Ciwujianoside BCAS# 114902-16-8 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 114902-16-8 | SDF | Download SDF |
| PubChem ID | 195123 | Appearance | Powder |
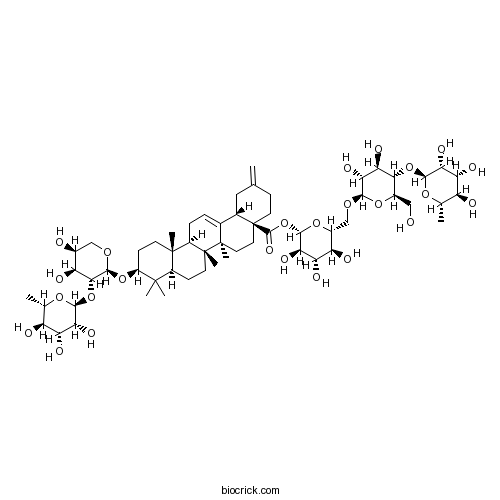
| Formula | C58H92O25 | M.Wt | 1189.35 |
| Type of Compound | Triterpenoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | [(2S,3R,4S,5S,6R)-6-[[(2R,3R,4R,5S,6R)-3,4-dihydroxy-6-(hydroxymethyl)-5-[(2S,3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxyoxan-2-yl]oxymethyl]-3,4,5-trihydroxyoxan-2-yl] (4aS,6aR,6aS,6bR,8aR,10S,12aR,14bS)-10-[(2S,3R,4S,5S)-4,5-dihydroxy-3-[(2S,3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxyoxan-2-yl]oxy-6a,6b,9,9,12a-pentamethyl-2-methylidene-1,3,4,5,6,6a,7,8,8a,10,11,12,13,14b-tetradecahydropicene-4a-carboxylate | ||
| SMILES | CC1C(C(C(C(O1)OC2C(OC(C(C2O)O)OCC3C(C(C(C(O3)OC(=O)C45CCC(=C)CC4C6=CCC7C8(CCC(C(C8CCC7(C6(CC5)C)C)(C)C)OC9C(C(C(CO9)O)O)OC1C(C(C(C(O1)C)O)O)O)C)O)O)O)CO)O)O)O | ||
| Standard InChIKey | UPROOJBJZLZCGS-CHTHVDMYSA-N | ||
| Standard InChI | InChI=1S/C58H92O25/c1-23-11-16-58(53(73)83-51-44(71)40(67)37(64)30(79-51)22-75-48-45(72)41(68)46(29(20-59)78-48)81-49-42(69)38(65)34(61)24(2)76-49)18-17-56(7)26(27(58)19-23)9-10-32-55(6)14-13-33(54(4,5)31(55)12-15-57(32,56)8)80-52-47(36(63)28(60)21-74-52)82-50-43(70)39(66)35(62)25(3)77-50/h9,24-25,27-52,59-72H,1,10-22H2,2-8H3/t24-,25-,27-,28-,29+,30+,31-,32+,33-,34-,35-,36-,37+,38+,39+,40-,41+,42+,43+,44+,45+,46+,47+,48+,49-,50-,51-,52-,55-,56+,57+,58-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Ciwujianoside B can relieve fatigue, enhance memory, and improve human cognition, it may enhance reuse of choline to accelerate the synthesis of acetylcholine in hippocampal neurons by activation of cholinesterase. Ciwujianoside B has inhibitory effects on apoptosis induced by MIRI and cardiomyocytes apoptosis induced by H 2 O 2 in rats; it also shows radioprotective effects on the hematopoietic system in mice, which is associated with changes in the cell cycle, a reduction in DNA damage, and down-regulation of the ratio of Bax/Bcl-2 in bone marrow cells exposed to radiation. |
| Targets | P450 (e.g. CYP17) | AChR | NF-kB | Bcl-2/Bax |
| In vitro | Development of sample preparation method for Ciwujianoside B and E analysis in Acanthopanax senticosus by ionic liquids-ultrasound based extraction and high-performance liquid chromatography detection.[Pubmed: 23870977]Food Chem. 2013 Dec 1;141(3):2426-33.An ionic liquids-based ultrasonic-assisted extraction (ILUAE) method was successfully developed for extracting eleutheroside B and E from Radix Acanthopanax senticosus. |
| In vivo | Ciwujianoside B or E enhances learning and memory in experimentally aged rats.[Pubmed: 25206404]Neural Regen Res. 2013 Apr 25;8(12):1103-12.Eleutheroside B or E, the main component of Acanthopanax, can relieve fatigue, enhance memory, and improve human cognition. Numerous studies have confirmed that high doses of acetylcholine significantly attenuate clinical symptoms and delay the progression of Alzheimer's disease. |
| Kinase Assay | Effects of Ciwujianoside B and eleutheroside E on activity of cytochrome P450 in rat liver microsomes.[Pubmed: 24383621]BMC Complement Altern Med. 2014 Jan 2;14:1.Chemicals of herbal products may cause unexpected toxicity or adverse effect by the potential for alteration of the activity of CYP450 when co-administered with other drugs. Eleutherococcus senticosus (ES), has been widely used as a traditional herbal medicine and popular herbal dietary supplements, and often co-administered with many other drugs. The main bioactive constituents of ES were considered to be eleutherosides including eleutheroside B (EB) and eleutheroside E (EE). This study was to investigate the effects of EB and EE on CYP2C9, CYP2D6, CYP2E1 and CYP3A4 in rat liver microsomes in vitro.
|
| Animal Research | Comparative investigations on the protective effects of rhodioside, ciwujianoside-B and astragaloside IV on radiation injuries of the hematopoietic system in mice.[Pubmed: 21031634]Phytother Res. 2011 May;25(5):644-53.The aim of this study was to investigate the protective effects of three glycosides (rhodioside, Ciwujianoside B and astragaloside IV) on the hematopoietic system in the mice exposed to γ-rays, and to examine the possible mechanisms involved. |

Ciwujianoside B Dilution Calculator

Ciwujianoside B Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 0.8408 mL | 4.204 mL | 8.408 mL | 16.8159 mL | 21.0199 mL |
| 5 mM | 0.1682 mL | 0.8408 mL | 1.6816 mL | 3.3632 mL | 4.204 mL |
| 10 mM | 0.0841 mL | 0.4204 mL | 0.8408 mL | 1.6816 mL | 2.102 mL |
| 50 mM | 0.0168 mL | 0.0841 mL | 0.1682 mL | 0.3363 mL | 0.4204 mL |
| 100 mM | 0.0084 mL | 0.042 mL | 0.0841 mL | 0.1682 mL | 0.2102 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Z-Val-OH
Catalog No.:BCC2734
CAS No.:1149-26-4
- Trabectedin
Catalog No.:BCC2012
CAS No.:114899-77-3
- Boc-Phe(3-Cl)-OH
Catalog No.:BCC2640
CAS No.:114873-03-9
- Boc-Phe(2-F)-OH
Catalog No.:BCC3223
CAS No.:114873-00-6
- ST-836
Catalog No.:BCC1968
CAS No.:1148156-63-1
- Z-Pro-OH
Catalog No.:BCC2754
CAS No.:1148-11-4
- Losartan
Catalog No.:BCC4090
CAS No.:114798-26-4
- 4-Bromomethyl-2-cyanobiphenyl
Catalog No.:BCC8702
CAS No.:114772-54-2
- tert-Butyl4'-(bromomethyl)biphenyl-2-carboxylate
Catalog No.:BCC9164
CAS No.:114772-40-6
- Methyl 4'-bromomethyl biphenyl-2-carboxylate
Catalog No.:BCC9039
CAS No.:114772-38-2
- Methyl 4'-methylbiphenyl-2-carboxylate
Catalog No.:BCC9040
CAS No.:114772-34-8
- 2-(2-Aminobenzoyl)-benzoic acid
Catalog No.:BCC8476
CAS No.:1147-43-9
- Ciwujianoside E
Catalog No.:BCN3505
CAS No.:114912-36-6
- Docetaxel intermediate
Catalog No.:BCN8360
CAS No.:114915-14-9
- 2-Chloro-1-(5'-(prop-1-ynyl)-2,2'-bithiophen-5-yl)ethanol
Catalog No.:BCN1614
CAS No.:114916-00-6
- N1,N10-Bis(p-coumaroyl)spermidine
Catalog No.:BCN6027
CAS No.:114916-05-1
- XL-888
Catalog No.:BCC2339
CAS No.:1149705-71-4
- Docetaxel
Catalog No.:BCN5342
CAS No.:114977-28-5
- Bromophenol Blue
Catalog No.:BCC8029
CAS No.:115-39-9
- Azacyclonol
Catalog No.:BCC4761
CAS No.:115-46-8
- Sinomenine
Catalog No.:BCN6265
CAS No.:115-53-7
- Ambenonium dichloride
Catalog No.:BCC6630
CAS No.:115-79-7
- Linalyl Acetate
Catalog No.:BCC8200
CAS No.:115-95-7
- 9-Phenylcarbazole
Catalog No.:BCN2259
CAS No.:1150-62-5
Effects of eleutheroside B and eleutheroside E on activity of cytochrome P450 in rat liver microsomes.[Pubmed:24383621]
BMC Complement Altern Med. 2014 Jan 2;14:1.
BACKGROUND: Chemicals of herbal products may cause unexpected toxicity or adverse effect by the potential for alteration of the activity of CYP450 when co-administered with other drugs. Eleutherococcus senticosus (ES), has been widely used as a traditional herbal medicine and popular herbal dietary supplements, and often co-administered with many other drugs. The main bioactive constituents of ES were considered to be eleutherosides including eleutheroside B (EB) and eleutheroside E (EE). This study was to investigate the effects of EB and EE on CYP2C9, CYP2D6, CYP2E1 and CYP3A4 in rat liver microsomes in vitro. METHOD: Probe drugs of tolbutamide (TB), dextromethorphan (DM), chlorzoxazone (CLZ) and testosterone (TS) as well as eleutherosides of different concentrations were added to incubation systems of rat liver microsomes in vitro. After incubation, validated HPLC methods were used to quantify relevant metabolites. RESULTS: The results suggested that EB and EE exhibited weak inhibition against the activity of CYP2C9 and CYP2E1, but no effects on CYP2D6 and CYP3A4 activity. The IC50 values for EB and EE were calculated to be 193.20 muM and 188.36 muM for CYP2E1, 595.66 muM and 261.82 muM for CYP2C9, respectively. Kinetic analysis showed that inhibitions of CYP2E1 by EB and EE were best fit to mixed-type with Ki value of 183.95 muM and 171.63 muM, respectively. CONCLUSIONS: These results indicate that EB and EE may inhibit the metabolism of drugs metabolized via CYP2C9 and CYP2E1, and have the potential to increase the toxicity of the drugs.
Eleutheroside B or E enhances learning and memory in experimentally aged rats.[Pubmed:25206404]
Neural Regen Res. 2013 Apr 25;8(12):1103-12.
Eleutheroside B or E, the main component of Acanthopanax, can relieve fatigue, enhance memory, and improve human cognition. Numerous studies have confirmed that high doses of acetylcholine significantly attenuate clinical symptoms and delay the progression of Alzheimer's disease. The present study replicated a rat model of aging induced by injecting quinolinic acid into the hippocampal CA1 region. These rats were intraperitoneally injected with low, medium and high doses of eleutheroside B or E (50, 100, 200 mg/kg), and rats injected with Huperzine A or PBS were used as controls. At 4 weeks after administration, behavioral tests showed that the escape latencies and errors in searching for the platform in a Morris water maze were dose-dependently reduced in rats treated with medium and high-dose eleutheroside B or E. Hematoxylin-eosin staining showed that the number of surviving hippocampal neurons was greater and pathological injury was milder in three eleutheroside B or E groups compared with model group. Hippocampal homogenates showed enhanced cholinesterase activity, and dose-dependent increases in acetylcholine content and decreases in choline content following eleutheroside B or E treatment, similar to those seen in the Huperzine A group. These findings indicate that eleutheroside B or E improves learning and memory in aged rats. These effects of eleutheroside B or E may be mediated by activation of cholinesterase or enhanced reuse of choline to accelerate the synthesis of acetylcholine in hippocampal neurons.
Development of sample preparation method for eleutheroside B and E analysis in Acanthopanax senticosus by ionic liquids-ultrasound based extraction and high-performance liquid chromatography detection.[Pubmed:23870977]
Food Chem. 2013 Dec 1;141(3):2426-33.
An ionic liquids-based ultrasonic-assisted extraction (ILUAE) method was successfully developed for extracting eleutheroside B and E from Radix Acanthopanax senticosus. Thirteen 1-alkyl-3-methylimidazolium ionic liquids with different cations and anions were investigated and 1-butyl-3-methylimidazolium bromide ([C4mim]Br) solution was selected as the solvent. The conditions for ILUAE, including the ionic liquid concentration, soaking time, ultrasonic power, ultrasonic time, solid-liquid ratio and number of extraction cycles, were optimized. With the proposed method, the energy consumption time was reduced to 30 min, whereas conventional method requires about 4h. The proposed method had good recovery (97.96-103.39%) and reproducibility (RSD, n=5; 3.3% for eleutheroside B, 4.6% for eleutheroside E). ILUAE was an efficient, rapid and simple sample preparation technique that showed high reproducibility and was environmental friendly.
Comparative investigations on the protective effects of rhodioside, ciwujianoside-B and astragaloside IV on radiation injuries of the hematopoietic system in mice.[Pubmed:21031634]
Phytother Res. 2011 May;25(5):644-53.
The aim of this study was to investigate the protective effects of three glycosides (rhodioside, ciwujianoside-B and astragaloside IV) on the hematopoietic system in the mice exposed to gamma-rays, and to examine the possible mechanisms involved. Mice were pretreated with the glycosides (40 mg/kg, i.g.) daily for 7 days prior to radiation. The survival of mice pretreated with three glycosides after total body irradiation (6.0 Gy) was examined. Peripheral blood leucocytes and endogenous spleen colony counts, colony-forming unit-granulocyte macrophage assay, analysis of DNA content and apoptosis rate determination were performed to evaluate the effects of the three glycosides on hematogenesis. The fragmentation of double-stranded DNA in lymphocytes was detected by the comet assay. The changes in cell cycle were analysed by flow cytometry. Furthermore, the expression levels of Bcl-2, Bax and nuclear factor-kappa B (NF-kappaB) were measured by western blot and the electrophoretic mobility shift assay. The results showed that pretreatment with all of the glycosides improved survival time and increased the number of leucocytes, spleen colonies and granulocyte-macrophage colonies in mice exposed to 6.0 Gy gamma-radiation. Rhodioside showed more protective efficacy than both ciwujianoside-B and astragaloside IV. All three glycosides significantly increased the proliferation abilities of bone marrow cells, and decreased the ratio of cells in G(0)/G(1) phase. Further analysis showed that these three glycosides were able to decrease DNA damage and the increment in the Bax/Bcl-2 ratio induced by radiation. In summary, the three glycosides showed radioprotective effects on the hematopoietic system in mice, which was associated with changes in the cell cycle, a reduction in DNA damage, and down-regulation of the ratio of Bax/Bcl-2 in bone marrow cells exposed to radiation.


