ConocarpanCAS# 221666-27-9 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 221666-27-9 | SDF | Download SDF |
| PubChem ID | 6474521 | Appearance | Powder |
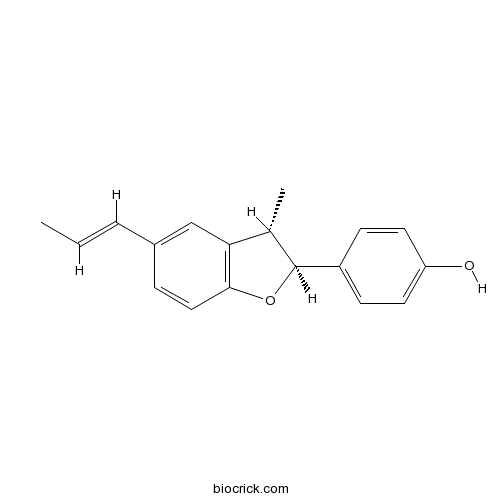
| Formula | C18H18O2 | M.Wt | 266.3 |
| Type of Compound | Lignans | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | 4-[(2S,3S)-3-methyl-5-[(E)-prop-1-enyl]-2,3-dihydro-1-benzofuran-2-yl]phenol | ||
| SMILES | CC=CC1=CC2=C(C=C1)OC(C2C)C3=CC=C(C=C3)O | ||
| Standard InChIKey | GXJSAHXNLJFDPO-OFXNJDNMSA-N | ||
| Standard InChI | InChI=1S/C18H18O2/c1-3-4-13-5-10-17-16(11-13)12(2)18(20-17)14-6-8-15(19)9-7-14/h3-12,18-19H,1-2H3/b4-3+/t12-,18-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. Conocarpan has antinociceptive effects. 2. Conocarpan is quite active against S. aureus and B. subtilis with MIC of 6.25 micrograms/ml, it also shows activity against M. tuberculosis (MIC=15.6 ug/ml). 3. Conocarpan shows considerable activity against epimastigote forms of T. cruzi, with 50% inhibition concentrations (IC50) of 8.0 microg/ml. |
| Targets | Antifection |

Conocarpan Dilution Calculator

Conocarpan Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.7552 mL | 18.7758 mL | 37.5516 mL | 75.1033 mL | 93.8791 mL |
| 5 mM | 0.751 mL | 3.7552 mL | 7.5103 mL | 15.0207 mL | 18.7758 mL |
| 10 mM | 0.3755 mL | 1.8776 mL | 3.7552 mL | 7.5103 mL | 9.3879 mL |
| 50 mM | 0.0751 mL | 0.3755 mL | 0.751 mL | 1.5021 mL | 1.8776 mL |
| 100 mM | 0.0376 mL | 0.1878 mL | 0.3755 mL | 0.751 mL | 0.9388 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- OBAA
Catalog No.:BCC6716
CAS No.:221632-26-4
- L-Menthol
Catalog No.:BCN4971
CAS No.:2216-51-5
- Boron tributoxide
Catalog No.:BCN8281
CAS No.:688-74-4
- 2,6-Dimethoxy-1-acetonylquinol
Catalog No.:BCN5052
CAS No.:2215-96-5
- 1-Benzyl-1H-indazol-3-ol
Catalog No.:BCC8458
CAS No.:2215-63-6
- Stigmastane-3,6-dione
Catalog No.:BCN5051
CAS No.:22149-69-5
- Hopane-3beta,22-diol
Catalog No.:BCN4852
CAS No.:22149-65-1
- Kaempferol 3-gentiobioside
Catalog No.:BCN3345
CAS No.:22149-35-5
- 11-Hydroxytabersonine
Catalog No.:BCN5050
CAS No.:22149-28-6
- 2 beta-Hydroxykolavelool
Catalog No.:BCN4671
CAS No.:221466-42-8
- 2-Oxokolavelool
Catalog No.:BCN4672
CAS No.:221466-41-7
- Fischeria A
Catalog No.:BCN3779
CAS No.:221456-63-9
- Methylswertianin
Catalog No.:BCN8505
CAS No.:22172-17-4
- Zotarolimus(ABT-578)
Catalog No.:BCC5481
CAS No.:221877-54-9
- Macrocarpal N
Catalog No.:BCN5811
CAS No.:221899-21-4
- L-Canavanine sulfate
Catalog No.:BCC6746
CAS No.:2219-31-0
- Lyn peptide inhibitor
Catalog No.:BCC5895
CAS No.:222018-18-0
- Pyrantel Pamoate
Catalog No.:BCC4958
CAS No.:22204-24-6
- Naproxen
Catalog No.:BCC9091
CAS No.:22204-53-1
- Dihydroseselin
Catalog No.:BCN8258
CAS No.:2221-66-1
- Dehydroglaucine
Catalog No.:BCN2548
CAS No.:22212-26-6
- Cucurbitacin I
Catalog No.:BCC2439
CAS No.:2222-07-3
- Thalrugosaminine
Catalog No.:BCN7745
CAS No.:22226-73-9
- Fern-7-en-19-one
Catalog No.:BCN6443
CAS No.:222294-61-3
Activity of neolignans isolated from Piper regnellii (MIQ.) C. DC. var. pallescens (C. DC.) YUNCK against Trypanosoma cruzi.[Pubmed:17015964]
Biol Pharm Bull. 2006 Oct;29(10):2126-30.
The in vitro antiproliferative effects of 4 neolignans purified from the ethyl-acetate extract from leaves of Piper regnellii (MIQ.) C. DC. var. pallescens (C. DC.) YUNCK against Trypanosoma cruzi were investigated. These isolated compounds were identified through spectral analyses of UV, EI-MS, 1H-, 13C-NMR, H-H COSY, gNOE, HETCOR, and HMBC. The compounds eupomatenoid-5, eupomatenoid-6, and Conocarpan showed considerable activity against epimastigote forms of T. cruzi, with 50% inhibition concentrations (IC50) of 7.0, 7.5, and 8.0 microg/ml respectively. After methylation, these compounds showed a lessened inhibitory activity to the growth of the protozoan, suggesting that loss of the hydroxyl group from their molecules reduces the activity. The compound eupomatenoid-3 showed lower activity than the hexane fraction. Eupomatenoid-5 was significantly more active than benznidazole, the antiparasitic drug of choice for treatment of Chagas' disease. The crude extract, hexane fraction, and eupomatenoid-5 caused no lysis in sheep blood at concentrations which inhibit the growth of epimastigote forms. The compound eupomatenoid-5 showed low cytotoxic effects against Vero cells. These results provide new perspectives on the development of novel drugs obtained from natural products with trypanocidal activity. However, the extracts and active compound isolated from P. regnellii var. pallescens should be further studied in animal models for in vivo efficacy.
Antinociceptive properties of conocarpan and orientin obtained from Piper solmsianum C. DC. var. solmsianum (Piperaceae).[Pubmed:20473574]
J Nat Med. 2010 Oct;64(4):402-8.
The antinociceptive properties of some fractions and two pure compounds, Conocarpan and orientin, obtained from P. solmsianum leaves were investigated in several models of pain in mice. The results indicated that this plant exhibits a promising antinociceptive profile, as it produces active principles which are several times more active than some reference drugs used for comparison. The main compound tested, orientin, caused potent and dose-dependent effects against acetic acid-induced writhing and capsaicin- and glutamate-induced nociception, being more effective against the first one, with an ID(50) value of 6.5 mg/kg (14.5 micromol/kg). Orientin was about 20-fold more potent than acetylsalicylic acid and 3.5-fold more active than indomethacin. The antinociceptive effects of this plant may be attributed, at least partially, to the presence of Conocarpan and, in particular, to the flavonoid orientin.
Anti-tuberculosis neolignans from Piper regnellii.[Pubmed:23474218]
Phytomedicine. 2013 May 15;20(7):600-4.
The present study determined the anti-Mycobacterium tuberculosis activities of supercritical CO2 extracts, neolignans eupomatenoid-5 (1), Conocarpan (4) and eupomatenoid-3 (7) and their derivatives (2, 3, 5, 6, and 8) from Piper regnellii, as well as their cytotoxicities. The supercritical CO2 extract from leaves was purified by chromatographic methods, yielding compounds (1), (4) and (7), which were identified by (1)H NMR and comparison with literature data. Anti-M. tuberculosis activity (H37Rv and clinical isolates) was evaluated using a resazurin microtiter assay plate (REMA) to determine the MIC. The cytotoxicity assay was carried out in macrophages J774G.8 by sulforhodamine B colorimetric assay. The supercritical CO2 extracts from leaves and stems, and compound (4) showed activity against M. tuberculosis (MIC 15.6 mug/ml). Compound (1) showed the best activity (MIC 1.9 mug/ml), with good SI. Compounds (7) and (8) showed low activity against M. tuberculosis H37Rv. The derivative compounds did not show increased anti-M. tuberculosis activity. This is the first report, to our knowledge, to describe neolignans from P. regnellii with activity against M. tuberculosis, and compound (1) is a potential candidate for future antituberculosis drugs.
Antibacterial activity of extracts and neolignans from Piper regnellii (Miq.) C. DC. var. pallescens (C. DC.) Yunck.[Pubmed:15049100]
Mem Inst Oswaldo Cruz. 2003 Dec;98(8):1115-20. Epub 2004 Mar 9.
The evaluation of the activity of the aqueous and ethyl acetate extracts of the leaves of Piper regnellii was tested against gram-positive and gram-negative bacteria. The aqueous extract displayed a weak activity against Staphylococcus aureus and Bacillus subtilis with minimal inhibitory concentration (MIC) and minimal bactericidal concentration (MBC) of 1000 micrograms/ml. The ethyl acetate extract presented a good activity against S. aureus and B. subtilis with MIC and MBC at 15.62 micrograms/ml. In contrast to the relative low MICs for gram-positive bacteria, gram-negative bacteria were not inhibited by the extracts at concentrations < or = 1000 mg/ml. The ethyl acetate extract was fractionated on silica gel into nine fractions. The hexane and chloroform fractions were active against S. aureus (MIC at 3.9 micrograms/ml) and B. subtilis (MIC at 3.9 and 7.8 micrograms/ml, respectively). Using bioactivity-directed fractionation, the hexane fraction was rechromatographed to yield the antimicrobial compounds 1, 2, 5, and 6 identified as eupomatenoid-6, eupomatenoid-5, eupomatenoid-3, and Conocarpan, respectively. The pure compounds 1 and 2 showed a good activity against S. aureus with MIC of 1.56 micrograms/ml and 3.12 micrograms/ml, respectively. Both compounds presented MIC of 3.12 micrograms/ml against B. subtilis. The pure compound 6 named as Conocarpan was quite active against S. aureus and B. subtilis with MIC of 6.25 micrograms/ml. The antibacterial properties of P. regnellii justify its use in traditional medicine for the treatment of wounds, contaminated through bacteria infections.


