid, protocatechuic acid, protocatechualdehyde, trans-caftaric acid, syringic acid, isovanillin or tyrosol, among others, were present. This is the first time that HSCCC has been used to characterize the phenolic composition of Sherry wines.
Metabolomic differences of seminal plasma between boars with high and low average conception rates after artificial insemination.[Pubmed:33176019]
Reprod Domest Anim. 2021 Jan;56(1):161-171.
Seminal plasma is a complex biological fluid containing many metabolites including amino acids, fructose, carbohydrates and lipids Metabolites play important roles in multiple biological processes, but details and significance of the seminal plasma metabolome related to boar fertility are unknown. The aim of the present study was to compare the comprehensive metabolome of seminal plasma from boars with different conception rate after artificial insemination and to identify the potential biomarkers. Semen samples were collected from boars which divided into two groups according to the conception rates in the offspring. Seminal plasma metabolites were isolated, purified, and then subjected to Ultra-high Performance Liquid Chromatography-Quadrupole Time-of-Flight Mass Spectrometry (UHPLC-qTOF-MS) procession. A total of 576 (Positive ion mode) and 377 (Negative ion mode) metabolites were identified in seminal plasma. Metabolites were identified and categorized according to their major chemical classes, including carboxylic acids and derivatives, organooxygen compounds, amino acids, peptides, and alogues, fatty amides, fatty acyls, benzene and substituted derivatives, purine nucleotides, pyrimidine nucleotides, glycosyl compounds, fatty acids and conjugates. The results showed that 4-Aminobenzoate, Pro-Asn, Ile-Tyr, Homoveratric acid and D-Biotin were higher in semen of boar with higher conception rate (HG) versus lower conception rate (LG) (p < .05), whereas L-Serine, Butoxyacetic acid, S-Methyl-5'-thioadenosine, Capsaicin and 1-O-(cis-9-Octadecenyl)-2-O-acetyl-sn-glycero-3-phosphocholine (PAF) were lower in HG than in LG (p < .05). These metabolites may be considered as candidate biomarkers for different fertility in boars.
Identification/quantification of free and bound phenolic acids in peel and pulp of apples (Malus domestica) using high resolution mass spectrometry (HRMS).[Pubmed:27542479]
Food Chem. 2017 Jan 15;215:301-10.
Free and bound phenolic acids were measured in the pulp and peel of four varieties of apples using high resolution mass spectrometry. Twenty-five phenolic acids were identified and included: 8 hydroxybenzoic acids, 11 hydroxycinnamic acids, 5 hydroxyphenylacetic acids, and 1 hydoxyphenylpropanoic acid. Several phenolics are tentatively identified for the first time in apples and include: methyl gallate, ethyl gallate, hydroxy phenyl acetic acid, three phenylacetic acid isomers, 3-(4-hydroxyphenyl)propionic acid, and Homoveratric acid. With exception of chlorogenic and caffeic acid, most phenolic acids were quantified for the first time in apples. Significant varietal differences (p<0.05) were observed in both peel and pulp. The levels of total phenolic acids were higher in the pulp as compared to apple peel (dry weight) in all varieties. Coumaroylquinic, protocatechuic, 4-hydroxybenzoic, vanillic and t-ferulic acids were present in free forms. With exception of chlorogenic acid, all other phenolic acids were present only as bound forms.
Synthesis of homoveratric acid-imprinted polymers and their evaluation as selective separation materials.[Pubmed:21546882]
Molecules. 2011 May 5;16(5):3826-44.
A bulk polymerization method was used to easily and efficiently prepare Homoveratric acid (3,4-dimethoxyphenylacetic acid)-imprinted polymers from eight basic monomers: 2-vinylpyridine, 4-vinylpyridine, 1-vinylimidazole, N-allylaniline, N-allylpiperazine, allylurea, allylthiourea, and allylamine, in the presence of Homoveratric acid as a template in N,N-dimethylformamide as a porogen. The imprinted polymer prepared from allylamine had the highest affinity to the template, showing an imprinting factor of 3.43, and allylamine polymers MIP8/NIP8 were selected for further studies. Their binding properties were analyzed using the Scatchard method. The results showed that the imprinted polymers have two classes of heterogeneous binding sites characterized by two pairs of K(d), B(max) values: K(d)(1) = 0.060 mumol/mL, B(max)(1) = 0.093 mumol/mg for the higher affinity binding sites, and K(d)(2) = 0.455 mumol/mL, B(max)(2) = 0.248 mumol/mg for the lower affinity binding sites. Non-imprinted polymer has only one class of binding site, with K(d) = 0.417 mumol/mL and B(max) = 0.184 mumol/mg. A computational analysis of the energies of the prepolymerization complexes was in agreement with the experimental results. It showed that the selective binding interactions arose from cooperative three point interactions between the carboxylic acid and the two methoxy groups in the template and amino groups in the polymer cavities. Those results were confirmed by the recognition studies performed with the set of structurally related compounds. Allylamine polymer MIP8 had no affinity towards biogenic amines. The obtained imprinted polymer could be used for selective separation of Homoveratric acid.
Racemization-free synthesis of (S)-(+)-tylophorine from L-proline by radical cyclization.[Pubmed:20377275]
Org Lett. 2010 May 7;12(9):2140-1.
The phenanthroindolizidine alkaloid (S)-(+)-tylophorine was synthesized from L-proline in nine linear steps including a double bromination and a free-radical cyclization of an N-aziridinylimine as the key steps. The phenanthrene moiety was prepared from Homoveratric acid and veratraldehyde and permits the variation of each oxygen-substituted ring.
Enzyme production by Mycena galopus mycelium in artificial media and in Picea sitchensis F1 horizon needle litter.[Pubmed:14531622]
Mycol Res. 2003 Aug;107(Pt 8):996-1008.
Mycena galopus is among the most important leaf litter decomposers in UK coniferous and angiosperm woodlands, having the potential to utilise all the major constituents of plant litter. Even so, the enzyme or combination of enzymes produced by M. galopus responsible for lignin depolymerisation was previously unknown. A range of media from liquid and semi-solid cultures to more natural substrata was tested to determine whether laccase was produced by an isolate of M. galopus, M9053. Malt extract liquid medium (MEL) with 2,5-xylidine favoured laccase production as compared with the same medium containing the inducers veratryl alcohol, veratryl aldehyde, veratric acid, Homoveratric acid, vanillic acid or p-anisic acid. A semi-solid medium of cereal bran in phosphate buffer and a solid medium of Picea sitchensis F1 horizon needle litter were also not as effective as MEL with 2,5-xylidine as an inducer. Compared with six other isolates of the same species grown in MEL without inducers, M9053 exhibited rates of laccase activity fairly typical for M. galopus. An isolate from a dark coloured basidiome of M. galopus, but not var. nigra, exhibited the greatest activity while var. candida showed relatively low laccase activity. Marasmius androsaceus exhibited peak laccase production several days later than M. galopus. In addition, a manganese-dependent peroxidase that was responsible for 15% (in MEL culture fluid) and 39% (in needle litter extract III) of ligninolytic activity was produced by M9053. A further peroxidase was found to be the major ligninolytic constituent in MEL extracts (53%), and had a similar contribution to total activity (29%) as laccase (32%) in needle litter fraction III. Mycena galopus produced water- and buffer-extractable mannases and xylanases when grown on needle litter.
Quantitative investigation on the urinary excretion and metabolism of 3,4-dimethoxyphenylethylamine in schizophrenics and normal individuals.[Pubmed:7059639]
Biol Psychiatry. 1982 Jan;17(1):49-59.
A quantitative method for the detection of DMPEA in urine was developed. It is based on the fluorometric determination of DMPEA in the form of its phosphopyridoxyl derivate. The limit of detection is 2 microgram DMPEA per 1 g creatinine. The DMPEA content was measured in urine from healthy persons, from schizophrenics, and from psychiatric patients without schizophrenia hospitalized with the schizophrenics. From each person five to ten 24-hr urine samples were investigated. DMPEA could be found neither in schizophrenics nor in controls or healthy persons. Finally, the urinary excretion of parenterally applied 14C-DMPEA was determined in three healthy volunteers and in three rats. In man about 25% of the label was excreted as DMPEA. The main metabolite in urine was Homoveratric acid. Both compounds were excreted as conjugates.


