D-XyloseCAS# 31178-70-8 |
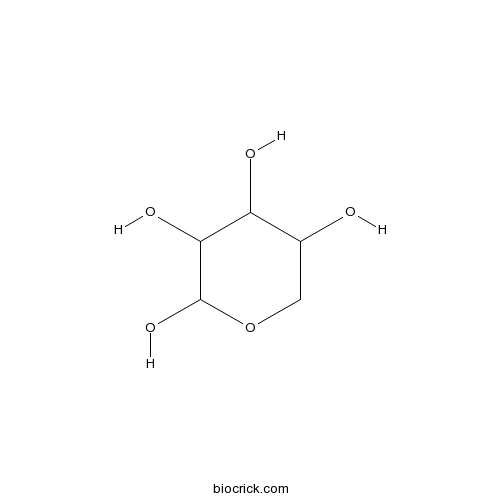
- Xylose
Catalog No.:BCC4880
CAS No.:25990-60-7
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 31178-70-8 | SDF | Download SDF |
| PubChem ID | 229 | Appearance | Powder |
| Formula | C5H10O5 | M.Wt | 150 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | oxane-2,3,4,5-tetrol | ||
| SMILES | C1C(C(C(C(O1)O)O)O)O | ||
| Standard InChIKey | SRBFZHDQGSBBOR-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C5H10O5/c6-2-1-10-5(9)4(8)3(2)7/h2-9H,1H2 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

D-Xylose Dilution Calculator

D-Xylose Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 6.6667 mL | 33.3333 mL | 66.6667 mL | 133.3333 mL | 166.6667 mL |
| 5 mM | 1.3333 mL | 6.6667 mL | 13.3333 mL | 26.6667 mL | 33.3333 mL |
| 10 mM | 0.6667 mL | 3.3333 mL | 6.6667 mL | 13.3333 mL | 16.6667 mL |
| 50 mM | 0.1333 mL | 0.6667 mL | 1.3333 mL | 2.6667 mL | 3.3333 mL |
| 100 mM | 0.0667 mL | 0.3333 mL | 0.6667 mL | 1.3333 mL | 1.6667 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- SYM 2081
Catalog No.:BCC6840
CAS No.:31137-74-3
- RFRP 3 (human)
Catalog No.:BCC6261
CAS No.:311309-27-0
- 3-Methyl-4-nitrobenzoic acid
Catalog No.:BCN2261
CAS No.:3113-71-1
- Taxifolin 3'-O-glucoside
Catalog No.:BCN6808
CAS No.:31106-05-5
- Benzoquinonium dibromide
Catalog No.:BCC6641
CAS No.:311-09-1
- 5,7,3'-Trihydroxy-6,4',5'-trimethoxyflavanone
Catalog No.:BCN1461
CAS No.:310888-07-4
- PRT 4165
Catalog No.:BCC6354
CAS No.:31083-55-3
- Decylic acid vanillylamide
Catalog No.:BCN7836
CAS No.:31078-36-1
- Alytesin
Catalog No.:BCC7203
CAS No.:31078-12-3
- Cedeodarin
Catalog No.:BCN4784
CAS No.:31076-39-8
- Spaglumic acid
Catalog No.:BCC6632
CAS No.:3106-85-2
- Echinophyllin C
Catalog No.:BCN5225
CAS No.:310433-44-4
- Sudan II
Catalog No.:BCN8383
CAS No.:3118-97-6
- Methylionene
Catalog No.:BCN7120
CAS No.:31197-54-3
- H-D-Ser-OH
Catalog No.:BCC2676
CAS No.:312-84-5
- Sideroxylin
Catalog No.:BCN5226
CAS No.:3122-87-0
- Eucalyptin
Catalog No.:BCN5227
CAS No.:3122-88-1
- Cimigenol-3-one
Catalog No.:BCN7430
CAS No.:31222-32-9
- IQ 3
Catalog No.:BCC8093
CAS No.:312538-03-7
- Tetrahydrocannabivarin
Catalog No.:BCN6935
CAS No.:31262-37-0
- SKI II
Catalog No.:BCC5029
CAS No.:312636-16-1
- THIQ
Catalog No.:BCC7539
CAS No.:312637-48-2
- gamma-Mangostin
Catalog No.:BCN5228
CAS No.:31271-07-5
- Indacaterol
Catalog No.:BCC1650
CAS No.:312753-06-3
In vitro reconstitution and characterisation of the oxidative D-xylose pathway for production of organic acids and alcohols.[Pubmed:30972503]
AMB Express. 2019 Apr 11;9(1):48.
The oxidative D-Xylose pathway, i.e. Dahms pathway, can be utilised to produce from cheap biomass raw material useful chemical intermediates. In vitro metabolic pathways offer a fast way to study the rate-limiting steps and find the most suitable enzymes for each reaction. We have constructed here in vitro multi-enzyme cascades leading from D-Xylose or D-xylonolactone to ethylene glycol, glycolic acid and lactic acid, and use simple spectrophotometric assays for the read-out of the efficiency of these pathways. Based on our earlier results, we focussed particularly on the less studied xylonolactone ring opening (hydrolysis) reaction. The bacterial Caulobacter crescentus lactonase (Cc XylC), was shown to be a metal-dependent enzyme clearly improving the formation of D-xylonic acid at pH range from 6 to 8. The following dehydration reaction by the ILVD/EDD family D-xylonate dehydratase is a rate-limiting step in the pathway, and an effort was made to screen for novel enolase family D-xylonate dehydratases, however, no suitable replacing enzymes were found for this reaction. Concerning the oxidation of glycolaldehyde to glycolic acid, several enzyme candidates were also tested. Both Escherichia coli aldehyde dehydrogenase (Ec AldA) and Azospirillum brasilense alpha-ketoglutarate semialdehyde dehydrogenase (Ab AraE) proved to be suitable enzymes for this reaction.
Hydrothermal Dehydration of Monosaccharides Promoted by Seawater: Fundamentals on the Catalytic Role of Inorganic Salts.[Pubmed:30968011]
Front Chem. 2019 Mar 22;7:132.
In biorefining, the conversion of carbohydrates under subcritical water conditions is a field of extensive studies. In particular, the hydrothermal decomposition of benchmark C6- and C5-monosaccharides, i.e., D-glucose and D-Xylose, into furanics and/or organic acids is fully considered. Herein, we propose to establish the fundamentals of the decomposition of D-glucose and D-Xylose under subcritical water conditions in the presence of specific salts (i.e., NaCl and KI) and in seawater. Our results demonstrated that the introduction of inorganic salts was found to modify sugars dehydration yields. Different NaCl concentrations from 0.21 to 1.63 mol L(-1) promoted the conversion of D-Xylose to 2-furfural (2-F) from 28 to 44% (molar yield). NaCl also improved 5-hydroxymethylfurfural (5-HMF) generation from D-glucose as well as rehydration of 5-HMF to levulinic and formic acid. KI favored other pathways toward formic acid production from D-glucose, reaching 20% in the upper concentration. Compared to a solution of equivalent NaCl concentration, seawater enhanced selectivity toward lactic acid which was raised by 10% for both monosaccharides, and sugars conversion, especially for D-glucose whose conversion was increased by 20%. 5-HMF molar yield around 30% were achieved from D-glucose in seawater at 211 degrees C and 20 bars after 15 min.


