DecursinolCAS# 23458-02-8 |
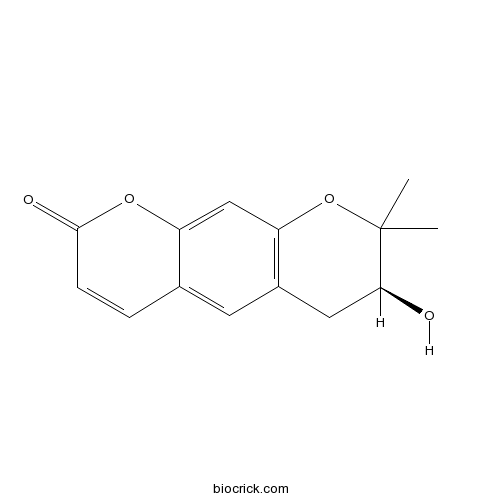
- 3',4'-dihydro-3'-hydroxy-Xanthyletin
Catalog No.:BCN3680
CAS No.:5993-18-0
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 23458-02-8 | SDF | Download SDF |
| PubChem ID | 442127 | Appearance | White powder |
| Formula | C14H14O4 | M.Wt | 246.26 |
| Type of Compound | Coumarins | Storage | Desiccate at -20°C |
| Synonyms | Smyrinol | ||
| Solubility | DMSO : 250 mg/mL (1015.19 mM; Need ultrasonic) | ||
| Chemical Name | (3S)-3-hydroxy-2,2-dimethyl-3,4-dihydropyrano[3,2-g]chromen-8-one | ||
| SMILES | CC1(C(CC2=C(O1)C=C3C(=C2)C=CC(=O)O3)O)C | ||
| Standard InChIKey | BGXFQDFSVDZUIW-LBPRGKRZSA-N | ||
| Standard InChI | InChI=1S/C14H14O4/c1-14(2)12(15)6-9-5-8-3-4-13(16)17-10(8)7-11(9)18-14/h3-5,7,12,15H,6H2,1-2H3/t12-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Decursinol may be a beneficial antimetastatic agent, targeting MMPs and its upstream signaling molecules; it inhibits the proliferation and invasion of CT-26 colon carcinoma cells, might via downregulated ERK and JNK phosphorylation. Aspirin-decursinol has neuroprotective effects, may be closely related to the attenuation of ischemia-induced gliosis and maintenance of antioxidants. |
| Targets | NADPH-oxidase | P450 (e.g. CYP17) | ERK | JNK | MMP(e.g.TIMP) |
| In vitro | In vitro metabolism of pyranocoumarin isomers decursin and decursinol angelate by liver microsomes from man and rodents.[Pubmed: 24026903]Planta Med. 2013 Nov;79(16):1536-44.The aim of this study is to investigate and compare the metabolic rate and profiles of pyranocoumarin isomers decursin and Decursinol angelate using liver microsomes from humans and rodents, and to characterize the major metabolites of decursin and Decursinol angelate in human liver microsomal incubations using LC-MS/MS. |
| In vivo | Single oral dose pharmacokinetics of decursin and decursinol angelate in healthy adult men and women.[Pubmed: 25695490]PLoS One. 2015 Feb 19;10(2):e0114992.The ethanol extract of Angelica gigas Nakai (AGN) root has promising anti-cancer and other bioactivities in rodent models. It is currently believed that the pyranocoumarin isomers decursin (D) and Decursinol angelate (DA) contribute to these activities. We and others have documented that D and DA were rapidly converted to Decursinol (DOH) in rodents. However, our in vitro metabolism studies suggested that D and DA might be metabolized differently in humans. |
| Kinase Assay | Decursin and decursinol from Angelica gigas inhibit the lung metastasis of murine colon carcinoma.[Pubmed: 21170925]Phytother Res. 2011 Jul;25(7):959-64.The principal objective of the present study was to evaluate the antimetastatic activity of decursin and Decursinol isolated from Angelica gigas. |

Decursinol Dilution Calculator

Decursinol Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 4.0607 mL | 20.3037 mL | 40.6075 mL | 81.215 mL | 101.5187 mL |
| 5 mM | 0.8121 mL | 4.0607 mL | 8.1215 mL | 16.243 mL | 20.3037 mL |
| 10 mM | 0.4061 mL | 2.0304 mL | 4.0607 mL | 8.1215 mL | 10.1519 mL |
| 50 mM | 0.0812 mL | 0.4061 mL | 0.8121 mL | 1.6243 mL | 2.0304 mL |
| 100 mM | 0.0406 mL | 0.203 mL | 0.4061 mL | 0.8121 mL | 1.0152 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- alpha-Spinasterone
Catalog No.:BCN5086
CAS No.:23455-44-9
- Trenbolone cyclohexylmethylcarbonate
Catalog No.:BCC9185
CAS No.:23454-33-3
- Alternariol monomethyl ether
Catalog No.:BCN7384
CAS No.:23452-05-3
- Physcion 1-glucoside
Catalog No.:BCN8170
CAS No.:23451-01-6
- Irisolidone
Catalog No.:BCN8496
CAS No.:2345-17-7
- Swertianolin
Catalog No.:BCN2759
CAS No.:23445-00-3
- Tetrahydropiperin
Catalog No.:BCN6708
CAS No.:23434-88-0
- 7-Isopentenyloxy-gamma-fagarine
Catalog No.:BCN5085
CAS No.:23417-92-7
- Phalaenopsine La
Catalog No.:BCN2015
CAS No.:23412-99-9
- Phalaenopsine T
Catalog No.:BCN2014
CAS No.:23412-97-7
- Theviridoside
Catalog No.:BCN5084
CAS No.:23407-76-3
- (1S,2R)-2-Amino-1,2-diphenylethanol
Catalog No.:BCC8385
CAS No.:23364-44-5
- trans-Khellactone
Catalog No.:BCN6920
CAS No.:23458-04-0
- 2-Palmitoylglycerol
Catalog No.:BCC7289
CAS No.:23470-00-0
- U 99194 maleate
Catalog No.:BCC7029
CAS No.:234757-41-6
- 2-Amino-5-mercapto-1,3,4-thiadiazole
Catalog No.:BCC8536
CAS No.:2349-67-9
- Hoechst 33258
Catalog No.:BCC1623
CAS No.:23491-45-4
- Hoechst 33342
Catalog No.:BCC1629
CAS No.:23491-52-3
- Hoechst 33258 analog 2
Catalog No.:BCC1625
CAS No.:23491-54-5
- Hoechst 33258 analog 5
Catalog No.:BCC1627
CAS No.:23491-55-6
- Peimine
Catalog No.:BCN1094
CAS No.:23496-41-5
- Axillaridine
Catalog No.:BCN2060
CAS No.:23506-96-9
- MEN 11270
Catalog No.:BCC6094
CAS No.:235082-52-7
- Humulon
Catalog No.:BCC8186
CAS No.:23510-81-8
In vitro metabolism of pyranocoumarin isomers decursin and decursinol angelate by liver microsomes from man and rodents.[Pubmed:24026903]
Planta Med. 2013 Nov;79(16):1536-44.
The aim of this study is to investigate and compare the metabolic rate and profiles of pyranocoumarin isomers decursin and Decursinol angelate using liver microsomes from humans and rodents, and to characterize the major metabolites of decursin and Decursinol angelate in human liver microsomal incubations using LC-MS/MS. First, we conducted liver microsomal incubations of decursin and Decursinol angelate in the presence or absence of NADPH. We found that in the absence of NADPH, decursin was efficiently hydrolyzed to Decursinol by hepatic esterase(s), but Decursinol angelate was not. In contrast, formation of Decursinol from Decursinol angelate was mediated mainly by cytochrome P450(s). Second, we measured the metabolic rate of decursin and Decursinol angelate in liver S9 fractions from mice and humans. We found that human liver S9 fractions metabolized both decursin and Decursinol angelate more slowly than those of the mouse. Third, we characterized the major metabolites of decursin and Decursinol angelate from human liver microsomes incubations using HPLC-UV and LC-MS/MS methods and assessed the in vivo metabolites in mouse plasma from a one-dose PK study. Decursin and Decursinol angelate have different metabolite profiles. Nine metabolites of decursin and nine metabolites of Decursinol angelate were identified in human liver microsome incubations besides Decursinol using a hybrid triple quadruple linear ion trap LC-MS/MS system, and many of them were later verified to be also present in plasma samples from rodent PK studies.
Single oral dose pharmacokinetics of decursin and decursinol angelate in healthy adult men and women.[Pubmed:25695490]
PLoS One. 2015 Feb 19;10(2):e0114992.
UNLABELLED: The ethanol extract of Angelica gigas Nakai (AGN) root has promising anti-cancer and other bioactivities in rodent models. It is currently believed that the pyranocoumarin isomers decursin (D) and Decursinol angelate (DA) contribute to these activities. We and others have documented that D and DA were rapidly converted to Decursinol (DOH) in rodents. However, our in vitro metabolism studies suggested that D and DA might be metabolized differently in humans. To test this hypothesis and address a key question for human translatability of animal model studies of D and DA or AGN extract, we conducted a single oral dose human pharmacokinetic study of D and DA delivered through an AGN-based dietary supplement Cogni.Q (purchased from Quality of Life Labs, Purchase, NY) in twenty healthy subjects, i.e., 10 men and 10 women, each consuming 119 mg D and 77 mg DA from 4 vegicaps. Analyses of plasma samples using UHPLC-MS/MS showed mean time to peak concentration (Tmax) of 2.1, 2.4 and 3.3 h and mean peak concentration (Cmax) of 5.3, 48.1 and 2,480 nmol/L for D, DA and DOH, respectively. The terminal elimination half-life (t1/2) for D and DA was similar (17.4 and 19.3 h) and each was much longer than that of DOH (7.4 h). The mean area under the curve (AUC0-48h) for D, DA and DOH was estimated as 37, 335 and 27,579 hnmol/L, respectively. Gender-wise, men absorbed the parent compounds faster and took shorter time to reach DOH peak concentration. The human data supported an extensive conversion of D and DA to DOH, even though they metabolized DA slightly slower than rodents. Therefore, the data generated in rodent models concerning anti-cancer efficacy, safety, tissue distribution and pharmacodynamic biomarkers will likely be relevant for human translation. TRIAL REGISTRATION: ClinicalTrials.gov NCT02114957.
Single oral dose pharmacokinetics of decursin, decursinol angelate, and decursinol in rats.[Pubmed:23364885]
Planta Med. 2013 Mar;79(3-4):275-80.
Decursin and Decursinol angelate are the major components in the alcoholic extract of the root of Angelica gigas Nakai. Our previous work convincingly demonstrated that both decursin and Decursinol angelate were rapidly converted to Decursinol in mice after administration by either oral gavage or i. p. injection. In the current study, we compared for the first time the plasma profiles of Decursinol, when equal moles of decursin/Decursinol angelate or Decursinol were given to rats by oral gavage, and investigated the effect of different formulas and other chemicals in Angelica gigas extract on the bioavailability of Decursinol. Our results show that gavage of Decursinol led to a faster attainment of plasma Decursinol peak (Tmax ~ 0.7 h) and much higher peak levels than an equal molar amount administered as decursin/Decursinol angelate mixture or as Angelica gigas ethanol extract, resulting in 2-3 fold higher bioavailability as estimated by the area under the curve of the respective regimens (65 012 vs. 27 033 h . ng/mL for Decursinol and decursin/Decursinol angelate treatment groups, respectively). Compared to a formula based on ethanol-PEG400-Tween80, carboxyl methyl cellulose was a less optimized vehicle. In addition, we detected peak levels of decursin and Decursinol angelate in the plasma of rats administered with decursin/Decursinol angelate or Angelica gigas extract in the nM range (Tmax ~ 0.5 h) with a newly established sensitive UHPLC-MS/MS method. Furthermore, our data support the liver, instead of intestine, as a major organ site where decursin and Decursinol angelate were hydrolyzed to Decursinol with a S9 microsomal in vitro metabolism assay. Taken together, our study provided important PK, LC-MS/MS methodology, formulation and metabolism insights in a rodent model for the rational design of in vivo efficacy studies of the corresponding chemicals in the future.
Decursin and decursinol from Angelica gigas inhibit the lung metastasis of murine colon carcinoma.[Pubmed:21170925]
Phytother Res. 2011 Jul;25(7):959-64.
The principal objective of the present study was to evaluate the antimetastatic activity of decursin and Decursinol isolated from Angelica gigas. Decursin and Decursinol inhibited the proliferation and invasion of CT-26 colon carcinoma cells. The expressions of matrix metalloproteinase (MMP)-2 and MMP-9 in cells and the activities in the culture medium were also reduced by decursin and Decursinol treatment. In CT-26 cells, the extracellular signal-regulated kinase (ERK) inhibitor inhibited cell proliferation, invasion and MMP-9 expression, and the c-Jun N-terminal kinase (JNK) inhibitor suppressed the expression of both MMPs, as well as cell proliferation and cell invasion. The phosphatidylinositol-3 kinase (PI3K) inhibitor reduced only the expression of MMP-2. In addition, the invasion of CT-26 cells was inhibited by the treatment with anti-MMP-9 antibody, rather than anti-MMP-2 antibody. These results indicate that MMP-9 expression via ERK and JNK plays a critical role for the invasion of CT26 cells. Decursin and Decursinol downregulated ERK and JNK phosphorylation. Moreover, oral administration of decursin and Decursinol reduced the formation of tumor nodules in the lungs and the increase in lung weight caused by CT-26 metastases. Therefore, both decursin and Decursinol may be beneficial antimetastatic agents, targeting MMPs and their upstream signaling molecules.


