Schisantherin ECAS# 64917-83-5 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

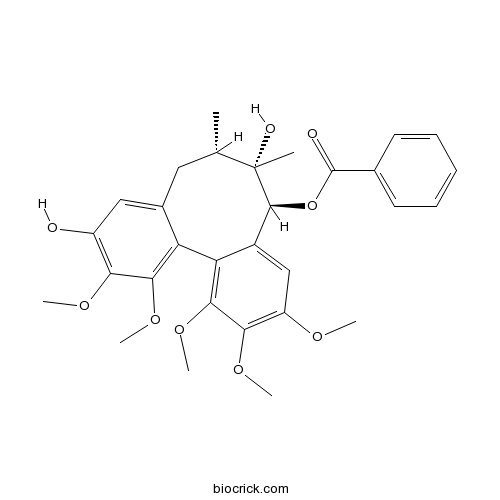
| Cas No. | 64917-83-5 | SDF | Download SDF |
| PubChem ID | 13844274 | Appearance | Powder |
| Formula | C30H34O9 | M.Wt | 538.6 |
| Type of Compound | Lignans | Storage | Desiccate at -20°C |
| Synonyms | Schizantherin-E | ||
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| SMILES | CC1CC2=CC(=C(C(=C2C3=C(C(=C(C=C3C(C1(C)O)OC(=O)C4=CC=CC=C4)OC)OC)OC)OC)OC)O | ||
| Standard InChIKey | ZNXDFTKQSCEJGE-DSASHONVSA-N | ||
| Standard InChI | InChI=1S/C30H34O9/c1-16-13-18-14-20(31)24(35-4)26(37-6)22(18)23-19(15-21(34-3)25(36-5)27(23)38-7)28(30(16,2)33)39-29(32)17-11-9-8-10-12-17/h8-12,14-16,28,31,33H,13H2,1-7H3/t16-,28-,30-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Schisantherin E is a natural compound isolated from the active fraction of the fruits of Schisandra sphenanthera Rehd. et Wils. |
| Structure Identification | Sci Sin. 1978 Jul-Aug;21(4):483-502.Studies on the active principles of Schisandra sphenanthera Rehd. et Wils. The structures of schisantherin A, B, C, D, E, and the related compounds.[Pubmed: 233922]Deoxyschisandrin (VIII) and five new lignans, named schisantherin A, schisantherin B, schisantherin C, schisantherin D, and Schisantherin E, were isolated from the active fraction of the fruits of Schisandra sphenanthera Rehd. et Wils. |

Schisantherin E Dilution Calculator

Schisantherin E Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.8567 mL | 9.2833 mL | 18.5667 mL | 37.1333 mL | 46.4166 mL |
| 5 mM | 0.3713 mL | 1.8567 mL | 3.7133 mL | 7.4267 mL | 9.2833 mL |
| 10 mM | 0.1857 mL | 0.9283 mL | 1.8567 mL | 3.7133 mL | 4.6417 mL |
| 50 mM | 0.0371 mL | 0.1857 mL | 0.3713 mL | 0.7427 mL | 0.9283 mL |
| 100 mM | 0.0186 mL | 0.0928 mL | 0.1857 mL | 0.3713 mL | 0.4642 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Schisantherin E is a natural compound isolated from the active fraction of the fruits of Schisandra sphenanthera Rehd. et Wils.
- Schisantherin D
Catalog No.:BCN7010
CAS No.:64917-82-4
- Boc-D-Trp(For)-OH
Catalog No.:BCC2598
CAS No.:64905-10-8
- BAY 61-3606 dihydrochloride
Catalog No.:BCC1407
CAS No.:648903-57-5
- trans-Norpterosin C
Catalog No.:BCN6918
CAS No.:64890-70-6
- Urapidil HCl
Catalog No.:BCC5044
CAS No.:64887-14-5
- Rubusoside
Catalog No.:BCN2313
CAS No.:64849-39-4
- AS-605240
Catalog No.:BCC2495
CAS No.:648450-29-7
- AS-604850
Catalog No.:BCC4989
CAS No.:648449-76-7
- ophocarpine hydrobromide
Catalog No.:BCN7541
CAS No.:78003-71-1
- Vitexin-2''-O-rhamnoside
Catalog No.:BCN5025
CAS No.:64820-99-1
- Neomangiferin
Catalog No.:BCN4970
CAS No.:64809-67-2
- Heraclenol acetonide
Catalog No.:BCN4192
CAS No.:64790-68-7
- Sendanolactone
Catalog No.:BCN4193
CAS No.:64929-59-5
- Schisantherin C
Catalog No.:BCN3621
CAS No.:64938-51-8
- Ditryptophenaline
Catalog No.:BCN7408
CAS No.:64947-43-9
- H-D-Asp(OtBu)-OH
Catalog No.:BCC2899
CAS No.:64960-75-4
- Brivanib (BMS-540215)
Catalog No.:BCC1231
CAS No.:649735-46-6
- Brivanib Alaninate (BMS-582664)
Catalog No.:BCC1240
CAS No.:649735-63-7
- Pifithrin-μ
Catalog No.:BCC2412
CAS No.:64984-31-2
- 7alpha-Hydroxystigmasterol
Catalog No.:BCN4194
CAS No.:64998-19-2
- 1-Testosterone
Catalog No.:BCC8474
CAS No.:65-06-5
- Yohimbine Hydrochloride
Catalog No.:BCN6268
CAS No.:65-19-0
- Pyridoxine
Catalog No.:BCC8355
CAS No.:65-23-6
- Phentolamine Mesylate
Catalog No.:BCC4353
CAS No.:65-28-1
Studies on the active principles of Schisandra sphenanthera Rehd. et Wils. The structures of schisantherin A, B, C, D, E, and the related compounds.[Pubmed:233922]
Sci Sin. 1978 Jul-Aug;21(4):483-502.
Deoxyschisandrin (VIII) and five new lignans, named schisantherin A, B, C, D, and E, were isolated from the active fraction of the fruits of Schisandra sphenanthera Rehd. et Wils. Their configurations and conformations were established by exhaustive spectral analysis as well as chemical degradations as shown in Ia, Ib; IIa, IIb; IIIa, IIIb; IVa, IVb, and Va, Vb respectively, and their absolute configurations at biphenyl, at C6, C7, and C8 were all assigned to be S form. The position of the methylenedioxyl group in the structures of gamma-schisandrin and Wuweizisu C (as described in the literature), isolated from Schisandra chinensis, must be corrected as shown in VI and VII respectively. In pharmacologica studies and preliminary clinical trials, schisantherin A, B, C, and D showed good effect in lowering the serum glutamic-pyruvic transaminase level of the patients suffering from chronic virus hepatitis. Schisantherin E and deoxyschisandrin were not effective.


