TrimethoxystilbeneCAS# 22255-22-7 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 22255-22-7 | SDF | Download SDF |
| PubChem ID | 5388063 | Appearance | Powder |
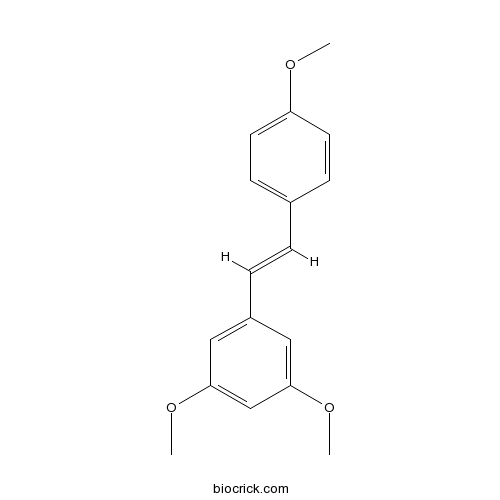
| Formula | C17H18O3 | M.Wt | 270.3 |
| Type of Compound | Polyphenols | Storage | Desiccate at -20°C |
| Synonyms | 3,4',5-Trimethoxystilbene | ||
| Solubility | DMSO : ≥ 50 mg/mL (184.97 mM) H2O : < 0.1 mg/mL (insoluble) *"≥" means soluble, but saturation unknown. | ||
| Chemical Name | 1,3-dimethoxy-5-[(E)-2-(4-methoxyphenyl)ethenyl]benzene | ||
| SMILES | COC1=CC=C(C=C1)C=CC2=CC(=CC(=C2)OC)OC | ||
| Standard InChIKey | GDHNBPHYVRHYCC-SNAWJCMRSA-N | ||
| Standard InChI | InChI=1S/C17H18O3/c1-18-15-8-6-13(7-9-15)4-5-14-10-16(19-2)12-17(11-14)20-3/h4-12H,1-3H3/b5-4+ | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 3,4',5-Trimethoxystilbene, an inhibitor of tubulin polymerization, which exerts antitumor, anti-HCV, antiallergic, anti-mitotic properties, it also exerts antiangiogenic and vascular-disrupting effects in zebrafish through the downregulation of VEGFR2 and cell-cycle modulation. 3,4',5-Trimethoxystilbene has anti-inflammatory activity, the ability of it to induce HO-1 expression may provide one of possible mechanisms of its anti-inflammatory action. |
| Targets | HCV | AMPK | mTOR | Calcium Channel | TNF-α | HO-1 | IL Receptor | VEGFR | HCV |
| In vitro | Structural modification of resveratrol leads to increased anti-tumor activity, but causes profound changes in the mode of action.[Pubmed: 26044878 ]Toxicol Appl Pharmacol. 2015 Aug 15;287(1):67-76.(Z)-3,5,4'-Trimethoxystilbene (3,4',5-Trimethoxystilbene, Z-TMS) is a resveratrol analog with increased antiproliferative activity towards a number of cancer cell lines compared to resveratrol, which has been shown to inhibit tubulin polymerization in vitro. The purpose of this study was to investigate if Z-TMS still shows potential for the prevention of metabolic diseases as known for resveratrol.
Differential effects of resveratrol and its natural analogs, piceatannol and 3,5,4'-trans-trimethoxystilbene, on anti-inflammatory heme oxigenase-1 expression in RAW264.7 macrophages.[Pubmed: 23861314 ]Biofactors. 2014 Jan-Feb;40(1):138-45.
beta-Cyclodextrins influence on E-3,5,4'-trimethoxystilbene absorption across biological membrane model: a differential scanning calorimetry evidence.[Pubmed: 20045042 ]Int J Pharm. 2010 Mar 30;388(1-2):144-50.E-3,5,4'-trimethoxystilbene (TMS) is a naturally occurring analog of resveratrol. The anti-neoplastic, antiallergic and anti-angiogenic activities of TMS have been recently reported.
|
| In vivo | (Z)-3,5,4'-Trimethoxystilbene Limits Hepatitis C and Cancer Pathophysiology by Blocking Microtubule Dynamics and Cell-Cycle Progression.[Pubmed: 27287718]Cancer Res. 2016 Aug 15;76(16):4887-96.Hepatocellular carcinoma (HCC) is the third most common cause of cancer-related deaths worldwide. Chronic hepatitis C virus (HCV) infection causes induction of several tumors/cancer stem cell (CSC) markers and is known to be a major risk factor for development of HCC. Therefore, drugs that simultaneously target viral replication and CSC properties are needed for a risk-free treatment of advanced stage liver diseases, including HCC.
|
| Kinase Assay | Novel role for TRPC4 in regulation of macroautophagy by a small molecule in vascular endothelial cells.[Pubmed: 25476892 ]Biochim Biophys Acta. 2015 Feb;1853(2):377-87.Macroautophagy (autophagy) is an important factor affecting the function of vascular endothelial cells (VECs) and must be tightly regulated in these cells. However, the precise mechanisms underlying this process, particularly in the presence of serum, remain obscure.
|
| Structure Identification | Biofactors. 2006;27(1-4):37-46.Anti-mitotic properties of resveratrol analog (Z)-3,5,4'-trimethoxystilbene.[Pubmed: 17012762]
|

Trimethoxystilbene Dilution Calculator

Trimethoxystilbene Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.6996 mL | 18.498 mL | 36.9959 mL | 73.9919 mL | 92.4898 mL |
| 5 mM | 0.7399 mL | 3.6996 mL | 7.3992 mL | 14.7984 mL | 18.498 mL |
| 10 mM | 0.37 mL | 1.8498 mL | 3.6996 mL | 7.3992 mL | 9.249 mL |
| 50 mM | 0.074 mL | 0.37 mL | 0.7399 mL | 1.4798 mL | 1.8498 mL |
| 100 mM | 0.037 mL | 0.185 mL | 0.37 mL | 0.7399 mL | 0.9249 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Trans-Trimethoxyresveratrol is a derivative of Resveratrol (RSV),and it may be a more potent anti-inflammatory, antiangiogenic and vascular-disrupting agent when compared with resveratrol. In vitro: The in vitro study of resveratrol and trans-Trimethoxyresveratrol showed rather weak cytotoxic effects on three cancer cell lines (HepG2, MCF-7, and MDA-MB-231), which contradicted a previous study reporting that resveratrol inhibited MCF-7 cells with an IC50 of about 10?μM. This discrepancy might be explained by the fact that the measurements were made 24?h after drug treatment, whereas the measurements of the previous study were taken 6 days after. The fact that the cytotoxic effect of trans-Trimethoxyresveratrol was lower than that of resveratrol is surprising, because in many studies, trans-Trimethoxyresveratrol is the most active analogue of resveratrol , although resveratrol shows much stronger antioxidant effects than that of trans-Trimethoxyresveratrol.[1] In vivo: Zebrafish embryos offer great advantage over their adults as well as other in vivo models because of the external development and optical transparency during their first few days, making them invaluable in the inspection of developmental processes. These unique advantages can even be made more useful when specific cell types are labeled with fluorescent probes. Zebrafish embryo in vivo, suggests that trans-Trimethoxyresveratrol has both more potent antiangiogenic activity and more importantly, stronger specific cytotoxic effects on endothelial cells than does resveratrol.[1]
References:
[1]. Alex, D. et al. Resveratrol derivative, trans-3,5,4'-trimethoxystilbene, exerts antiangiogenic and vascular-disrupting effects in zebrafish through the downregulation of VEGFR2 and cell-cycle modulation. Journal of cellular biochemistry 109, 339-346, doi:
- Guaijaverin
Catalog No.:BCN5056
CAS No.:22255-13-6
- alpha-Amyrin palmitate
Catalog No.:BCN5055
CAS No.:22255-10-3
- Methyl 6-hydroxyangolensate
Catalog No.:BCN5054
CAS No.:22255-07-8
- Ipratropium Bromide
Catalog No.:BCC3795
CAS No.:22254-24-6
- 7-Amino-3-methyl-3-cephem-4-carboxylic acid
Catalog No.:BCC8776
CAS No.:22252-43-3
- Fern-7-en-19-one
Catalog No.:BCN6443
CAS No.:222294-61-3
- Thalrugosaminine
Catalog No.:BCN7745
CAS No.:22226-73-9
- Cucurbitacin I
Catalog No.:BCC2439
CAS No.:2222-07-3
- Dehydroglaucine
Catalog No.:BCN2548
CAS No.:22212-26-6
- Dihydroseselin
Catalog No.:BCN8258
CAS No.:2221-66-1
- Naproxen
Catalog No.:BCC9091
CAS No.:22204-53-1
- Pyrantel Pamoate
Catalog No.:BCC4958
CAS No.:22204-24-6
- Lucidin 3-O-glucoside
Catalog No.:BCN8249
CAS No.:22255-29-4
- Loganic acid
Catalog No.:BCN5057
CAS No.:22255-40-9
- Adoprazine
Catalog No.:BCC1329
CAS No.:222551-17-9
- Tempol
Catalog No.:BCC4862
CAS No.:2226-96-2
- Bromocriptine mesylate
Catalog No.:BCC6642
CAS No.:22260-51-1
- Antidesmone
Catalog No.:BCN5058
CAS No.:222629-77-8
- ACBC
Catalog No.:BCC6584
CAS No.:22264-50-2
- Chysin A
Catalog No.:BCN2020
CAS No.:22269-11-0
- SZL P1-41
Catalog No.:BCC8004
CAS No.:222716-34-9
- Noladin ether
Catalog No.:BCC5756
CAS No.:222723-55-9
- Finasteride acetate
Catalog No.:BCC4204
CAS No.:222989-99-3
- SEA0400
Catalog No.:BCC1941
CAS No.:223104-29-8
The resveratrol analogue trimethoxystilbene inhibits cancer cell growth by inducing multipolar cell mitosis.[Pubmed:27739192]
Mol Carcinog. 2017 Mar;56(3):1117-1126.
Natural compounds are extensively studied for their potential use in traditional and non-traditional medicine. Several natural and synthetic Resveratrol analogues have shown interesting biological activities in the field of cancer chemoprevention. In the present study, we have focused on the ability of Resveratrol and two methoxylated derivatives (Trimethoxystilbene and Pterostilbene) to inhibit human cancer cell growth particularly analyzing their ability to interfere with tubulin dynamics at mitosis. We show that Trimethoxystilbene, differently from Resveratrol and Pterostilbene, alters microtubule polymerization dynamics in HeLa cells specifically inducing multipolar spindles and mitotic arrest coupled to a reduction of cell growth and an increase in apoptotic death by mitotic catastrophe. This work demonstrates that the structural modification of Rsv causes substantial changes in the mechanism of action of the derivatives. The presence of three extra methyl groups renders Trimethoxy very efficient in impairing cell proliferation by inducing mitotic catastrophe in cancer cells. (c) 2016 Wiley Periodicals, Inc.


