Norcepharadione BCAS# 57576-41-7 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 57576-41-7 | SDF | Download SDF |
| PubChem ID | 189168 | Appearance | Brown powder |
| Formula | C18H13NO4 | M.Wt | 307.3 |
| Type of Compound | Alkaloids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
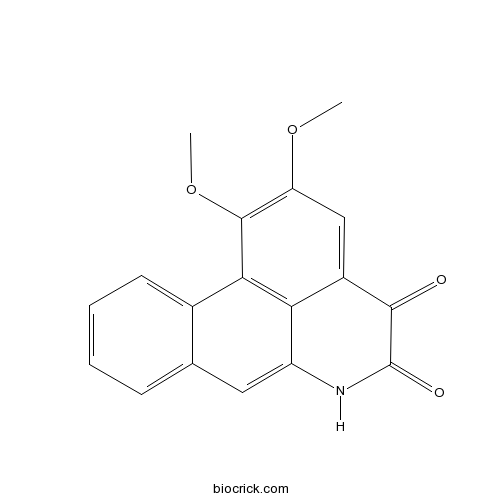
| SMILES | COC1=C(C2=C3C(=C1)C(=O)C(=O)NC3=CC4=CC=CC=C42)OC | ||
| Standard InChIKey | BAGGDUOPTSQTHD-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C18H13NO4/c1-22-13-8-11-14-12(19-18(21)16(11)20)7-9-5-3-4-6-10(9)15(14)17(13)23-2/h3-8H,1-2H3,(H,19,21) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Norcepharadione B shows good inhibitory activity against the replication of HSV-1, it also shows antimalarial activity with EC50 values of 7.5mug/ml. Norcepharadione B exhibits significant cytotoxicity against five human tumor cell lines (A-549, SK-OV-3, SK-MEL-2, XF-498 and HCT-15) in vitro. Norcepharadione B shows significant inhibitory effects on both ADP-induced and thrombin-induced platelet aggregation. |
| Targets | PAFR | HSV |
| In vitro | The constituents and their bioactivities of Houttuynia cordata.[Pubmed: 19881272]Chem Pharm Bull (Tokyo). 2009 Nov;57(11):1227-30.
Cytotoxic alkaloids from Houttuynia cordata.[Pubmed: 11794526]Arch Pharm Res. 2001 Dec;24(6):518-21.
Alkaloids from Houttuynia cordata and Their Antiplatelet Aggregation Activities.[Reference: WebLink]Chinese Journal of Natural Medicines, 2011, 9(6):425-8.To study the chemical constituents from Houttuynia cordata and to test their antiplatelet aggregation activities.Methods The chemical constituents were isolated and purified by silica gel column chromatography and their structures were elucidated on the basis of spectral analysis. The antiplatelet aggregation activities were evaluated by Born's method, using rat PRP induced by ADP and thrombin.ResultsNine alkaloids were isolated and their structures were identified as Norcepharadione B (1), 4, 5-dioxodehydroasimilobine (2), cepharadione B (3), aristololactam B II (4), aristololactam A II (5), sauristolactam (6), piperolactam A (7), splendidine (8), and aristololactam F II (9). Antiplatelet aggregation test indicated significant inhibitory activities of compounds 1, 2, 4–7, 9 induced by ADP and 1, 2, 4, 7 induced by thrombin.Conclusion Compounds 2, 6, 7, 9 were isolated from the genus Houttuynia for the first time. Compounds 1, 2, 4, 7 showed significant inhibitory effects on both ADP-induced and thrombin-induced platelet aggregation. |
| Structure Identification | Chulalongkorn University. 1996.Antimalarial compounds from Goniothalamus tenuifolius.[Reference: WebLink]
|

Norcepharadione B Dilution Calculator

Norcepharadione B Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.2541 mL | 16.2707 mL | 32.5415 mL | 65.083 mL | 81.3537 mL |
| 5 mM | 0.6508 mL | 3.2541 mL | 6.5083 mL | 13.0166 mL | 16.2707 mL |
| 10 mM | 0.3254 mL | 1.6271 mL | 3.2541 mL | 6.5083 mL | 8.1354 mL |
| 50 mM | 0.0651 mL | 0.3254 mL | 0.6508 mL | 1.3017 mL | 1.6271 mL |
| 100 mM | 0.0325 mL | 0.1627 mL | 0.3254 mL | 0.6508 mL | 0.8135 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- (2,4-Dihydroxyphenyl)acetonitrile
Catalog No.:BCN5783
CAS No.:57576-34-8
- (24S)-Cycloartane-3,24,25-triol 24,25-acetonide
Catalog No.:BCN1414
CAS No.:57576-31-5
- (3beta,24xi)-Cycloartane-3,24,25-triol
Catalog No.:BCN5782
CAS No.:57576-29-1
- PNU 37883 hydrochloride
Catalog No.:BCC7262
CAS No.:57568-80-6
- Isofuranodiene
Catalog No.:BCN5781
CAS No.:57566-47-9
- SNOG
Catalog No.:BCC6714
CAS No.:57564-91-7
- Notoginsenoside S
Catalog No.:BCN8371
CAS No.:575446-95-6
- H-His(Nτ-Me)-OMe.2HCl
Catalog No.:BCC2958
CAS No.:57519-09-2
- 2-Methylsulfanylpyrimidin-4(3H)-one
Catalog No.:BCC8582
CAS No.:5751-20-2
- 6-Benzyloxypurine
Catalog No.:BCC8770
CAS No.:57500-07-9
- 3-Benzalphthalide
Catalog No.:BCC8621
CAS No.:575-61-1
- 3,4'-Di-O-methylellagic acid
Catalog No.:BCN3710
CAS No.:57499-59-9
- Aclacinomycin A
Catalog No.:BCC1232
CAS No.:57576-44-0
- Cycloartane-3,24,25-triol
Catalog No.:BCC8922
CAS No.:57586-98-8
- Biocytin
Catalog No.:BCC7659
CAS No.:576-19-2
- Piperenone
Catalog No.:BCN6578
CAS No.:57625-31-7
- Fenobam
Catalog No.:BCC7345
CAS No.:57653-26-6
- Baccatin IV
Catalog No.:BCN5785
CAS No.:57672-77-2
- 1-Dehydroxybaccatin IV
Catalog No.:BCN7211
CAS No.:57672-78-3
- Baccatin VI
Catalog No.:BCN7229
CAS No.:57672-79-4
- Palmitic acid-1-13C
Catalog No.:BCC8229
CAS No.:57677-53-9
- Kansuinine B
Catalog No.:BCN3766
CAS No.:57685-46-8
- Flavanomarein
Catalog No.:BCN6429
CAS No.:577-38-8
- 2-Acetylbenzoic acid
Catalog No.:BCN5786
CAS No.:577-56-0
Cytotoxic alkaloids from Houttuynia cordata.[Pubmed:11794526]
Arch Pharm Res. 2001 Dec;24(6):518-21.
Six bioactive alkaloids, aristolactam B(1), piperolactam A(2), aristolactam A(3), Norcepharadione B(4), cepharadione B(5) and splendidine(6) were isolated by bioactivity-guided fractionation of a methanolic extract of the aerial part of Houttuynia cordata. Several of them exhibited significant cytotoxicity against five human tumor cell lines (A-549, SK-OV-3, SK-MEL-2, XF-498 and HCT-15) in vitro.
The constituents and their bioactivities of Houttuynia cordata.[Pubmed:19881272]
Chem Pharm Bull (Tokyo). 2009 Nov;57(11):1227-30.
Chemical investigation on the whole plant of Houttuynia cordata has resulted in the isolation of two new compounds, named as houttuynoside A (1) and houttuynamide A (2), together with thirty-eight known compounds. The structures of 1 and 2 were elucidated on the basis of spectroscopic analysis. In the inhibitory effects on herpes simplex virus type 1 (HSV-1) assay, Norcepharadione B (10) showed good inhibitory activity against the replication of HSV-1. In addition, the antioxidant and antityrosinase activities of some isolated compounds were also evaluated. Among these compounds, quercitrin (25) and quercetin-3-O-beta-D-galactopyranoside (26) showed excellent 2,2-diphenyl-1-picrylhydrazyl radical-scavenging property with IC50 values of 31 and 63 microM, respectively. Cepharadione B (9) exhibited strong tyrosinase inhibitory activity with an IC50 value of 170 microM.


