1,3,5-tricaffeoylquinic acidCAS# 1073897-80-9 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 1073897-80-9 | SDF | Download SDF |
| PubChem ID | 131636627 | Appearance | Powder |
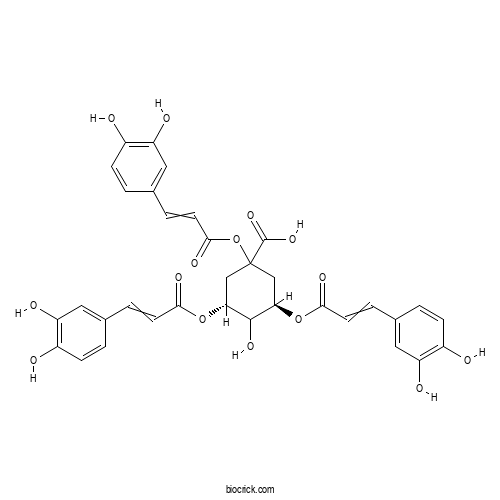
| Formula | C34H30O15 | M.Wt | 678.6 |
| Type of Compound | Caffeoylquinic acids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | (3R,5R)-1,3,5-tris[3-(3,4-dihydroxyphenyl)prop-2-enoyloxy]-4-hydroxycyclohexane-1-carboxylic acid | ||
| SMILES | C1C(C(C(CC1(C(=O)O)OC(=O)C=CC2=CC(=C(C=C2)O)O)OC(=O)C=CC3=CC(=C(C=C3)O)O)O)OC(=O)C=CC4=CC(=C(C=C4)O)O | ||
| Standard InChIKey | MSKVJEAKVWAQTA-QTLOJIOFSA-N | ||
| Standard InChI | InChI=1S/C34H30O15/c35-21-7-1-18(13-24(21)38)4-10-29(41)47-27-16-34(33(45)46,49-31(43)12-6-20-3-9-23(37)26(40)15-20)17-28(32(27)44)48-30(42)11-5-19-2-8-22(36)25(39)14-19/h1-15,27-28,32,35-40,44H,16-17H2,(H,45,46)/t27-,28-,32?,34?/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

1,3,5-tricaffeoylquinic acid Dilution Calculator

1,3,5-tricaffeoylquinic acid Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.4736 mL | 7.3681 mL | 14.7362 mL | 29.4724 mL | 36.8406 mL |
| 5 mM | 0.2947 mL | 1.4736 mL | 2.9472 mL | 5.8945 mL | 7.3681 mL |
| 10 mM | 0.1474 mL | 0.7368 mL | 1.4736 mL | 2.9472 mL | 3.6841 mL |
| 50 mM | 0.0295 mL | 0.1474 mL | 0.2947 mL | 0.5894 mL | 0.7368 mL |
| 100 mM | 0.0147 mL | 0.0737 mL | 0.1474 mL | 0.2947 mL | 0.3684 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Ginsenoside Ra3
Catalog No.:BCN0860
CAS No.:90985-77-6
- Ginsenoside Rh8
Catalog No.:BCN0859
CAS No.:343780-69-8
- Ginsenoside F11
Catalog No.:BCN0858
CAS No.:115038-42-1
- Cirenshenoside S
Catalog No.:BCN0857
CAS No.:226572-11-8
- Ginsenoyne E
Catalog No.:BCN0856
CAS No.:126146-63-2
- Notoginsenoside T1
Catalog No.:BCN0855
CAS No.:343962-53-8
- Ciwujianoside C3
Catalog No.:BCN0854
CAS No.:114906-74-0
- Ciwujianoside A1
Catalog No.:BCN0853
CAS No.:120768-65-2
- Siraitic acid A
Catalog No.:BCN0852
CAS No.:183374-15-4
- Siraitic acid B
Catalog No.:BCN0851
CAS No.:183374-16-5
- 11-Deoxymogroside IIIE
Catalog No.:BCN0850
CAS No.:1793003-47-0
- 11-Oxomogroside I
Catalog No.:BCN0849
CAS No.:918972-06-2
- Heratomol
Catalog No.:BCN0862
CAS No.:61265-07-4
- Secologanin
Catalog No.:BCN0863
CAS No.:19351-63-4
- Clinoposaponin D
Catalog No.:BCN0864
CAS No.:1822328-43-7
- Clinoposaponin VIII
Catalog No.:BCN0865
CAS No.:152020-04-7
- Clinoposaponin X
Catalog No.:BCN0866
CAS No.:159122-00-6
- Clinoposaponin XI
Catalog No.:BCN0867
CAS No.:159122-01-7
- Clinoposaponin I
Catalog No.:BCN0868
CAS No.:152580-76-2
- Clinoposaponin IX
Catalog No.:BCN0869
CAS No.:159121-99-0
- Clinoposaponin VI
Catalog No.:BCN0870
CAS No.:152020-03-6
- Clinopodiside B
Catalog No.:BCN0871
CAS No.:155762-41-7
- Ginkgolic Acid C17:2
Catalog No.:BCN0872
CAS No.:102811-39-2
- Onjisaponin A
Catalog No.:BCN0873
CAS No.:82410-33-1
Identification of anti-HIV active dicaffeoylquinic- and tricaffeoylquinic acids in Helichrysum populifolium by NMR-based metabolomic guided fractionation.[Pubmed:25841639]
Fitoterapia. 2015 Jun;103:155-64.
South Africa being home to more than 35% of the world's Helichrysum species (c.a. 244) of which many are used in traditional medicine, is seen potentially as a significant resource in the search of new anti-HIV chemical entities. It was established that five of the 30 Helichrysum species selected for this study had significant anti-HIV activity ranging between 12 and 21 mug/mL (IC50) by using an in-house developed DeCIPhR method on a full virus model. Subsequent toxicity tests also revealed little or no toxicity for these active extracts. With the use of NMR-based metabolomics, the search for common chemical characteristics within the plant extract was conducted, which resulted in specific chemical shift areas identified that could be linked to the anti-HIV activity of the extracts. The NMR chemical shifts associated with the activity were identified to be 2.56-3.08 ppm, 5.24-6.28 ppm, 6.44-7.04 ppm and 7.24-8.04 ppm. This activity profile was then used to guide the fractionation process by narrowing down and focusing the fractionation and purification processes to speed up the putative identification of five compounds with anti-HIV activity in the most active species, Helichrysum populifolium. The anti-HIV compounds identified for the first time from H. populifolium were three dicaffeoylquinic acid derivatives, i.e. 3,4-dicaffeoylquinic acid, 3,5-dicaffeoylquinic acid and 4,5-dicaffeoylquinic acid as well as two tricaffeoylquinic acid derivatives i.e. 1,3,5-tricaffeoylquinic acid and either 5-malonyl-1,3,4-tricaffeoylquinic or 3-malonyl-1,4,5-tricaffeoylquinic acid, with the latter being identified for the first time in the genus.


