THIQPotent and selective MC4 receptor agonist CAS# 312637-48-2 |
- Lomeguatrib
Catalog No.:BCC1133
CAS No.:192441-08-0
- 5-Azacytidine
Catalog No.:BCC1130
CAS No.:320-67-2
- Zebularine
Catalog No.:BCC1136
CAS No.:3690-10-6
- RG 108
Catalog No.:BCC1134
CAS No.:48208-26-0
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 312637-48-2 | SDF | Download SDF |
| PubChem ID | 9938402 | Appearance | Powder |
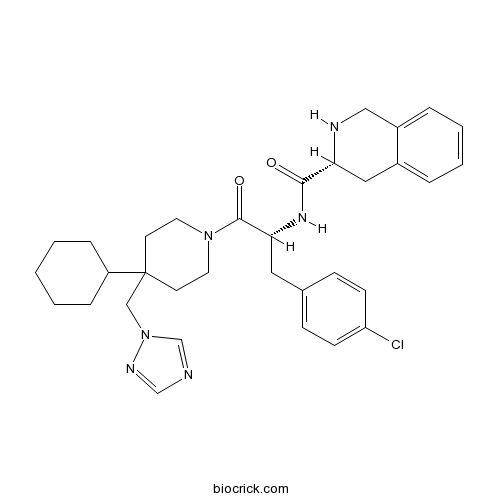
| Formula | C33H41ClN6O2 | M.Wt | 589.17 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | DMSO : 250 mg/mL (424.33 mM; Need ultrasonic) | ||
| Chemical Name | (3R)-N-[(2R)-3-(4-chlorophenyl)-1-[4-cyclohexyl-4-(1,2,4-triazol-1-ylmethyl)piperidin-1-yl]-1-oxopropan-2-yl]-1,2,3,4-tetrahydroisoquinoline-3-carboxamide | ||
| SMILES | C1CCC(CC1)C2(CCN(CC2)C(=O)C(CC3=CC=C(C=C3)Cl)NC(=O)C4CC5=CC=CC=C5CN4)CN6C=NC=N6 | ||
| Standard InChIKey | HLCHESOMJVGDSJ-LOYHVIPDSA-N | ||
| Standard InChI | InChI=1S/C33H41ClN6O2/c34-28-12-10-24(11-13-28)18-30(38-31(41)29-19-25-6-4-5-7-26(25)20-36-29)32(42)39-16-14-33(15-17-39,21-40-23-35-22-37-40)27-8-2-1-3-9-27/h4-7,10-13,22-23,27,29-30,36H,1-3,8-9,14-21H2,(H,38,41)/t29-,30-/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Potent and selective melanocortin 4 (MC4) receptor agonist (IC50 values are 1.2, 761 and 2067 nM for human MC4, MC3 and MC1 receptors respectively). Enhances intracavernosal pressure and stimulates erectile activity in rats ex copula following systemic administration. |

THIQ Dilution Calculator

THIQ Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.6973 mL | 8.4865 mL | 16.973 mL | 33.9461 mL | 42.4326 mL |
| 5 mM | 0.3395 mL | 1.6973 mL | 3.3946 mL | 6.7892 mL | 8.4865 mL |
| 10 mM | 0.1697 mL | 0.8487 mL | 1.6973 mL | 3.3946 mL | 4.2433 mL |
| 50 mM | 0.0339 mL | 0.1697 mL | 0.3395 mL | 0.6789 mL | 0.8487 mL |
| 100 mM | 0.017 mL | 0.0849 mL | 0.1697 mL | 0.3395 mL | 0.4243 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- SKI II
Catalog No.:BCC5029
CAS No.:312636-16-1
- Tetrahydrocannabivarin
Catalog No.:BCN6935
CAS No.:31262-37-0
- IQ 3
Catalog No.:BCC8093
CAS No.:312538-03-7
- Cimigenol-3-one
Catalog No.:BCN7430
CAS No.:31222-32-9
- Eucalyptin
Catalog No.:BCN5227
CAS No.:3122-88-1
- Sideroxylin
Catalog No.:BCN5226
CAS No.:3122-87-0
- H-D-Ser-OH
Catalog No.:BCC2676
CAS No.:312-84-5
- Methylionene
Catalog No.:BCN7120
CAS No.:31197-54-3
- Sudan II
Catalog No.:BCN8383
CAS No.:3118-97-6
- D-Xylose
Catalog No.:BCC8320
CAS No.:31178-70-8
- SYM 2081
Catalog No.:BCC6840
CAS No.:31137-74-3
- RFRP 3 (human)
Catalog No.:BCC6261
CAS No.:311309-27-0
- gamma-Mangostin
Catalog No.:BCN5228
CAS No.:31271-07-5
- Indacaterol
Catalog No.:BCC1650
CAS No.:312753-06-3
- Hygromycin B
Catalog No.:BCC1204
CAS No.:31282-04-9
- Raucaffricine
Catalog No.:BCN4653
CAS No.:31282-07-2
- TCS JNK 5a
Catalog No.:BCC5148
CAS No.:312917-14-9
- LDN-27219
Catalog No.:BCC6236
CAS No.:312946-37-5
- Arjunic acid
Catalog No.:BCN5229
CAS No.:31298-06-3
- Estradiol Cypionate
Catalog No.:BCC4477
CAS No.:313-06-4
- Aristolochic acid A
Catalog No.:BCN6262
CAS No.:313-67-7
- ICA 121431
Catalog No.:BCC6358
CAS No.:313254-51-2
- Regadenoson
Catalog No.:BCC6438
CAS No.:313348-27-5
- Reversan
Catalog No.:BCC7764
CAS No.:313397-13-6
Mild-condition synthesis of allenes from alkynes and aldehydes mediated by tetrahydroisoquinoline (THIQ).[Pubmed:24266693]
J Org Chem. 2013 Dec 6;78(23):11783-93.
A practical 1,2,3,4-tetrahydroisoquinoline (THIQ)-mediated synthesis of 1,3-disubstituted allenes from terminal alkynes and aldehydes under mild conditions in the presence of CuBr first and then ZnI2 was reported. This telescoped allene synthesis reaction includes three consecutive steps and two reactions: first, a room-temperature CuBr-catalyzed synthesis of propargylamines, exo-yne-THIQs, from terminal alkynes, aldehydes, and THIQ, then filtration of the CuBr catalyst, and finally the ZnI2-mediated allene synthesis from the generated exo-yne-THIQs under mild conditions (either at room temperature or heating at 50 or 75 degrees C). A wide range of aliphatic or aromatic aldehydes and terminal alkynes are tolerated, affording the allene products in up to 92% yield. Especially, temperature-sensitive aldehydes can be used in the reaction system. Preliminary exploration of the asymmetric allene synthesis has also been conducted, and a moderate enantioselectivity has been achieved. Finally, the relative reactivities of several secondary amines were compared with THIQ, showing that THIQ is the best of these amines in the synthesis of allenes under mild reaction conditions.
Structure-activity relationship (SAR) of the alpha-amino acid residue of potent tetrahydroisoquinoline (THIQ)-derived LFA-1/ICAM-1 antagonists.[Pubmed:21109434]
Bioorg Med Chem Lett. 2011 Jan 1;21(1):307-10.
This letter describes the structure-activity relationship (SAR) of the 'right-wing' alpha-amino acid residue of potent tetrahydroisoquinoline (THIQ)-derived LFA-1/ICAM-1 antagonists. Novel (S)-substituted heteroaryl-bearing alpha-amino acids have been identified as replacements of the 'right-wing' (S)-2,3-diaminopropanoic acid (DAP) moiety. Improvement of potency in the Hut-78 assay in the presence of 10% human serum has also been achieved.
Discovery of tetrahydroisoquinoline (THIQ) derivatives as potent and orally bioavailable LFA-1/ICAM-1 antagonists.[Pubmed:20655213]
Bioorg Med Chem Lett. 2010 Sep 1;20(17):5269-73.
This letter describes the discovery of a novel series of tetrahydroisoquinoline (THIQ)-derived small molecules that potently inhibit both human T-cell migration and super-antigen induced T-cell activation through disruption of the binding of integrin LFA-1 to its receptor, ICAM-1. In addition to excellent in vitro potency, 6q shows good pharmacokinetic properties and its ethyl ester (6t) demonstrates good oral bioavailability in both mouse and rat. Either intravenous administration of 6q or oral administration of its ethyl ester (6t) produced a significant reduction of neutrophil migration in a thioglycollate-induced murine peritonitis model.
A small molecule agonist THIQ as a novel pharmacoperone for intracellularly retained melanocortin-4 receptor mutants.[Pubmed:25076858]
Int J Biol Sci. 2014 Jul 20;10(8):817-24.
Although mutations in the melanocortin-4 receptor (MC4R) gene cause severe early-onset obesity, we still do not have effective approaches to correct the defects of these mutations. Several antagonists have been identified as pharmacoperones of the MC4R whereas no agonist of the MC4R has been reported. In the present study, we investigated the effect of a small molecule agonist of the MC4R, THIQ, on the cell surface expression and signaling of ten intracellularly retained MC4R mutants using different cell lines. We showed that THIQ increased the cell surface expression of three mutants (N62S, C84R, and C271Y) and two of them (N62S and C84R) had increased signaling in HEK293 cells. Interestingly, THIQ increased the signaling of two other mutants (P78L and P260Q) without increasing their cell surface expression in HEK293 cells. In neuronal cells, THIQ exhibited a more potent effect, correcting the cell surface expression and signaling of seven mutants (N62S, I69R, P78L, C84R, W174C, P260Q, and C271Y). Other mutants were not rescued by THIQ. We also showed that THIQ did not rescue MC4R mutants defective in ligand binding or signaling or one intracellularly retained mutant of the melanocortin-3 receptor. In summary, we demonstrated that a small molecule agonist acted as a pharmacoperone of the MC4R rescuing the cell surface expression and signaling of some intracellularly retained MC4R mutants.
New substituted piperazines as ligands for melanocortin receptors. Correlation to the X-ray structure of "THIQ".[Pubmed:15317471]
J Med Chem. 2004 Aug 26;47(18):4613-26.
A series of piperazine analogues of the melanocortin 4 receptor (MC4R) specific small-molecule agonist "THIQ" was synthesized and characterized structurally and pharmacologically. First, several THIQ imitations lacking the triazole moiety were prepared. Syntheses included acylation of 4-phenylpiperazine or 4-cyclohexylpiperazine. In two cases the tertiary amine function was replaced by the corresponding N-oxide. To obtain more complex structures, a 4-substituted piperazine ring was formed by alkylation of the primary amino group of cyclohexane-derived amino alcohols with N,N-bis(2-chloroethyl)benzylamine. The hydroxylic group of the intermediate was first activated with methanesulfonyl chloride, and the sulfonic ester formed in situ was introduced into the reaction with the sodium salt of 1,2,4-triazole. In one case (i.e., preparation of 23c) introduction of the 1,2,4-triazole moiety was performed at a carbon of the cyclohexane ring. In addition, this intermediate contained a piperazine moiety connected via its nitrogen atom to a cyclohexane ring carbon neighboring the reaction center. As established in NMR and X-ray investigations herein, this substitution proceeded with retention of the initial trans configuration of 1,2-disubstituted cyclohexane. To obtain pure enantiomers of 23c, its precursor 21c was subjected to chiral chromatography on a Chirobiotic V column. The derivatives (R,R)-21cand (S,S)-21c obtained were introduced into further syntheses steps, giving (R,R)-23c and (S,S)-23c, respectively. Melanocortin MC(1,3-5) receptor binding studies showed that all tested piperazine derivatives were active. Several compounds showed clear selectivity for MC4R, with submicromolar affinities being obtained. Among them, one substance, (R,R)-23c, displayed a biphasic curve in displacement of [125I]NDP-MSH on MC4R [K(i)high = 1 nM and K(i)low = 260 nM]. This biphasic competition curve was similarly biphasic to the competition curve obtained herein using THIQ. An X-ray study performed on crystals of the THIQ sulfate salt revealed two closely related conformations, which resemble the shape of the letter "Y", where piperidine and 4-chlorophenyl groups are situated close to each other, but the 1,2,3,4-tetrahydroisoquinoline residue is remote, the triazole function being highly exposed to the environment. The crystals of the dinitrate salt of (R,R)-23c showed a different conformation, where parts of the molecule are spread out almost symmetrically around the central section. Molecular modeling, based on the THIQ crystal structure and the functional similarity of THIQ and (R,R)-23c, allowed us to suggest a possible "bioactive" conformation of (R,R)-23c that is similar to the crystal conformation of THIQ.
Activation of melanocortin MC(4) receptors increases erectile activity in rats ex copula.[Pubmed:12409007]
Eur J Pharmacol. 2002 Nov 1;454(1):71-9.
Melanocortin peptide agonists, alpha-melanocyte stimulating hormone (alpha-MSH) and melanotan-II, stimulate erectile activity in a variety of species, including man. Since neither peptide discriminates amongst melanocortin receptors, it is not clear which subtype mediates these pro-erectile effects. Here, we present data that melanocortin-induced erectogenesis is mediated by melanocortin MC(4) receptors. Systemic administration of a melanocortin MC(4) receptor agonist (N-[(3R)-1,2,3,4-tetrahydroisoquinolinium-3-ylcarbonyl]-(1R)-1-(4-chlorobenzyl)-2 -[4-cyclohexyl-4-(1H-1,2,4-triazol-1ylmethyl)piperidin-1-yl]-2-oxoethylamine; THIQ) with high selectivity over other melanocortin receptors enhanced intracavernosal pressure and stimulated erectile activity in rats ex copula. THIQ dose-dependently (1-5 mg/kg, i.v.) increased the total number of erections, to an extent comparable or greater than that produced by apomorphine (0.025 mg/kg, s.c.). Central administration of THIQ (20 microg, intracerebroventricular (i.c.v.)) increased the number of reflexive penile erections; whereas administration of both a nonselective endogenous melanocortin MC(4) receptor antagonist (agouti-related protein (AgRP), 5.5. microg, i.c.v.) and a melanocortin MC(4) receptor preferring antagonist (MPB10, 1 mg/kg, i.v.) blocked THIQ-induced erectogenesis. These pro-erectile effects were also attenuated by systemic or central administration of an oxytocin antagonist (L-368899, 1 mg/kg, i.v.). Thus, melanocortin MC(4) receptor activation is sufficient for erectogenesis and these effects may involve oxytocinergic pathways.
Design and pharmacology of N-[(3R)-1,2,3,4-tetrahydroisoquinolinium- 3-ylcarbonyl]-(1R)-1-(4-chlorobenzyl)- 2-[4-cyclohexyl-4-(1H-1,2,4-triazol- 1-ylmethyl)piperidin-1-yl]-2-oxoethylamine (1), a potent, selective, melanocortin subtype-4 receptor agonist.[Pubmed:12361385]
J Med Chem. 2002 Oct 10;45(21):4589-93.
Synthetic and natural peptides that act as nonselective melanocortin receptor agonists have been found to be anorexigenic and to stimulate erectile activity. We report the design and development of 1, a potent, selective (1184-fold vs MC3R, 350-fold vs MC5R), small-molecule agonist of the MC4 receptor. Pharmacological testing confirms the food intake lowering effects of MC4R agonism and suggests another role for the receptor in the stimulation of erectile activity.


