NeorauflavaneCAS# 53734-74-0 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 53734-74-0 | SDF | Download SDF |
| PubChem ID | 101643011 | Appearance | Powder |
| Formula | C21H22O5 | M.Wt | 354.4 |
| Type of Compound | Flavonoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
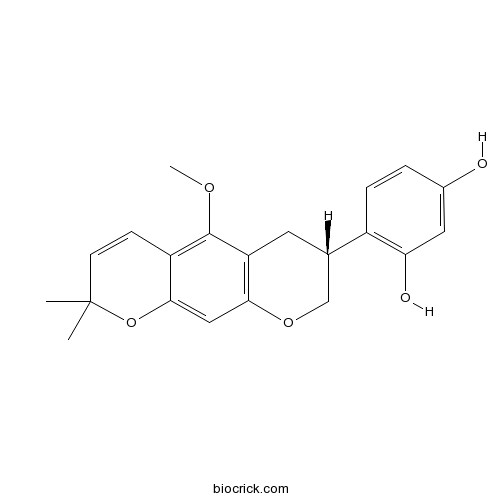
| Chemical Name | 4-[(7R)-5-methoxy-2,2-dimethyl-7,8-dihydro-6H-pyrano[3,2-g]chromen-7-yl]benzene-1,3-diol | ||
| SMILES | CC1(C=CC2=C(C3=C(C=C2O1)OCC(C3)C4=C(C=C(C=C4)O)O)OC)C | ||
| Standard InChIKey | FLKVRTIAGSMQLN-LBPRGKRZSA-N | ||
| Standard InChI | InChI=1S/C21H22O5/c1-21(2)7-6-15-19(26-21)10-18-16(20(15)24-3)8-12(11-25-18)14-5-4-13(22)9-17(14)23/h4-7,9-10,12,22-23H,8,11H2,1-3H3/t12-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. Neorauflavane shows tyrosinase inhibition, it can inhibit tyrosinase monophenolase activity with an IC50 of 30 nM. 2. Neorauflavane can efficiently reduce melanin content in B16 melanoma cells with 12.95 uM of IC50. |
| Targets | Tyrosinase |

Neorauflavane Dilution Calculator

Neorauflavane Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.8217 mL | 14.1084 mL | 28.2167 mL | 56.4334 mL | 70.5418 mL |
| 5 mM | 0.5643 mL | 2.8217 mL | 5.6433 mL | 11.2867 mL | 14.1084 mL |
| 10 mM | 0.2822 mL | 1.4108 mL | 2.8217 mL | 5.6433 mL | 7.0542 mL |
| 50 mM | 0.0564 mL | 0.2822 mL | 0.5643 mL | 1.1287 mL | 1.4108 mL |
| 100 mM | 0.0282 mL | 0.1411 mL | 0.2822 mL | 0.5643 mL | 0.7054 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Luteolin-7-O-glucoside
Catalog No.:BCN5388
CAS No.:5373-11-5
- Bruceantinol
Catalog No.:BCN7616
CAS No.:53729-52-5
- Oxfendazole
Catalog No.:BCC4817
CAS No.:53716-50-0
- Carprofen
Catalog No.:BCC4645
CAS No.:53716-49-7
- Tubacin
Catalog No.:BCC2428
CAS No.:537049-40-4
- BML-210(CAY10433)
Catalog No.:BCC6479
CAS No.:537034-17-6
- 2-Acetyl-5-bromothiophene
Catalog No.:BCC8513
CAS No.:5370-25-2
- Mexiletine HCl
Catalog No.:BCC4677
CAS No.:5370-01-4
- beta-Amyrenonol methylthiomethyl ether
Catalog No.:BCN3354
CAS No.:
- N-Acetyl-m-toluidine
Catalog No.:BCC9083
CAS No.:537-92-8
- Soyasaponin Be Methyl Ester
Catalog No.:BCN5925
CAS No.:117210-13-6
- N-Acetyl-L-tyrosine
Catalog No.:BCC9082
CAS No.:537-55-3
- Neorauflavene
Catalog No.:BCN4848
CAS No.:53734-75-1
- Lyclaninol
Catalog No.:BCN5710
CAS No.:53755-76-3
- Lycernuic acid A
Catalog No.:BCN5711
CAS No.:53755-77-4
- UF 010
Catalog No.:BCC6478
CAS No.:537672-41-6
- CP-724714
Catalog No.:BCC1188
CAS No.:537705-08-1
- Ribostamycin Sulfate
Catalog No.:BCC4710
CAS No.:53797-35-6
- DCC
Catalog No.:BCC2810
CAS No.:538-75-0
- 1-Benzothiophene-3-carbaldehyde
Catalog No.:BCC8454
CAS No.:5381-20-4
- Onitin
Catalog No.:BCN5712
CAS No.:53823-02-2
- Onitisin
Catalog No.:BCN5713
CAS No.:53823-03-3
- Trifloroside
Catalog No.:BCN6915
CAS No.:53823-10-2
- 3-(beta-D-Glucopyranosyloxy)-2-hydroxybenzoic acid methyl ester
Catalog No.:BCN7734
CAS No.:53827-68-2
Highly potent tyrosinase inhibitor, neorauflavane from Campylotropis hirtella and inhibitory mechanism with molecular docking.[Pubmed:26706112]
Bioorg Med Chem. 2016 Jan 15;24(2):153-9.
Tyrosinase inhibition may be a means to alleviate not only skin hyperpigmentation but also neurodegeneration associated with Parkinson's disease. In the course of metabolite analysis from tyrosinase inhibitory methanol extract (80% inhibition at 20 mug/ml) of Campylotropis hirtella, we isolated fourteen phenolic compounds, among which Neorauflavane 3 emerged as a lead structure for tyrosinase inhibition. Neorauflavane 3 inhibited tyrosinase monophenolase activity with an IC50 of 30 nM. Thus this compound is 400-fold more active than kojic acid. It also inhibited diphenolase (IC50=500 nM), significantly. Another potent inhibitor 1 (IC50=2.9 muM) was found to be the most abundant metabolite in C. hirtella. In kinetic studies, compounds 3 showed competitive inhibitory behavior against both monophenolase and diphenolase. It manifested simple reversible slow-binding inhibition against monophenolase with the following kinetic parameters: Ki(app)=1.48 nM, k3=0.0033 nM(-1) min(-1) and k4=0.0049 min(-1). Neorauflavane 3 efficiently reduced melanin content in B16 melanoma cells with 12.95 muM of IC50. To develop a pharmacophore model, we explored the binding mode of neuroflavane 3 in the active site of tyrosinase. Docking results show that resorcinol motif of B-ring and methoxy group in A-ring play crucial roles in the binding the enzyme.


