GlyH-101CFTR Inhibitor II CAS# 328541-79-3 |
- Betamethasone
Catalog No.:BCC4765
CAS No.:378-44-9
- Beclomethasone dipropionate
Catalog No.:BCC4257
CAS No.:5534-09-8
- Methylprednisolone
Catalog No.:BCC2256
CAS No.:83-43-2
- Mifepristone
Catalog No.:BCC4486
CAS No.:84371-65-3
- Betamethasone hydrochloride
Catalog No.:BCC4256
CAS No.:956901-32-9
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 328541-79-3 | SDF | Download SDF |
| PubChem ID | 11260414 | Appearance | Powder |
| Formula | C19H15Br2N3O3 | M.Wt | 493.15 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | DMSO : ≥ 58 mg/mL (117.61 mM) *"≥" means soluble, but saturation unknown. | ||
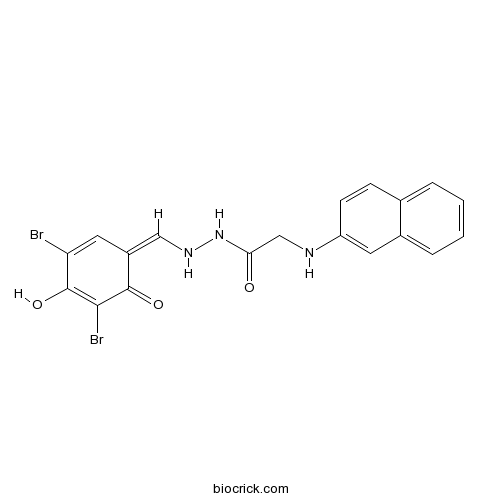
| Chemical Name | N'-[(Z)-(3,5-dibromo-4-hydroxy-6-oxocyclohexa-2,4-dien-1-ylidene)methyl]-2-(naphthalen-2-ylamino)acetohydrazide | ||
| SMILES | C1=CC=C2C=C(C=CC2=C1)NCC(=O)NNC=C3C=C(C(=C(C3=O)Br)O)Br | ||
| Standard InChIKey | FLGYCSPPJQJZCH-LCYFTJDESA-N | ||
| Standard InChI | InChI=1S/C19H15Br2N3O3/c20-15-8-13(18(26)17(21)19(15)27)9-23-24-16(25)10-22-14-6-5-11-3-1-2-4-12(11)7-14/h1-9,22-23,27H,10H2,(H,24,25)/b13-9- | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Reversible, voltage-dependent CFTR chloride channel blocker (Ki = 4.3 μM). Inhibits forskolin-induced hyperpolarization in nasal potential differences and inhibits cholera toxin-induced intestinal fluid secretion in mice. |

GlyH-101 Dilution Calculator

GlyH-101 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.0278 mL | 10.1389 mL | 20.2778 mL | 40.5556 mL | 50.6945 mL |
| 5 mM | 0.4056 mL | 2.0278 mL | 4.0556 mL | 8.1111 mL | 10.1389 mL |
| 10 mM | 0.2028 mL | 1.0139 mL | 2.0278 mL | 4.0556 mL | 5.0695 mL |
| 50 mM | 0.0406 mL | 0.2028 mL | 0.4056 mL | 0.8111 mL | 1.0139 mL |
| 100 mM | 0.0203 mL | 0.1014 mL | 0.2028 mL | 0.4056 mL | 0.5069 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
GlyH-101 is a cell-permeable glycinyl hydrazone compound that blocks CFTR with Ki of 1.4 uM.
- Lannaconitine
Catalog No.:BCN2504
CAS No.:32854-75-4
- (H-Cys-OMe)2.2HCl
Catalog No.:BCC2916
CAS No.:32854-09-4
- Coniferyl alcohol
Catalog No.:BCN4651
CAS No.:32811-40-8
- Phortress
Catalog No.:BCC3901
CAS No.:328087-38-3
- Ceranib 1
Catalog No.:BCC6186
CAS No.:328076-61-5
- H-D-Leu-OH
Catalog No.:BCC2975
CAS No.:328-38-1
- Nomifensine
Catalog No.:BCC7226
CAS No.:32795-47-4
- Panaxatriol
Catalog No.:BCN1081
CAS No.:32791-84-7
- Labetalol HCl
Catalog No.:BCC5489
CAS No.:32780-64-6
- Protopanaxatriol
Catalog No.:BCC9245
CAS No.:32773-56-1
- Macrocarpal O
Catalog No.:BCN7371
CAS No.:327622-65-1
- BIO 5192
Catalog No.:BCC8002
CAS No.:327613-57-0
- AG-14361
Catalog No.:BCC2209
CAS No.:328543-09-5
- Benzylamine hydrochloride
Catalog No.:BCN6908
CAS No.:3287-99-8
- Cajanin
Catalog No.:BCN5249
CAS No.:32884-36-9
- Lasiodiplodin
Catalog No.:BCN4770
CAS No.:32885-81-7
- De-O-methyllasiodiplodin
Catalog No.:BCN7187
CAS No.:32885-82-8
- Tildipirosin
Catalog No.:BCC5478
CAS No.:328898-40-4
- pep2-SVKI
Catalog No.:BCC5784
CAS No.:328944-75-8
- C646
Catalog No.:BCC4546
CAS No.:328968-36-1
- 4E1RCat
Catalog No.:BCC5338
CAS No.:328998-25-0
- Norepinephrine hydrochloride
Catalog No.:BCC5133
CAS No.:329-56-6
- DL-Adrenaline
Catalog No.:BCC4318
CAS No.:329-65-7
- PMSF
Catalog No.:BCC1229
CAS No.:329-98-6
Revisiting CFTR inhibition: a comparative study of CFTRinh -172 and GlyH-101 inhibitors.[Pubmed:24758416]
Br J Pharmacol. 2014 Aug;171(15):3716-27.
BACKGROUND AND PURPOSE: For decades, inhibitors of the cystic fibrosis transmembrane conductance regulator (CFTR) chloride channel have been used as tools to investigate the role and function of CFTR conductance in cystic fibrosis research. In the early 2000s, two new and potent inhibitors of CFTR, CFTRinh -172 and GlyH-101, were described and are now widely used to inhibit specifically CFTR. However, despite some evidence, the effects of both drugs on other types of Cl(-) -conductance have been overlooked. In this context, we explore the specificity and the cellular toxicity of both inhibitors in CFTR-expressing and non-CFTR-expressing cells. EXPERIMENTAL APPROACH: Using patch-clamp technique, we tested the effects of CFTRinh -172 and GlyH-101 inhibitors on three distinct types of Cl(-) currents: the CFTR-like conductance, the volume-sensitive outwardly rectifying Cl(-) conductance (VSORC) and finally the Ca(2+) -dependent Cl(-) conductance (CaCC). We also explored the effect of both inhibitors on cell viability using live/dead and cell proliferation assays in two different cell lines. KEY RESULTS: We confirmed that these two compounds were potent inhibitors of the CFTR-mediated Cl(-) conductance. However,GlyH-101 also inhibited the VSORC conductance and the CaCC at concentrations used to inhibit CFTR. The CFTRinh -172 did not affect the CaCC but did inhibit the VSORC, at concentrations higher than 5 microM. Neither inhibitor (20 microM; 24 h exposure) affected cell viability, but both were cytotoxic at higher concentrations. CONCLUSIONS AND IMPLICATIONS: Both inhibitors affected Cl(-) conductances apart from CFTR. Our results provided insights into their use in mouse models.
Divergent CFTR orthologs respond differently to the channel inhibitors CFTRinh-172, glibenclamide, and GlyH-101.[Pubmed:21940661]
Am J Physiol Cell Physiol. 2012 Jan 1;302(1):C67-76.
Comparison of diverse orthologs is a powerful tool to study the structure and function of channel proteins. We investigated the response of human, killifish, pig, and shark cystic fibrosis transmembrane conductance regulator (CFTR) to specific inhibitors of the channel: CFTR(inh)-172, glibenclamide, and GlyH-101. In three systems, including organ perfusion of the shark rectal gland, primary cultures of shark rectal gland tubules, and expression studies of each ortholog in cRNA microinjected Xenopus laevis oocytes, we observed fundamental differences in the sensitivity to inhibition by these channel blockers. In organ perfusion studies, shark CFTR was insensitive to inhibition by CFTR(inh)-172. This insensitivity was also seen in short-circuit current experiments with cultured rectal gland tubular epithelial cells (maximum inhibition 4 +/- 1.3%). In oocyte expression studies, shark CFTR was again insensitive to CFTR(inh)-172 (maximum inhibition 10.3 +/- 2.5% at 25 muM), pig CFTR was insensitive to glibenclamide (maximum inhibition 18.4 +/- 4.4% at 250 muM), and all orthologs were sensitive to GlyH-101. The amino acid residues considered responsible by previous site-directed mutagenesis for binding of the three inhibitors are conserved in the four CFTR isoforms studied. These experiments demonstrate a profound difference in the sensitivity of different orthologs of CFTR proteins to inhibition by CFTR blockers that cannot be explained by mutagenesis of single amino acids. We believe that the potency of the inhibitors CFTR(inh)-172, glibenclamide, and GlyH-101 on the CFTR chloride channel protein is likely dictated by the local environment and the three-dimensional structure of additional residues that form the vestibules, the chloride pore, and regulatory regions of the channel.
Locating a plausible binding site for an open-channel blocker, GlyH-101, in the pore of the cystic fibrosis transmembrane conductance regulator.[Pubmed:22923500]
Mol Pharmacol. 2012 Dec;82(6):1042-55.
High-throughput screening has led to the identification of small-molecule blockers of the cystic fibrosis transmembrane conductance regulator (CFTR) chloride channel, but the structural basis of blocker binding remains to be defined. We developed molecular models of the CFTR channel on the basis of homology to the bacterial transporter Sav1866, which could permit blocker binding to be analyzed in silico. The models accurately predicted the existence of a narrow region in the pore that is a likely candidate for the binding site of an open-channel pore blocker such as N-(2-naphthalenyl)-[(3,5-dibromo-2,4-dihydroxyphenyl)methylene]glycine hydrazide (GlyH-101), which is thought to act by entering the channel from the extracellular side. As a more-stringent test of predictions of the CFTR pore model, we applied induced-fit, virtual, ligand-docking techniques to identify potential binding sites for GlyH-101 within the CFTR pore. The highest-scoring docked position was near two pore-lining residues, Phe337 and Thr338, and the rates of reactions of anionic, thiol-directed reagents with cysteines substituted at these positions were slowed in the presence of the blocker, consistent with the predicted repulsive effect of the net negative charge on GlyH-101. When a bulky phenylalanine that forms part of the predicted binding pocket (Phe342) was replaced with alanine, the apparent affinity of the blocker was increased approximately 200-fold. A molecular mechanics-generalized Born/surface area analysis of GlyH-101 binding predicted that substitution of Phe342 with alanine would substantially increase blocker affinity, primarily because of decreased intramolecular strain within the blocker-protein complex. This study suggests that GlyH-101 blocks the CFTR channel by binding within the pore bottleneck.
Discovery of glycine hydrazide pore-occluding CFTR inhibitors: mechanism, structure-activity analysis, and in vivo efficacy.[Pubmed:15277574]
J Gen Physiol. 2004 Aug;124(2):125-37.
The cystic fibrosis transmembrane conductance regulator (CFTR) protein is a cAMP-regulated epithelial Cl- channel that, when defective, causes cystic fibrosis. Screening of a collection of 100,000 diverse small molecules revealed four novel chemical classes of CFTR inhibitors with Ki < 10 microM, one of which (glycine hydrazides) had many active structural analogues. Analysis of a series of synthesized glycine hydrazide analogues revealed maximal inhibitory potency for N-(2-naphthalenyl) and 3,5-dibromo-2,4-dihydroxyphenyl substituents. The compound N-(2-naphthalenyl)-[(3,5-dibromo-2,4-dihydroxyphenyl)methylene]glycine hydrazide (GlyH-101) reversibly inhibited CFTR Cl- conductance in <1 min. Whole-cell current measurements revealed voltage-dependent CFTR block by GlyH-101 with strong inward rectification, producing an increase in apparent inhibitory constant Ki from 1.4 microM at +60 mV to 5.6 microM at -60 mV. Apparent potency was reduced by lowering extracellular Cl- concentration. Patch-clamp experiments indicated fast channel closures within bursts of channel openings, reducing mean channel open time from 264 to 13 ms (-60 mV holding potential, 5 microM GlyH-101). GlyH-101 inhibitory potency was independent of pH from 6.5-8.0, where it exists predominantly as a monovalent anion with solubility approximately 1 mM in water. Topical GlyH-101 (10 microM) in mice rapidly and reversibly inhibited forskolin-induced hyperpolarization in nasal potential differences. In a closed-loop model of cholera, intraluminal GlyH-101 (2.5 microg) reduced by approximately 80% cholera toxin-induced intestinal fluid secretion. Compared with the thiazolidinone CFTR inhibitor CFTR(inh)-172, GlyH-101 has substantially greater water solubility and rapidity of action, and a novel inhibition mechanism involving occlusion near the external pore entrance. Glycine hydrazides may be useful as probes of CFTR pore structure, in creating animal models of CF, and as antidiarrheals in enterotoxic-mediated secretory diarrheas.


