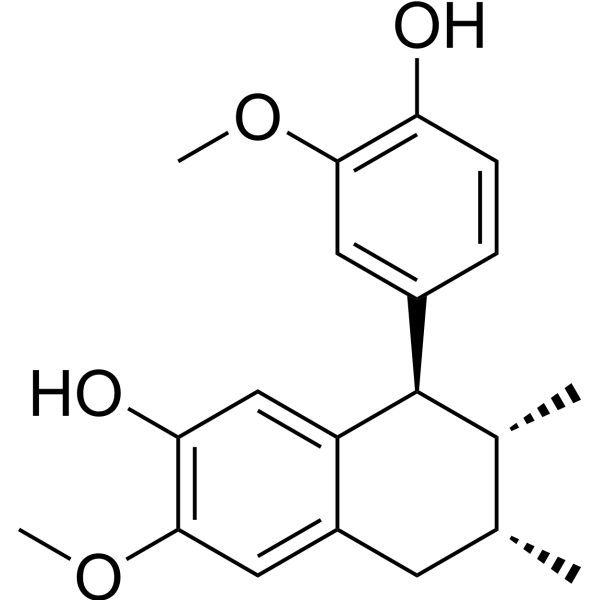
IsoguaiacinCAS# 78341-26-1 |
Quality Control & MSDS
Package In Stock
Number of papers citing our products

| Cas No. | 78341-26-1 | SDF | File under preparation. |
| PubChem ID | N/A | Appearance | Powder |
| Formula | C20H24O4 | M.Wt | 328.4 |
| Type of Compound | Lignanoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Isoguaiacin Dilution Calculator

Isoguaiacin Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.0451 mL | 15.2253 mL | 30.4507 mL | 60.9013 mL | 76.1267 mL |
| 5 mM | 0.609 mL | 3.0451 mL | 6.0901 mL | 12.1803 mL | 15.2253 mL |
| 10 mM | 0.3045 mL | 1.5225 mL | 3.0451 mL | 6.0901 mL | 7.6127 mL |
| 50 mM | 0.0609 mL | 0.3045 mL | 0.609 mL | 1.218 mL | 1.5225 mL |
| 100 mM | 0.0305 mL | 0.1523 mL | 0.3045 mL | 0.609 mL | 0.7613 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 5-Hydroxy-2-(2′-hydroxy-2-phenylethyl)chromone
Catalog No.:BCX1824
CAS No.:357637-15-1
- (S)-(+)-Imperanene
Catalog No.:BCX1823
CAS No.:163634-08-0
- Puberulin A
Catalog No.:BCX1822
CAS No.:164268-04-6
- (7S,8S,1′S)-3,4,1′-Trimethoxy-1′,6′-dihydro-7,4′-epoxy-8,3′-neoligna-8′-en-6′-one
Catalog No.:BCX1821
CAS No.:61240-30-0
- Grandivine A
Catalog No.:BCX1820
CAS No.:200574-22-7
- Manwuweizic acid
Catalog No.:BCX1819
CAS No.:116963-87-2
- Changnanic acid
Catalog No.:BCX1818
CAS No.:136040-44-3
- Foenumoside B
Catalog No.:BCX1817
CAS No.:877661-00-2
- Lysimachigenoside C
Catalog No.:BCX1816
CAS No.:952304-73-3
- Ipalbine
Catalog No.:BCX1815
CAS No.:23544-46-9
- Ipalbidine
Catalog No.:BCX1814
CAS No.:26294-41-7
- Piperkadsin A
Catalog No.:BCX1813
CAS No.:895543-36-9
- Rubriflorin B
Catalog No.:BCX1826
CAS No.:835875-96-2
- Kadsuralignan A
Catalog No.:BCX1827
CAS No.:913237-64-6
- Acetylbinankadsurin A
Catalog No.:BCX1828
CAS No.:77174-33-5
- Kadsulignan H
Catalog No.:BCX1829
CAS No.:144049-91-2
- Schiarisanrin E
Catalog No.:BCX1830
CAS No.:697228-90-3
- Schisanlignone B
Catalog No.:BCX1831
CAS No.:135459-86-8
- Elephanoside D
Catalog No.:BCX1832
CAS No.:1005340-21-5
- Interiorin D
Catalog No.:BCX1833
CAS No.:133360-40-4
- Schiarisanrin C
Catalog No.:BCX1834
CAS No.:130252-44-7
- Angeloylbinankadsurin A
Catalog No.:BCX1835
CAS No.:77165-80-1
- (+)-Osbeckic acid
Catalog No.:BCX1836
CAS No.:112923-64-5
- Kadsurenin C
Catalog No.:BCX1837
CAS No.:145722-88-9
3'-Demethoxy-6-O-Demethylisoguaiacin and Norisoguaiacin Nematocidal Lignans from Artemisia cina against Haemonchus contortus Infective Larvae.[Pubmed:36840168]
Plants (Basel). 2023 Feb 12;12(4):820.
Artemisia cina is a plant used in traditional Chinese medicine as a remedy for parasitic diseases. This study describes the isolation and chemical characterization of anthelmintic compounds of A. cina against Haemonchus contortus infective larvae (L(3)) through lethal testing. Previously, three extracts-n-hexane (HexAc), ethyl acetate (EtOAc) and methanol (MeOAc)-were evaluated at concentrations of 4 to 0.5 mg/mL, resulting in the HexAc extract with the greatest effect of 76.6% mortality of the larvae at 4 mg/mL. Then, this was chemically fractioned by polarity, obtaining seven fractions (C1F1-C1F7), and, when evaluated at concentrations from 2 to 0.25 mg/mL, the 2 mg/mL C1F5 fraction produced an effect against the nematode H. contortus of 100% mortality of the larvae. Thus, this fraction was fractionated again by column chromatography, obtaining twelve subfractions (C2F1-C2F12) which were evaluated from 1 to 0.125 mg/mL, with the C2F5 subfraction causing a nematicidal effect of 100% mortality. NMR analysis of one ((1)H, (13)C and DEPT) and two dimensions (COSY, HSQC and HMBC) and mass spectrometry of this fraction allowed us to identify the mixture of 3'-demethoxy-6-O-demethylIsoguaiacin and norIsoguaiacin. Therefore, it can be assumed that the mixture of these compounds is responsible for the anthelmintic effect. These results indicate that A. cina containing anthelmintic compounds and might be used as an antiparasitic drug against H. contortus.
An Artemisia cina n-hexane extract reduces the Haemonchus contortus and Teladorsagia circumcincta fecal egg count in naturally infected periparturient goats.[Pubmed:35138496]
Trop Anim Health Prod. 2022 Feb 9;54(2):95.
The objective of this study was to evaluate an n-hexane extract of Artemisia cina (Acn-h) as a natural anthelmintic treatment for periparturient goats naturally infected with the nematodes Haemonchus contortus and Teladorsagia circumcincta. A total of 200 periparturient Alpine and Nubian goats were used. Deworming criteria were based on the following parameters: fecal egg account (epg), ocular mucosa color (OMC), and body condition (BC). A previous analysis using coprocultures of the flock revealed the presence of H. contortus (80%) and T. circumcincta (20%). The Acn-h contained two new compounds identified by mass spectrometry data as Isoguaiacin and norIsoguaiacin at 284.14 and 315 m/z. The lethal effects of Acn-h at 0.5, 1, 2, and 4 mg/mL on H. contortus adult stages were 31.6, 66.5, 81.3, and 86.9%, respectively (p < 0.05), showing similar efficacy at 2 and 4 mg/mL with albendazole (positive control group). Then, two experimental groups, with 100 goats in peripartum in each, were distributed randomly and treated at day 0 as follows: group 1 = 4 mg/kg of Acn-h as single oral dose, and group 2 = control group, treated with water (as a placebo). The epg, OMC, and BC parameters were recorded at 0 (periparturient period), 7 (birth period), and 23 (postpartum) days and analyzed using a completely randomized design with Duncan's test for comparison of means and analysis of variance. The following epg reductions were recorded in the Acn-h-treated group as follows: 20.1 +/- 34.4 and 31.7 +/- 38.2% at days 7 and 23 compared to the control group. During the whole experiment, no significant differences in OMC or BC were observed in relation to the control group, excepting at day 23 (p < 0.05) for BC in the group treated with A. cina. Thus, Acn-h can be a useful natural alternative tool for the control of the nematodes H. contortus and T. circumcincta in periparturient goat flocks.
Potential Mechanism of Action of 3'-Demethoxy-6-O-demethyl-isoguaiacin on Methicillin Resistant Staphylococcus aureus.[Pubmed:26184132]
Molecules. 2015 Jul 8;20(7):12450-8.
Bacterial infections represent one of the main threats to global public health. One of the major causative agents associated with high morbidity and mortality infections in hospitals worldwide is methicillin-resistant Staphylococcus aureus. Therefore, there is a need to develop new antibacterial agents to treat these infections, and natural products are a rich source of them. In previous studies, we reported that lignan 3'-demethoxy-6-O-demethylIsoguaiacin, isolated and characterized from Larrea tridentate, showed the best activity towards methicillin-resistant S. aureus. Thus, the aim of this study was to determine the potential molecular mechanism of the antibacterial activity of 3'-demethoxy-6-O-demethylIsoguaiacin against methicillin-resistant S. aureus using microarray technology. Results of microarray genome expression were validated by real-time polymerase chain reaction (RT-PCR). The genetic profile expression results showed that lignan 3'-demethoxy-6-O-demethylIsoguaiacin had activity on cell membrane affecting proteins of the ATP-binding cassette (ABC) transport system causing bacteria death. This molecular mechanism is not present in any antibacterial commercial drug and could be a new target for the development of novel antibacterial agents.
Screening of phytochemicals against protease activated receptor 1 (PAR1), a promising target for cancer.[Pubmed:25007158]
J Recept Signal Transduct Res. 2015 Feb;35(1):26-45.
CONTEXT: Drug resistance and drug-associated toxicity are the primary causes for withdrawal of many drugs, although patient recovery is satisfactory in many instances. Interestingly, the use of phytochemicals in the treatment of cancer as an alternative to synthetic drugs comes with a host of advantages; minimum side effects, good human absorption and low toxicity to normal cells. Protease activated receptor 1 (PAR1) has been established as a promising target in many diseases including various cancers. Strong evidences suggest its role in metastasis also. OBJECTIVE: There are no natural compounds known to inhibit its activity, so we aimed to identify phytochemicals with antagonist activity against PAR1. METHODS: We screened phytochemicals from Naturally Occurring Plant-based Anticancer Compound-Activity-Target database (NPACT, http://crdd.osdd.net/raghava/npact/ ) against PAR1 using virtual screening workflow of Schrodinger software. It analyzes pharmaceutically relevant properties using Qikprop and calculates binding energy using Glide at three accuracy levels (high-throughput virtual screening, standard precision and extra precision). RESULTS AND CONCLUSION: Our study led to the identification of phytochemicals, which showed interaction with at least one experimentally determined active site residue of PAR1, showed no violations to Lipinski's rule of five along with predicted high human absorption. Furthermore, structural interaction fingerprint analysis indicated that the residues H255, D256, E260, S344, V257, L258, L262, Y337 and S344 may play an important role in the hydrogen bond interactions of the phytochemicals screened. Of these residues, H255 and L258 residues were experimentally proved to be important for antagonist binding. The residues Y183, L237, L258, L262, F271, L332, L333, Y337, L340, A349, Y350, A352, and Y353 showed maximum hydrophobic interactions with the phytochemicals screened. The results of this work suggest that phytochemicals Reissantins D, 24,25-dihydro-27-desoxywithaferin A, Isoguaiacin, 20-hydroxy-12-deoxyphorbol angelate, etc. could be potential antagonist of PAR1. However, further experimental studies are necessary to validate their antagonistic activity against PAR1.
[Lignans from Machilus robusta].[Pubmed:24010288]
Zhongguo Zhong Yao Za Zhi. 2013 Jun;38(11):1740-6.
Eighteen lignans were isolated from an ethanol extract of Machilus robusta by a combination of various chromatographic techniques including column chromatography over silica gel, Sephadex LH-20 and reversed-phase HPLC. Their structures were identified by spectroscopic data analysis as isolariciresinol-9'-O-beta-D-xylopyranoside (1), (+)-5'-methoxy-isolariciresinol-9'-O-beta-D-xylopyranoside(2), lyoniresinol-9'-O-beta-D-xylopyranoside(3), (+)-(8S, 8'S) -4, 4'-dihydroxy-3, 3', 5, 5'-tetramethoxylignan-9, 9'-diol 9-O-beta-D-xylopyranoside (ssioriside, 4), lyoniresinol (5), meso-dihydroguaiaretic acid (6), (+)-(8S, 8'R)-3', 4, 4'-trihydroxy-3'-methoxylignan (7), (8S, 8'R)-4'-hydroxy-3, 3', 4-trimethoxylignan (meso-monomethyl dihydroguaiaretic acid, 8), (+)-guaiacin (9), Isoguaiacin (10), (-)-(7'R, 8R, 8'R)-4, 4'-dihydroxy-3, 3', 5-trimethoxy-2, 7'-cyclolignan (11), henricine B (12), (-)-(7S, 7'S, 8R, 8'R)-4, 4'-dihydroxy-3, 3', 5, 5'-tetramethoxy-7, 7'-epoxylignan-9, 9'-dio] (7S, 7'S, 8R, 8'R-icariol A2, 13), (+)-(7R, 8R, 7'E)-4-hydroxy-3, 5'-dimethoxy-7, 4'-epoxy-8, 3'-neolignan-7'-ene (licarin A, 14), nectandrin B (15), machilin-I (16), (-)-pinoresinol (17), and (-)-syringaresinol (18). All compounds were isolated from this plant for the first time. In the preliminary assay, compound 17 showed inhibitory activity against NO secretion of mouse peritoneal macrophages with an inhibition rate of 72.2% at 10 micromol x L(-1).
Stimulatory activity of lignans from Machilus thunbergii on osteoblast differentiation.[Pubmed:17409528]
Biol Pharm Bull. 2007 Apr;30(4):814-7.
Phytoestrogens are naturally occurring compounds exerting estrogenic activity, and include isoflavonoids, flavonoids and lignans. In the present study, we evaluated the stimulating activity of six lignans, meso-dihydroguaiaretic acid, nordihydroguaiaretic acid, machilin A, guaiacin, Isoguaiacin and Isoguaiacin dimethylether, from Machilus thunbergii, on osteoblast differentiation employing primary cultures of mouse osteoblast as an in vitro assay system. Among the six lignans tested, arylnaphthalene type lignans such as guaiacin, Isoguaiacin and Isoguaiacin dimethylether significantly increased alkaline phosphatase activity, whereas bibenzylbutane type lignans such as meso-dihydroguaiaretic acid, nordihydroguaiaretic acid and machilin A showed little effects. Isoguaiacin and Isoguaiacin dimethylether also increased collagen synthesis as well as calcium deposition. In addition, treatment of the mouse osteoblasts with tamoxifen markedly reduced ALP activity increased by Isoguaiacin or Isoguaiacin dimethylether, suggesting the involvement of estrogen receptor in the action of these lignans on osteoblast differentiation. Taken together, these results suggest that arylnaphthalene type lignans such as guaiacin, Isoguaiacin and Isoguaiacin dimethylether significantly increase osteoblast differentiation.
Neuroprotective lignans from the bark of Machilus thunbergii.[Pubmed:14765301]
Planta Med. 2004 Jan;70(1):79-80.
The CH (2)Cl (2) fraction of the bark of Machilus thunbergii Sieb. et Zucc. (Lauraceae) significantly protected primary cultures of rat cortical cells exposed to the excitotoxic amino acid, L-glutamate. (-)-Isoguaiacin, meso-dihydroguaiaretic acid, machilin A, (+)-galbelgin, licarin A, (-)-sesamin, and (+)-guaiacin were isolated by bioactivity-guided fractionation from the CH (2)Cl (2) fraction and further separated using chromatographic techniques. Isoguaiacin, meso-dihydroguaiaretic acid, licarin A and (+)-guaiacin had significant neuroprotective activities against glutamate-induced neurotoxicity in primary cultures of rat cortical cells at concentrations ranging from 0.1 microM to 10.0 microM.
Antioxidant lignans from Machilus thunbergii protect CCl4-injured primary cultures of rat hepatocytes.[Pubmed:11045899]
J Pharm Pharmacol. 2000 Sep;52(9):1163-9.
Eleven lignans (1-11) were isolated from the CH2Cl2 fraction of the bark of Machilus thunbergii Sieb. et Zucc. (Lauraceae). These were identified as (-)-acuminatin (1), (-)-Isoguaiacin (2), meso-dihydroguaiaretic acid (3), (+)-galbacin (4), (-)-sesamin (5), (+)-galbelgin (6), machilin A (7), machilin G (8), licarin A (9), and nectandrin A (10) and B (11). Primary cultures of rat hepatocytes were co-incubated for 90 min with the hepatotoxin CCl4 and each of the 11 lignans (50 microM). Hepatoprotective activity was determined by measuring the level of glutamic pyruvic transaminase released into the medium from the primary cultures of rat hepatocytes. (-)-Acuminatin, (-)-Isoguaiacin and meso-dihydroguaiaretic acid all significantly reduced the level of glutamic pyruvic transaminase released. Further investigation revealed that these three compounds significantly preserved the levels and the activities of glutathione, superoxide dismutase, glutathione peroxidase and catalase. (-)-Acuminatin, (-)-Isoguaiacin and meso-dihydroguaiaretic acid also ameliorated lipid peroxidation as demonstrated by a reduction of malondialdehyde production. These results suggest that (-)-acuminatin, (-)-Isoguaiacin and meso-dihydroguaiaretic acid exert diverse hepatoprotective activities, perhaps by serving as potent antioxidants.
Chemical constituents of two Chinese Magnoliaceae plants, Tsoongiodendron odorum and Manglietiastrum sinicum, and their inhibition of platelet aggregation.[Pubmed:10985075]
Planta Med. 2000 Aug;66(6):511-5.
Phytochemical investigations of Tsoongiodendron odorum and Manglietiastrum sinicum, both Magnoliaceae, led to the isolation of twenty compounds in total. Among them, one was a new sesquiterpene, 11-O-oleoyl-beta-eudesmol (2), and another, 1-(3,4-dimethoxypheny)-4-(3,4-methylenedioxyphenyl)-2,3-dimethy lbutane (12) was isolated as a natural product for the first time. Moreover, 13C-NMR spectral data of Isoguaiacin (16) are reported here for the first time. Structure elucidations for compounds reported here were mainly based on their spectral data. The ethanolic extracts of T. odorum and M. sinicum, and six pure compounds, 4(15)-eudesmen-11-ol (beta-eudesmol) (1), 1 beta-hydroxy-4(15),11(13)-eudesmadien-12,6 alpha-olide (reynosin) (3), 3,11(13)-eudesmadien-12,6 alpha-olide (alpha-cyclocostunolide) (5), erythro-1-(4-hydroxy-3-methoxyphenyl)-4-(3,4-methylenedioxyphenyl)-2,3- dimethylbutane (11), nectandrin-B (18), and syringaresinol (19), displayed considerable inhibition against platelet aggregation induced by AA, by ADP, or by PAF.


