6'''-FeruloylspinosinCAS# 77690-92-7 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 77690-92-7 | SDF | Download SDF |
| PubChem ID | 21597353 | Appearance | Yellow powder |
| Formula | C38H40O18 | M.Wt | 784.71 |
| Type of Compound | Flavonoids | Storage | Desiccate at -20°C |
| Synonyms | Spinosin B;319480-84-7 | ||
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
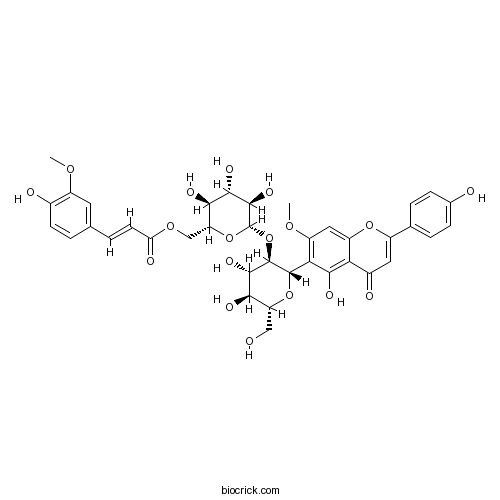
| Chemical Name | [(2R,3S,4S,5R,6S)-6-[(2S,3R,4S,5S,6R)-4,5-dihydroxy-2-[5-hydroxy-2-(4-hydroxyphenyl)-7-methoxy-4-oxochromen-6-yl]-6-(hydroxymethyl)oxan-3-yl]oxy-3,4,5-trihydroxyoxan-2-yl]methyl (E)-3-(4-hydroxy-3-methoxyphenyl)prop-2-enoate | ||
| SMILES | COC1=C(C=CC(=C1)C=CC(=O)OCC2C(C(C(C(O2)OC3C(C(C(OC3C4=C(C=C5C(=C4O)C(=O)C=C(O5)C6=CC=C(C=C6)O)OC)CO)O)O)O)O)O)O | ||
| Standard InChIKey | WZAXZHIVHPRTIU-IHIXZLSHSA-N | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. 6'''-Feruloylspinosin may possess anti-inflammatory activity. |
| Targets | NF-kB | p65 |

6'''-Feruloylspinosin Dilution Calculator

6'''-Feruloylspinosin Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.2744 mL | 6.3718 mL | 12.7436 mL | 25.4871 mL | 31.8589 mL |
| 5 mM | 0.2549 mL | 1.2744 mL | 2.5487 mL | 5.0974 mL | 6.3718 mL |
| 10 mM | 0.1274 mL | 0.6372 mL | 1.2744 mL | 2.5487 mL | 3.1859 mL |
| 50 mM | 0.0255 mL | 0.1274 mL | 0.2549 mL | 0.5097 mL | 0.6372 mL |
| 100 mM | 0.0127 mL | 0.0637 mL | 0.1274 mL | 0.2549 mL | 0.3186 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Enoximone
Catalog No.:BCC7155
CAS No.:77671-31-9
- ent-11,16-Epoxy-15-hydroxykauran-19-oic acid
Catalog No.:BCN1355
CAS No.:77658-46-9
- ent-9-Hydroxy-15-oxo-19-kauranoic acid
Catalog No.:BCN1356
CAS No.:77658-45-8
- ent-9-Hydroxy-15-oxo-16-kauren-19-oic acid
Catalog No.:BCN1357
CAS No.:77658-39-0
- Pterokaurene L3
Catalog No.:BCN4583
CAS No.:77658-38-9
- Boc-D-Ala-OH
Catalog No.:BCC3049
CAS No.:7764-95-6
- Toddanone
Catalog No.:BCN3430
CAS No.:77636-08-9
- 1,4,5,6-Tetrahydroxy-7,8-diprenylxanthone
Catalog No.:BCN1358
CAS No.:776325-66-7
- Fmoc-HomoArg-OH
Catalog No.:BCC2645
CAS No.:776277-76-0
- Isoscopoletin
Catalog No.:BCN4582
CAS No.:776-86-3
- Nestoron
Catalog No.:BCC1797
CAS No.:7759-35-5
- Ingenol-3,4:5,20-diacetonide
Catalog No.:BCN2959
CAS No.:77573-44-5
- Arctigenin
Catalog No.:BCN6291
CAS No.:7770-78-7
- (-)-Toddanol
Catalog No.:BCN3429
CAS No.:77715-99-2
- 1,7-Dihydroxy-3-methoxy-2-prenylxanthone
Catalog No.:BCN1354
CAS No.:77741-58-3
- 1,2-Dihydrotanshinquinone
Catalog No.:BCN2477
CAS No.:77769-21-2
- Potassium phosphate monobasic
Catalog No.:BCC7583
CAS No.:7778-77-0
- Dehydrocrebanine
Catalog No.:BCN4328
CAS No.:77784-22-6
- Carasinol B
Catalog No.:BCN8226
CAS No.:777857-86-0
- GNF 2
Catalog No.:BCC3891
CAS No.:778270-11-4
- GNF 5
Catalog No.:BCC3892
CAS No.:778277-15-9
- Nelumol A
Catalog No.:BCN4749
CAS No.:77836-86-3
- (1R)-(+)-Alpha-Pinene
Catalog No.:BCC8275
CAS No.:7785-70-8
- Oglemilast
Catalog No.:BCC1817
CAS No.:778576-62-8
A novel spinosin derivative from Semen Ziziphi Spinosae.[Pubmed:22115038]
J Asian Nat Prod Res. 2011 Dec;13(12):1151-7.
A novel spinosin derivative, 6'''-(4''''-O-beta-D-glucopyranosyl)-vanilloyl spinosin (1) was isolated from the methanol extract of Semen Ziziphi Spinosae, together with five known flavonoids, swertish, spinosin, 6'''-feruloylspinosin, isospinosin and kaempferol 3-O-alpha-L-rhamnopyranosyl-(1 --> 2)-O-[O-alpha-L-rhamnopyranosyl-(1 --> 6)]-beta-D-glucopyranoside, and two alkanoids, zizyphusine and 6-(2',3'-dihydroxyl-4'-hydroxymethyl-tetrahydro-furan-1'-yl)-cyclopentene[c]pyrro le-1,3-diol. The structure of compound 1 was elucidated by spectroscopic methods including UV, IR, ESI-TOF-MS, 1D, and 2D NMR techniques.
Evaluating ancient Egyptian prescriptions today: Anti-inflammatory activity of Ziziphus spina-christi.[Pubmed:26969383]
Phytomedicine. 2016 Mar 15;23(3):293-306.
BACKGROUND: Ziziphus spina-christi (L.) Desf. (Christ's Thorn Jujube) is a wild tree today found in Jordan, Israel, Egypt, and some parts of Africa, which was already in use as a medicinal plant in Ancient Egypt. In ancient Egyptian prescriptions, it was used in remedies against swellings, pain, and heat, and thus should have anti-inflammatory effects. Nowadays, Z. spina-christi, is used in Egypt (by Bedouins, and Nubians), the Arabian Peninsula, Jordan, Iraq, and Morocco against a wide range of illnesses, most of them associated with inflammation. Pharmacological research undertaken to date suggests that it possesses anti-inflammatory, hypoglycemic, hypotensive and anti-microbial effects. The transcription factor NF-kappaB (nuclear factor kappa-light-chain-enhancer of activated B cells) is critical in inflammation, proliferation and involved in various types of cancer. Identification of new anti-inflammatory compounds might be an effective strategy to target inflammatory disorders and cancer. Therefore, extracts from Z. spina-christi are investigated in terms of their anti-inflammatory effects. Our intention is to evaluate the effects of Z. spina-christi described in ancient Egyptian papyri, and to show whether the effects can be proven with modern pharmacological methods. Furthermore, we determine the active ingredients in crude extracts for their inhibitory activity toward NF-kappaB pathway. MATERIALS AND METHODS: To determine the active ingredients of Z. spina-christi, we fractionated the extracts for bioassays and identified the active compounds. Epigallocatechin, gallocatechin, spinosin, 6''' feruloylspinosin and 6''' sinapoylspinosin and crude extracts of seed, leaf, root or stem were analyzed for their effect on NF-kappaB DNA binding by electromobility shift assay (EMSA) and nuclear translocation of NF-kappaB-p65 by Western blot analysis. The binding mode of the compounds to NF-kappaB pathway proteins was compared with the known inhibitor, MG-132, by in silico molecular docking calculations. Log10IC50 values of gallocatechin and epigallocatechin as two main compounds of the plant were correlated to the microarray-based mRNA expression of 79 inflammation-related genes in cell lines of the National Cancer Institute (NCI, USA) as determined. The expression of 17 genes significantly correlated to the log10IC50 values for gallocatechin or epigallocatechin. RESULTS: Nuclear p65 protein level decreased upon treatment with each extract and compound. Root and seed extracts inhibited NF-kappaB-DNA binding as shown by EMSA. The compounds showed comparable binding energies and similar docking poses as MG-132 on the target proteins. CONCLUSION: Z. spina-christi might possess anti-inflammatory activity as assumed by ancient Egyptian prescriptions. Five compounds contributed to this bioactivity, i.e. epigallocatechin, gallocatechin, spinosin, 6''' feruloylspinosin and 6''' sinapoylspinosin as shown in vitro and in silico.
Distribution of free amino acids, flavonoids, total phenolics, and antioxidative activities of Jujube (Ziziphus jujuba) fruits and seeds harvested from plants grown in Korea.[Pubmed:21574660]
J Agric Food Chem. 2011 Jun 22;59(12):6594-604.
Fruit pulp and seeds from the jujube plant possess nutritional and medicinal properties. The bioactive components have been shown to vary both with cultivar and with growing conditions. Most studies report the components of varieties from China. We measured free amino acid, individual phenolic, and total phenolic content, and antioxidative activities in three jujube fruit pulp extracts from Boeun-deachu, Mechu, and Sanzoin cultivars and two seed extracts (Mechu and Sanzoin) from plants grown in Korea. In g/100 g dry weight, total free amino acid content measured by ion-exchange chromatography ranged from 5.2 to 9.8 in the pulp and from 4.0 to 5.3 in the seed. Total phenolic content measured by Folin-Ciocalteu ranged from 1.1 to 2.4 in the pulp and from 3.6 to 4.6 in the seed. Flavonoids were measured by HPLC and ranged from 0.7 to 1.8 in the pulp and from 3.2 to 4.0 in the seed. Flavonoids were identified by HPLC elution position and UV/vis and mass spectra. Fruits contained the following flavonoids: procyanidin B2, epicatechin, quercetin-3-O-rutinoside (Q-3-R), quercetin-3-O-galactoside (Q-3-G), kaempferol-glucosyl-rhamnoside (K-G-R), and two unidentified compounds. Seeds contained the following flavonoids: saponarin, spinosin, vitexin, swertish, 6'''-hydroxybenzoylspinosin (6'''-HBS), 6'''-feruloylspinosin (6'''-FS), and one unidentified substance. Dimensions and weights of the fresh fruit samples affected phenolic content. The distribution of the individual flavonoids among the different samples varied widely. Data determined by the FRAP antioxidative assay were well correlated with total phenolic content. In a departure from other studies, data from the DPPH free radical assay were not correlated with FRAP or with any of the measured compositional parameters. Because individual jujube flavonoids are reported to exhibit different health-promoting effects, knowledge of the composition and concentration of bioactive compounds of jujube products can benefit consumers.


