DoxifluridineCAS# 3094-09-5 |
- Salmefamol
Catalog No.:BCC1919
CAS No.:18910-65-1
- Guanfacine hydrochloride
Catalog No.:BCC1609
CAS No.:29110-48-3
- (R,R)-Formoterol
Catalog No.:BCC1293
CAS No.:67346-49-0
- Medetomidine
Catalog No.:BCC1736
CAS No.:86347-14-0
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 3094-09-5 | SDF | Download SDF |
| PubChem ID | 18343 | Appearance | Powder |
| Formula | C9H11FN2O5 | M.Wt | 246.19 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | DMSO : ≥ 100 mg/mL (406.19 mM) DMF : 100 mg/mL (406.19 mM; Need ultrasonic) H2O : 20 mg/mL (81.24 mM; Need ultrasonic) *"≥" means soluble, but saturation unknown. | ||
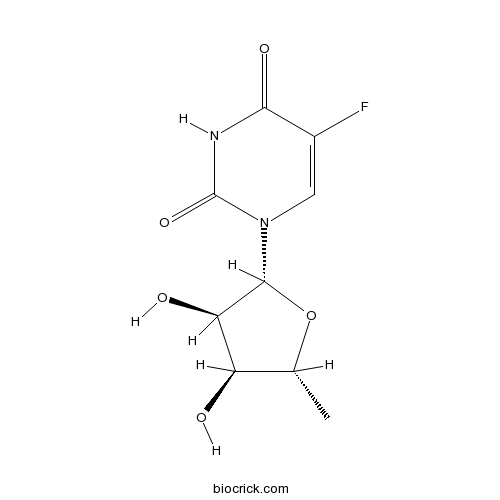
| Chemical Name | 1-[(2R,3R,4S,5R)-3,4-dihydroxy-5-methyloxolan-2-yl]-5-fluoropyrimidine-2,4-dione | ||
| SMILES | CC1C(C(C(O1)N2C=C(C(=O)NC2=O)F)O)O | ||
| Standard InChIKey | ZWAOHEXOSAUJHY-ZIYNGMLESA-N | ||
| Standard InChI | InChI=1S/C9H11FN2O5/c1-3-5(13)6(14)8(17-3)12-2-4(10)7(15)11-9(12)16/h2-3,5-6,8,13-14H,1H3,(H,11,15,16)/t3-,5-,6-,8-/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Doxifluridine Dilution Calculator

Doxifluridine Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 4.0619 mL | 20.3095 mL | 40.619 mL | 81.2381 mL | 101.5476 mL |
| 5 mM | 0.8124 mL | 4.0619 mL | 8.1238 mL | 16.2476 mL | 20.3095 mL |
| 10 mM | 0.4062 mL | 2.031 mL | 4.0619 mL | 8.1238 mL | 10.1548 mL |
| 50 mM | 0.0812 mL | 0.4062 mL | 0.8124 mL | 1.6248 mL | 2.031 mL |
| 100 mM | 0.0406 mL | 0.2031 mL | 0.4062 mL | 0.8124 mL | 1.0155 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Doxifluridine is a thymidine phosphorylase activator for PC9-DPE2 cells with IC50 of 0.62 μM.
- Inauhzin
Catalog No.:BCC5146
CAS No.:309271-94-1
- Boc-Asp-OBzl
Catalog No.:BCC3363
CAS No.:30925-18-9
- Boc-N-Me-Phg-OH
Catalog No.:BCC3350
CAS No.:30925-11-2
- 3β-Acetoxy-5α-androstan-17β-ol
Catalog No.:BCC8644
CAS No.:3090-70-8
- 8-Epixanthatin
Catalog No.:BCN7782
CAS No.:30890-35-8
- Kauran-18-Olc Acid,16,1719-Tnhydroxy-,(4A)
Catalog No.:BCC9235
CAS No.:308821-59-2
- Aloesin
Catalog No.:BCN8437
CAS No.:30861-27-9
- PM00104
Catalog No.:BCC4237
CAS No.:308359-57-1
- Spongouridine
Catalog No.:BCC9152
CAS No.:3083-77-0
- 4-Oxododecanedioic acid
Catalog No.:BCN5223
CAS No.:30828-09-2
- AEBSF.HCl
Catalog No.:BCC1219
CAS No.:30827-99-7
- L-Theanine
Catalog No.:BCN2571
CAS No.:3081-61-6
- Perillartine
Catalog No.:BCN8305
CAS No.:30950-27-7
- 2-Amino-9H-fluoren-9-one
Catalog No.:BCC8545
CAS No.:3096-57-9
- 1,5-Dicaffeoylquinic acid
Catalog No.:BCN5913
CAS No.:30964-13-7
- GW791343 HCl
Catalog No.:BCC4974
CAS No.:309712-55-8
- Vinyl Cinnamate
Catalog No.:BCN5041
CAS No.:3098-92-8
- Dauricinoline
Catalog No.:BCC8162
CAS No.:30984-80-6
- SX 011
Catalog No.:BCC7731
CAS No.:309913-42-6
- Boc-Aib-OH
Catalog No.:BCC3148
CAS No.:30992-29-1
- KL 001
Catalog No.:BCC6262
CAS No.:309928-48-1
- 3-(Boc-Amino)piperidine
Catalog No.:BCC8590
CAS No.:309956-78-3
- 7-O-Methylmangiferin
Catalog No.:BCN2804
CAS No.:31002-12-7
- 1-O-(3,4-Dimethoxybenzoyl)-beta-D-glucopyranose
Catalog No.:BCN3759
CAS No.:31002-27-4
Doxifluridine-conjugated 2-5A analog shows strong RNase L activation ability and tumor suppressive effect.[Pubmed:27364610]
Bioorg Med Chem. 2016 Aug 15;24(16):3870-4.
RNase L is activated by 2',5'-oligoadenylates (2-5A) at subnanomolar levels to cleave single-stranded RNA. We previously reported the hypothesis that the introduction of an 8-methyladenosine residue at the 2'-terminus of the 2-5A tetramer shifts the 2-5A binding site of RNase L. In this study, we synthesized various 5'-modified 2-5A analogs with 8-methyladenosine at the 2'-terminus. The Doxifluridine-conjugated 8-methyladenosine-substituted 2-5A analog was significantly more effective as an activator of RNase L than the parent 5'-monophophorylated 2-5A tetramer and showed a tumor suppressive effect against human cervical cancer cells.
Can thymidine phosphorylase be a predictive marker for gemcitabine and doxifluridine combination chemotherapy in cholangiocarcinoma?: case series.[Pubmed:25526478]
Medicine (Baltimore). 2014 Dec;93(28):e305.
Unresectable cholangiocarcinoma is poorly responded to chemotherapy, especially for the case refractory to gemcitabine and cisplatin. Here, we tested whether high expression of thymidine phosphorylase (TP) can be a predictive biomarker for the indicator for gemcitabine and Doxifluridine combination chemotherapy in the cholangiocarcinoma refractory to gemcitabine and cisplatin. Immunohistochemical staining for TP was performed with a biopsy specimen. We accepted the result as positive when more than 10% of cancer cells were stained with moderate intensity. Here, we report 2 cases of TP-positive cholangiocarcinoma well controlled with gemcitabine and Doxifluridine combination chemotherapy, which had been refractory to the first line treatment with gemcitabine and cisplatin combination chemotherapy.
Adjuvant chemotherapy for gastric cancer: a randomised phase 3 trial of mitomycin-C plus either short-term doxifluridine or long-term doxifluridine plus cisplatin after curative D2 gastrectomy (AMC0201).[Pubmed:23449357]
Br J Cancer. 2013 Apr 2;108(6):1245-51.
BACKGROUND: This phase 3 study evaluated the efficacy of new adjuvant chemotherapy (MFP), which intensified the mitomycin-C (MMC) plus short-term Doxifluridine (Mf) for gastric cancer. PATIENTS AND METHODS: A total of 855 patients (424 in Mf, 431 in MFP) with pathological stage II-IV (M0) gastric cancer after D2 gastrectomy were randomly assigned to receive either Mf (MMC 20 mg m(-2), followed by oral Doxifluridine 460-600 mg m(-2) per day for 3 months) or MFP (MMC 20 mg m(-2), followed by oral Doxifluridine 460-600 mg m(-2) per day for 12 months with 6 monthly infusions of 60 mg m(-2) of cisplatin) chemotherapy. RESULTS: With a median follow-up of 6.6 years, there was no difference between the two groups in recurrence-free survival (RFS) (5-year RFS 61.1% in Mf and 57.9% in MFP; hazard ratio 1.10 (95% CI 0.89-1.35); P=0.39) and overall survival (OS) (5-year OS 66.5% in Mf and 65.0% in MFP; hazard ratio 1.11 (95% CI 0.89-1.39); P=0.33). CONCLUSION: Intensification of Mf adjuvant chemotherapy by prolonging the duration of oral fluoropyrimidine and adding cisplatin was safe but not effective to improve the survivals in curatively resected gastric cancer patients.
Pharmacokinetic analysis of doxifluridine and its metabolites, 5-fluorouracil and 5-fluorouridine, after oral administration in beagle dogs.[Pubmed:23564503]
Eur J Drug Metab Pharmacokinet. 2013 Dec;38(4):295-9.
Doxifluridine (5'-deoxy-5-fluorouridine, 5'-dFUR) is a fluoropyrimidine derivative that is activated preferentially in malignant cells by thymidine phosphorylase to form 5-fluorouracil (5-FU). The purpose of this study was to investigate the pharmacokinetic properties of Doxifluridine and its two major metabolites, 5-FU, and 5-fluorouridine (5-FUrd), in beagle dogs following a single oral administration of 200 mg Doxifluridine capsule (Furtulon((R))). After the administration of 200 mg of Furtulon to 23 beagle dogs, the plasma concentrations of Doxifluridine, 5-FU, and 5-FUrd were measured simultaneously, using LC-MS/MS. The parent-metabolite compartment model with first-order absorption and Michaelis-Menten kinetics described the pharmacokinetics of Doxifluridine, 5-FU, and 5-FUrd. Michaelis-Menten kinetics sufficiently explained the generation and elimination processes of 5-FU and 5-FUrd. The studies described here are the first to evaluate the relationship between pharmacokinetics of Doxifluridine and its metabolites in dogs, and these findings will help in understanding the toxicity mechanism of Doxifluridine.


