ImbricatinCAS# 84504-71-2 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 84504-71-2 | SDF | Download SDF |
| PubChem ID | 14237636 | Appearance | Powder |
| Formula | C16H14O4 | M.Wt | 270.28 |
| Type of Compound | Phenols | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
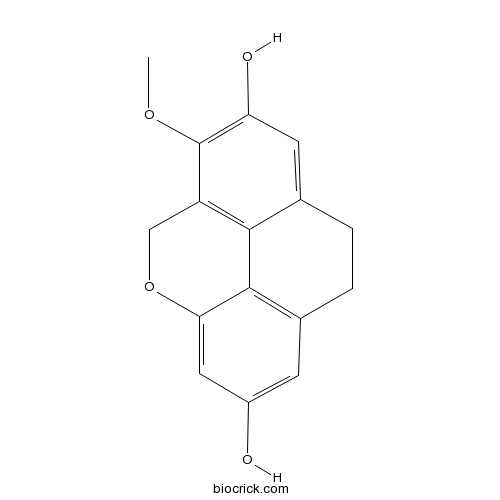
| Chemical Name | 5-methoxy-2-oxatetracyclo[6.6.2.04,16.011,15]hexadeca-1(14),4(16),5,7,11(15),12-hexaene-6,13-diol | ||
| SMILES | COC1=C(C=C2CCC3=C4C2=C1COC4=CC(=C3)O)O | ||
| Standard InChIKey | GNUBOQVVIAMPSG-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C16H14O4/c1-19-16-11-7-20-13-6-10(17)4-8-2-3-9(5-12(16)18)14(11)15(8)13/h4-6,17-18H,2-3,7H2,1H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Imbricatin Dilution Calculator

Imbricatin Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.6999 mL | 18.4993 mL | 36.9987 mL | 73.9973 mL | 92.4967 mL |
| 5 mM | 0.74 mL | 3.6999 mL | 7.3997 mL | 14.7995 mL | 18.4993 mL |
| 10 mM | 0.37 mL | 1.8499 mL | 3.6999 mL | 7.3997 mL | 9.2497 mL |
| 50 mM | 0.074 mL | 0.37 mL | 0.74 mL | 1.4799 mL | 1.8499 mL |
| 100 mM | 0.037 mL | 0.185 mL | 0.37 mL | 0.74 mL | 0.925 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Trigochinin B
Catalog No.:BCN9474
CAS No.:1210299-32-3
- Nauclefine
Catalog No.:BCN9473
CAS No.:57103-51-2
- (6E,12E)-Tetradeca-6,12-diene-8,10-diyne-1,3-diol diacetate
Catalog No.:BCN9472
CAS No.:89913-46-2
- (-)-Cedrusin
Catalog No.:BCN9471
CAS No.:404335-99-5
- Trigonothyrin D
Catalog No.:BCN9470
CAS No.:1254956-09-6
- Trigonosin D
Catalog No.:BCN9469
CAS No.:1262842-68-1
- Flavanthrinin
Catalog No.:BCN9468
CAS No.:130827-45-1
- Oxoflaccidin
Catalog No.:BCN9467
CAS No.:121817-24-1
- Lariciresinol p-coumarate
Catalog No.:BCN9466
CAS No.:864452-88-0
- Macedonic acid
Catalog No.:BCN9465
CAS No.:39022-00-9
- Shanciol H
Catalog No.:BCN9464
CAS No.:1114905-55-3
- 2',6'-Dimethoxypaulownin
Catalog No.:BCN9463
CAS No.:115196-22-0
- Kaempferol 3-O-(4''-O-trans-p-coumaroyl)rhamnopyranoside
Catalog No.:BCN9476
CAS No.:623927-14-0
- Helichrysoside
Catalog No.:BCN9477
CAS No.:56343-26-1
- Pipoxide chlorohydrin
Catalog No.:BCN9478
CAS No.:29228-15-7
- Eudesma-4(15),7(11)-dien-8-one
Catalog No.:BCN9479
CAS No.:54707-47-0
- Jacareubin
Catalog No.:BCN9480
CAS No.:3811-29-8
- 2-Hydroxy-1,8-dimethoxyxanthone
Catalog No.:BCN9481
CAS No.:38974-76-4
- 6-(3-Chloro-2-hydroxy-3-methylbutyl)-5,7-dimethoxycoumarin
Catalog No.:BCN9482
CAS No.:15575-50-5
- Leptostachyol acetate
Catalog No.:BCN9483
CAS No.:35770-58-2
- Rhodomyrtone
Catalog No.:BCN9484
CAS No.:468757-69-9
- Flaccidin
Catalog No.:BCN9485
CAS No.:115531-76-5
- Methoxyimbricatin
Catalog No.:BCN9486
CAS No.:233759-30-3
- 3-O-Debenzoylzeylenone
Catalog No.:BCN9487
CAS No.:1800008-77-8
Identification of phenanthrene derivatives in Aerides rosea (Orchidaceae) using the combined systems HPLC-ESI-HRMS/MS and HPLC-DAD-MS-SPE-UV-NMR.[Pubmed:25130411]
Phytochem Anal. 2015 Jan-Feb;26(1):34-9.
INTRODUCTION: In our continued efforts to contribute to the general knowledge on the chemical diversity of orchids, we have decided to focus our investigations on the Aeridinae subtribe. Following our previous phytochemical study of Vanda coerulea, which has led to the identification of phenanthrene derivatives, a closely related species, Aerides rosea Lodd. ex Lindl. & Paxton, was chosen for investigation. OBJECTIVE: To identify new secondary metabolites, and to avoid isolation of those already known, by means of the combined systems HPLC-DAD(diode-array detector) with high-resolution tandem mass spectrometry (HRMS/MS) and HPLC-DAD-MS-SPE(solid-phase extraction)-UV-NMR. METHODS: A dereplication strategy was developed using a HPLC-DAD-HRMS/MS targeted method and applied to fractions from A. rosea stem extract. Characterisation of unknown minor compounds was then performed using the combined HPLC-DAD-MS-SPE-UV-NMR system. RESULTS: The dereplication method allowed the characterisation of four compounds (gigantol, Imbricatin, methoxycoelonin and coelonin), previously isolated from Vanda coerulea stem extract. The analyses of two fractions permitted the identification of five additional minor constituents including one phenanthropyran, two phenanthrene and two dihydrophenanthrene derivatives. The full set of NMR data of each compound was obtained from microgram quantities. CONCLUSION: Nine secondary metabolites were characterised in A. rosea stems, utilising HPLC systems combined with high-resolution analytical systems. Two of them are newly described phenanthrene derivatives: aerosanthrene (5-methoxyphenanthrene-2,3,7-triol) and aerosin (3-methoxy-9,10-dihydro-2,5,7-phenanthrenetriol).
Inhibitory activities on nitric oxide production of stilbenoids from Pholidota yunnanensis.[Pubmed:24205813]
J Asian Nat Prod Res. 2013;15(12):1256-64.
Three new stilbenoids, 1-(4'-hydroxybenzyl)-Imbricatin, (E)-4'-hydroxy-2',3,3',5-tetramethoxystilbene, and (E)-3,4'-dihydroxy-2,6-bis(4-hydroxybenzyl)-2',3',5-trimethoxystilbene, together with 15 known stilbene derivatives, were isolated from Pholidota yunnanensis. Their structures were elucidated by spectroscopic methods and by comparison of their NMR data with those of related compounds. Furthermore, the inhibitory activities on nitric oxide (NO) production of the isolated compounds were examined in murine macrophages (RAW 264.7) activated by lipopolysaccharide. The cytotoxicity of 18 compounds was determined by the 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetr azolium assay. Among the tested compounds, eight stilbenoids, including three dihydrophenanthrenes, three stilbenes, and one bibenzyl derivative showed inhibitory effects on NO production without cytotoxicity with IC(5)(0) values ranging from 4.07 to 7.77 muM, as compared to MG-132, which was used as a positive control (IC(5)(0) of 0.10 muM). One dihydrophenanthrene, phoyunnanin C, showed cytotoxic effects at the test concentrations.
[Action of an extract of Vanda coerulea on the senescence of skin fibroblasts].[Pubmed:21570542]
Ann Pharm Fr. 2011 May;69(3):177-81.
The family of the orchids to date is poorly studied as a potential source of molecules with biological activity. The phytochemical analysis of extracts of Vanda coerulea stems (Orchidaceae), the isolation and the purification of the secondary metabolites realized by CLHP followed with high-resolution mass spectrometry and mono and two-dimensional nuclear magnetic resonance made it possible to identify the joint presence in an orchid of three stilbenoids i.e, Imbricatin, methoxycoelonin and gigantol. By flow cytometry, it is shown that the replicative senescence of human normal skin fibroblasts involves a reduction in the number of cells in phase S. A proteins chips technology dedicated to cell cycle proteins makes it possible to correlate this decrease of the number of cells in phase S to a decrease in cyclin E and cyclin dependant kinase 2, cdk2. The treatment by an ethanolic extract of stems of Vanda coerulea titrated in the three stilbenoids restores the percentage at an equivalent rate to that of young cells and the rate of cyclin E and, cdk2, thus bringing a beginning of explanation of their mechanism. These activities let predict an interesting potential as active ingredients to fight against the visible signs of cutaneous ageing.
Antioxidant biomarkers from Vanda coerulea stems reduce irradiated HaCaT PGE-2 production as a result of COX-2 inhibition.[Pubmed:21060890]
PLoS One. 2010 Oct 28;5(10):e13713.
BACKGROUND: In our investigations towards the isolation of potentially biologically active constituents from Orchidaceae, we carried out phytochemical and biological analyses of Vanda species. A preliminary biological screening revealed that Vanda coerulea (Griff. ex. Lindl) crude hydro-alcoholic stem extract displayed the best DPPH /(*)OH radical scavenging activity and in vitro inhibition of type 2 prostaglandin (PGE-2) release from UV(B) (60 mJ/cm(2)) irradiated HaCaT keratinocytes. PRINCIPAL FINDINGS: Bio-guided fractionation and phytochemical analysis led to the isolation of five stilbenoids: Imbricatin (1) methoxycoelonin (2) gigantol (3) flavidin (4) and coelonin (5). Stilbenoids (1-3) were the most concentrated in crude hydro-alcoholic stem extract and were considered as Vanda coerulea stem biomarkers. Dihydro-phenanthropyran (1) and dihydro-phenanthrene (2) displayed the best DPPH/(*)OH radical scavenging activities as well as HaCaT intracellular antioxidant properties (using DCFH-DA probe: IC(50) 8.8 microM and 9.4 microM, respectively) compared to bibenzyle (3) (IC(50) 20.6 microM). In turn, the latter showed a constant inhibition of PGE-2 production, stronger than stilbenoids (1) and (2) (IC(50) 12.2 microM and 19.3 microM, respectively). Western blot analysis revealed that stilbenoids (1-3) inhibited COX-2 expression at 23 microM. Interestingly, stilbenoids (1) and (2) but not (3) were able to inhibit human recombinant COX-2 activity. CONCLUSIONS: Major antioxidant stilbenoids (1-3) from Vanda coerulea stems displayed an inhibition of UV(B)-induced COX-2 expression. Imbricatin (1) and methoxycoelonin (2) were also able to inhibit COX-2 activity in a concentration-dependent manner thereby reducing PGE-2 production from irradiated HaCaT cells. Our studies suggest that stilbenoids (1-3) could be potentially used for skin protection against the damage caused by UV(B) exposure.
9, 10-Dihydrophenanthrene derivatives from Pholidota yunnanensis and scavenging activity on DPPH free radical.[Pubmed:17454314]
J Asian Nat Prod Res. 2007 Mar;9(2):165-74.
Three new 9,10-dihydrophenanthrene derivatives named phoyunnanins A-C, together with six known 9,10-dihydrophenanthrene constituents, 4,4',7,7'-tetrahydroxy-2,2'-dimethoxy-9,9',10,10'-tetrahydro-1,1'-biphenanthrene (4), lusianthridin (5), eulophiol (6), 2,4,7-trihydroxy-9,10-dihydrophenanthrene (7) and Imbricatin (8), were isolated from the 60% ethanolic extract of air-dried whole plant of Pholidota yunnanensis Rolfe. The structures of phoyunnanins A-C were established as 6-[2'-(3',3''-dihydroxy-5'-methoxybibenzy)]-4,7-dihydroxy-2-methoxy-9,10-dihydrop henanthrene (1), 6-[6'-(trans-3',3''-dihydroxy-5'-methoxystilbeny)]-4,7-dihydroxy-2-methoxy-9,10-d ihydrophenanthrene (2) and 4,4',7,7'-tetrahydroxy-2,2'-dimethoxy-9,9',10,10'-tetrahydro-1,6'-biphenanthrene (3), respectively, on the basis of the spectroscopic analysis. All eight compounds (1-8) were found to show the DPPH free radical scavenging activity with EC(50) from 8.8 to 55.9 microM.


