FrangufolineCAS# 19526-09-1 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 19526-09-1 | SDF | Download SDF |
| PubChem ID | 5317387.0 | Appearance | Powder |
| Formula | C31H42N4O4 | M.Wt | 534.69 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
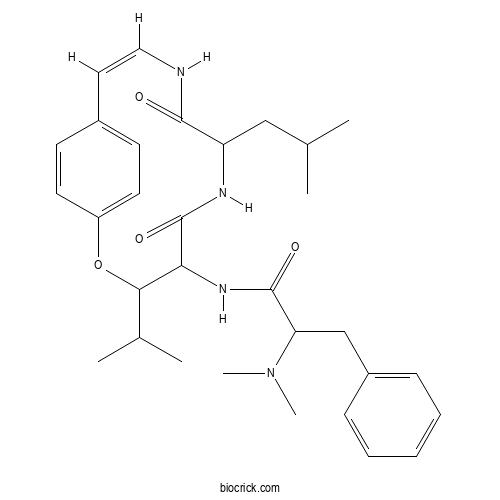
| Chemical Name | 2-(dimethylamino)-N-[(10Z)-7-(2-methylpropyl)-5,8-dioxo-3-propan-2-yl-2-oxa-6,9-diazabicyclo[10.2.2]hexadeca-1(14),10,12,15-tetraen-4-yl]-3-phenylpropanamide | ||
| SMILES | CC(C)CC1C(=O)NC=CC2=CC=C(C=C2)OC(C(C(=O)N1)NC(=O)C(CC3=CC=CC=C3)N(C)C)C(C)C | ||
| Standard InChIKey | TVUQUDJOLFMOKT-MSUUIHNZSA-N | ||
| Standard InChI | InChI=1S/C31H42N4O4/c1-20(2)18-25-29(36)32-17-16-22-12-14-24(15-13-22)39-28(21(3)4)27(31(38)33-25)34-30(37)26(35(5)6)19-23-10-8-7-9-11-23/h7-17,20-21,25-28H,18-19H2,1-6H3,(H,32,36)(H,33,38)(H,34,37)/b17-16- | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Frangufoline Dilution Calculator

Frangufoline Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.8702 mL | 9.3512 mL | 18.7024 mL | 37.4049 mL | 46.7561 mL |
| 5 mM | 0.374 mL | 1.8702 mL | 3.7405 mL | 7.481 mL | 9.3512 mL |
| 10 mM | 0.187 mL | 0.9351 mL | 1.8702 mL | 3.7405 mL | 4.6756 mL |
| 50 mM | 0.0374 mL | 0.187 mL | 0.374 mL | 0.7481 mL | 0.9351 mL |
| 100 mM | 0.0187 mL | 0.0935 mL | 0.187 mL | 0.374 mL | 0.4676 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Malonic acid
Catalog No.:BCX0780
CAS No.:90844-16-9
- Isogermafurenolide
Catalog No.:BCX0779
CAS No.:20267-89-4
- Nardoguaianone J
Catalog No.:BCX0778
CAS No.:443128-64-1
- Verrucosin
Catalog No.:BCX0777
CAS No.:83198-63-4
- Maltotriose
Catalog No.:BCX0776
CAS No.:1109-28-0
- 20-Deoxy,5-benzoyl-Ingenol
Catalog No.:BCX0775
CAS No.:54706-97-7
- 2,3-dihydroxypropyl 9-octadecenoate
Catalog No.:BCX0774
CAS No.:251983-54-7
- Nardoguaianone K
Catalog No.:BCX0773
CAS No.:443128-65-2
- 2-Hydroxycinnamicaldehyde
Catalog No.:BCX0772
CAS No.:3541-42-2
- (Z)-9-Nonadecene
Catalog No.:BCX0771
CAS No.:51865-02-2
- Hederoside D2
Catalog No.:BCX0770
CAS No.:20853-58-1
- Hirudonucleodisulfide B
Catalog No.:BCX0769
CAS No.:1072789-38-8
- Bixin
Catalog No.:BCX0782
CAS No.:6983-79-5
- 3-O-p-Coumaroylquinic acid
Catalog No.:BCX0783
CAS No.:5746-55-4
- Choerospondin
Catalog No.:BCX0784
CAS No.:81202-36-0
- Myricetin 3'-methyl-3-O-rutinoside
Catalog No.:BCX0785
CAS No.:55481-90-8
- Verbascose
Catalog No.:BCX0786
CAS No.:546-62-3
- Chrysotoxine
Catalog No.:BCX0787
CAS No.:156951-82-5
- 3-[[6-Deoxy-2-O-[6-O-[3-(4-hydroxyphenyl)-1-oxo-2-propen-1-yl]-β-D-glucopyranosyl]-α-L-mannopyranosyl]oxy]-2-(3,4-dihydroxyphenyl)-7-(β-D-glucopyranosyloxy)-5-hydroxy-4H-1-benzopyran-4-one
Catalog No.:BCX0788
CAS No.:1575672-58-0
- Yibeissine
Catalog No.:BCX0789
CAS No.:143502-51-6
- 6-hydroxyl kaempherol-3,6-O-diglucosyl-7-O-Glucuronic acid
Catalog No.:BCX0790
CAS No.:307950-53-4
- N-noratherosperminine
Catalog No.:BCX0791
CAS No.:74606-53-4
- Nor-rubrofusarin-6-O-β-D-gentiobioside
Catalog No.:BCX0792
CAS No.:245724-08-7
- 7,4′-Dihydroxyhomoisoflavane
Catalog No.:BCX0793
CAS No.:148462-00-4
Antibacterial activity of Discaria americana Gillies ex Hook (Rhamnaceae).[Pubmed:30543915]
J Ethnopharmacol. 2019 Jul 15;239:111635.
ETHNOPHARMACOLOGY RELEVANCE: Discaria americana Gillies ex Hook (sin. Discaria febrifuga and Discaria longispina) (Rhamnaceae) is a plant native from Rio Grande do Sul (Southern Brazil), Uruguay and Argentine, and has been used in Brazilian traditional medicine as antipyretic agent, and for stomach disorders. In Rio Grande do Sul, Uruguay and Argentine, the roots, in decoction, are used as tonic and febrifuge. Although it is a plant widely used by the population, there are no studies proving this popular use. MATERIAL AND METHODS: The crude neutral methanol extract, and pure isolated alkaloids, were investigated in vitro for antimicrobial activities against four Gram-positive bacteria: Staphylococcus aureus, Bacillus subtillis, Bacillus cereus, Enterococcus faecium; and five Gram-negative bacteria: Escherichia coli, Enterobacter cloacae, Enterobacter aerogenes, Salmonella enterica serovar Typhimurium and Pseudomonas aeruginosa. RESULTS: The crude neutral methanol (CME) extract of the root bark of Discaria americana showed antibacterial activity, ranging from 62.5 to 250 mug mL(-1) (MIC), against the tested bacteria. From the fractions obtained from the crude extract, the basic ethereal fraction (BEF) showed to be more effective, with MICs between 31.5 and 125 mug mL(-1) against the tested bacteria. The bioassay-guided fractionation of the ethyl ether basic fraction yielded eight cyclopeptide alkaloids: Frangufoline (1), frangulanine (2), adouetine Y' (3), discarine A (4) discarine B (5), discarine C (6), discarene C (7) and discarine D (8). When evaluated against the Gram-positive bacteria Enterococcus faecium, discarine B (5) proved to be the most active alkaloid with a MIC/MLC = 0.77/1.55 mug mL(-1), near the most active antibacterial agent levofloxacin (MIC/MLC = 0.77/0.77 mug mL(-1)). Moreover, discarine C (6) was the more active alkaloid against Salmonella enterica serovar Typhimurium, with a MIC/MLC = 3.1/6.2 mug mL(-1), the same observed for the antibacterial agent azithromycin. Kinetic measurements of the bacteriolytic activities of discarine B (5) against Enterococcus faecium (Gram-positive), and of discarine C (6) against Salmonella enterica serovar Typhimurium (Gram-negative) were determined by optical density based on real time assay, suggesting that both mode of action are partially bacteriolytic. CONCLUSION: In conclusion, five 14-membered cyclopeptide alkaloids isolated from Discaria americana Gillies ex Hook (Rhamnaceae) showed promising antibacterial activity, making this metabolites a class of scientific interest. The good activity presented by the extract and the alkaloids against the Gram-positive bacteria Enterococcus faecium and against the Gram-negative bacteria Salmonella enterica serovar Typhimurium, Enterobacter. aerogenes and Escherichia coli, corroborate with the popular use of this plant for stomach disorders and as antifebrile.
New bioactive cyclopeptide alkaloids with rare terminal unit from the root bark of Ziziphus cambodiana.[Pubmed:35541146]
RSC Adv. 2018 May 17;8(33):18204-18215.
Six new 14-membered ring cyclopeptide alkaloids, cambodines A-F (1-6), and two known compounds, Frangufoline (7) and lotusanine B (8), were isolated from the root bark extract of Ziziphus cambodiana Pierre. Their structures and configurations were established based on 1D and 2D NMR, HRMS, ECD, and X-ray crystallographic data. Compounds 1 and 3 are rare 5(14)-type cyclopeptide alkaloids that possess an imidazolidin-4-one ring in the terminal unit. The cyclopeptides were tested for their in vitro antiplasmodial, antitubercular, and cytotoxic effects against three cancer cell lines. Compound 3 showed significant antiplasmodial activity against the malarial parasite Plasmodium falciparum, with an IC(50) value of 6.09 muM.
[Chemical constituents from seeds of Ziziphus mauritiana].[Pubmed:25174108]
Zhong Yao Cai. 2014 Mar;37(3):432-5.
OBJECTIVE: To study the chemical constituents in the seeds of Ziziphus mauritiana. METHODS: The constituents were isolated by silica column chromatography and their structures were elucidated by physico-chemical properties and spectroscopic analysis. RESULTS: Twelve compounds were isolated from the seeds of Ziziphus mauritiana and identified as betulinic aldehyde (1), betulinic acid (2), ceanothic acid (3), Frangufoline (4), spinosin (5), beta-sitosterol (6), daucosterol (7), daucosterol-6'-octadecanoate (8), sucrose (9), docosanoic acid (10), stearic acid (11), palmitoleic acid (12). CONCLUSION: All the compounds are obtained from Ziziphus mauritiana seeds for the first time and compounds 4,5 and 8 are isolated from this plant for the first time.
Identification of marker compounds for Japanese Pharmacopoeia non-conforming jujube seeds from Myanmar.[Pubmed:25115227]
J Nat Med. 2015 Jan;69(1):68-75.
Jujube seed is a crude drug defined as the seed of Ziziphus jujuba Miller var. spinosa Hu ex H.F. Chou (Rhamnaceae) in the Japanese Pharmacopoeia (JP). Most of the jujube seed in the Japanese markets is imported from China, with the rest obtained from other Asian countries. Here we confirmed the botanical origins of jujube seeds from both China and Myanmar by a DNA sequencing analysis. We found that the botanical origins of the crude drugs from China and Myanmar were Z. jujuba and Z. mauritiana, respectively. Although the jujube seed from China conforms to the JP, that from Myanmar does not. A method for discriminating jujube seeds from China and Myanmar using a chemical approach is thus desirable, and here we sought to identify a compound specific to Z. jujuba. Jujuboside A (1) was identified as a compound specific to Z. jujuba. To establish a purity test of Jujube Seed in the JP against Z. mauritiana, we fractionated the extract of Z. mauritiana seeds and identified Frangufoline (2) and oleanolic acid (4) as the marker compounds specific to Z. mauritiana. Thin-layer chromatography (TLC) and gas chromatography-mass spectrometry analyses revealed that the latter compound was useful for testing by TLC analysis. The established TLC conditions were as follows: chromatographic support, silica gel; developing solvent, n-hexane:EtOAc:HCOOH = 10:5:1; developing length, 7 cm; visualization, diluted sulfuric acid; R f value, 0.43 (oleanolic acid).
Metabolic cleavage of frangufoline in rodents: in vitro and in vivo study.[Pubmed:9090868]
J Nat Prod. 1997 Mar;60(3):265-9.
Frangufoline, a sedative 14-membered frangulanine-type cyclopeptide alkaloid, was found to be rapidly converted, via enzymatic process, in vitro and in vivo in rodents to M1 ((S)-(N,N-dimethylphenylalanyl)-(2S,3S)-3-[(p-formylphenoxy) leucyl]-(S)-leucine); which is a substituted linear tripeptide. The reaction did not require low molecular weight cofactors, and mammalian serum failed to catalyze the reaction. Structure-reactivity study of cyclopeptide alkaloid analogs suggested that the enamide bond is the site being cleaved, and the reaction was inhibited by organophosphorus esters such as BPNP and by eserine at higher concentrations but not by eserine at lower concentrations or by EDTA and PCMB. On the basis of these results, a possible mechanism for metabolic conversion of Frangufoline to M1 was proposed, in which oxidation of the vinyl group and enzyme-catalyzed hydrolysis of the adjacent amide bond, possibly by B-esterase-like enzyme, proceed in a concerted manner.
[Peptide alkaloids from Discaria febrifuga Mart].[Pubmed:6662505]
Hoppe Seylers Z Physiol Chem. 1983 Nov;364(11):1641-3.
From the bark extract of Discaria febrifuga Mart. the known alkaloids Frangufoline (I), adouetine (II), scutianine B (III), franganine (IV) and two new ones, discarine C (V) and D (VI), were isolated. Their structures are elucidated by degradation reactions and spectroscopic investigations.


