Orientanol ACAS# 190381-82-9 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 190381-82-9 | SDF | Download SDF |
| PubChem ID | 91885009 | Appearance | Powder |
| Formula | C21H24O7 | M.Wt | 388.4 |
| Type of Compound | Phenols | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
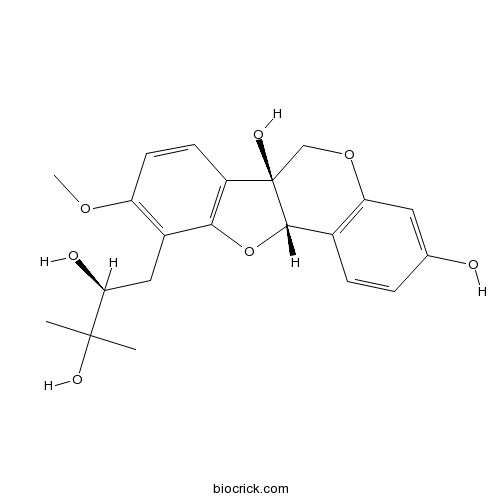
| Chemical Name | (6aS,11aS)-10-[(2S)-2,3-dihydroxy-3-methylbutyl]-9-methoxy-6,11a-dihydro-[1]benzofuro[3,2-c]chromene-3,6a-diol | ||
| SMILES | CC(C)(C(CC1=C(C=CC2=C1OC3C2(COC4=C3C=CC(=C4)O)O)OC)O)O | ||
| Standard InChIKey | TWRMBVPWOVPEPJ-HFSMHLIXSA-N | ||
| Standard InChI | InChI=1S/C21H24O7/c1-20(2,24)17(23)9-13-15(26-3)7-6-14-18(13)28-19-12-5-4-11(22)8-16(12)27-10-21(14,19)25/h4-8,17,19,22-25H,9-10H2,1-3H3/t17-,19-,21+/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Orientanol A is a natural product from Erythrina arborescens. |
| In vitro | Fuscacarpans A-C, new pterocarpans from the stems of Erythrina fusca.[Pubmed: 20079815 ]Fitoterapia. 2010 Sep;81(6):518-23.
|
| Structure Identification | J Asian Nat Prod Res. 2011 Feb;13(2):182-7.bis-Sigmodiol: a new prenylflavanone dimer from Erythrina sigmoidea Hua (Fabaceae) of Nigeria.[Pubmed: 21279883 ]
|

Orientanol A Dilution Calculator

Orientanol A Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.5747 mL | 12.8733 mL | 25.7467 mL | 51.4933 mL | 64.3666 mL |
| 5 mM | 0.5149 mL | 2.5747 mL | 5.1493 mL | 10.2987 mL | 12.8733 mL |
| 10 mM | 0.2575 mL | 1.2873 mL | 2.5747 mL | 5.1493 mL | 6.4367 mL |
| 50 mM | 0.0515 mL | 0.2575 mL | 0.5149 mL | 1.0299 mL | 1.2873 mL |
| 100 mM | 0.0257 mL | 0.1287 mL | 0.2575 mL | 0.5149 mL | 0.6437 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 3-Butyryloxytropane
Catalog No.:BCN1924
CAS No.:19038-34-7
- Sarracine N-oxide
Catalog No.:BCN2022
CAS No.:19038-27-8
- 7-Hydroxy-4-Methyl-8-Nitrocoumarin
Catalog No.:BCC9210
CAS No.:19037-69-5
- GR 135531
Catalog No.:BCC6836
CAS No.:190277-13-5
- Bripiodionene
Catalog No.:BCN1831
CAS No.:190265-70-4
- Spiratisanin C
Catalog No.:BCN6975
CAS No.:1902173-22-1
- Spiratisanin B
Catalog No.:BCN6976
CAS No.:1902173-19-6
- Spiratisanin A
Catalog No.:BCN6977
CAS No.:1902173-16-3
- Demethoxyencecalin
Catalog No.:BCN1175
CAS No.:19013-07-1
- Eupatoriochromene
Catalog No.:BCN1174
CAS No.:19013-03-7
- 7-Hydroxy-2,2-dimethylchromene
Catalog No.:BCN7784
CAS No.:19012-97-6
- N-Acetyl-O-phosphono-Tyr-Glu Dipentylamide
Catalog No.:BCC5855
CAS No.:190078-50-3
- SLIGKV-NH2
Catalog No.:BCC3959
CAS No.:190383-13-2
- 17β-Benzoyloxy-androsta-1,4-dien-3-one
Catalog No.:BCC8443
CAS No.:19041-66-8
- Beta-Amyloid (1-11)
Catalog No.:BCC1002
CAS No.:190436-05-6
- AT 1015
Catalog No.:BCC6194
CAS No.:190508-50-0
- Dioscin
Catalog No.:BCN6273
CAS No.:19057-60-4
- Prosapogenin A
Catalog No.:BCN2582
CAS No.:19057-67-1
- Ac-Gly-OEt
Catalog No.:BCC2944
CAS No.:1906-82-7
- Ro 32-3555
Catalog No.:BCC2377
CAS No.:190648-49-8
- Bepotastine Besilate
Catalog No.:BCC4538
CAS No.:190786-44-8
- Gracillin
Catalog No.:BCN5360
CAS No.:19083-00-2
- Isocupressic acid
Catalog No.:BCN1177
CAS No.:1909-91-7
- Triptocallic acid A
Catalog No.:BCN1176
CAS No.:190906-61-7
bis-Sigmodiol: a new prenylflavanone dimer from Erythrina sigmoidea Hua (Fabaceae) of Nigeria.[Pubmed:21279883]
J Asian Nat Prod Res. 2011 Feb;13(2):182-7.
A new prenylflavanone dimer named bis-sigmodiol was isolated from Erythrina sigmoidea, along with six known constituents isobavachin, lupiwighteone, Orientanol A, ergosta-4, 6, 8 (14), 22-tetraen-3-one, lupenyl acetate, and p-hydroxybenzoic acid. These known constituents have not been reported so far from E. sigmoidea. Their structures were elucidated by spectroscopic methods.
Fuscacarpans A-C, new pterocarpans from the stems of Erythrina fusca.[Pubmed:20079815]
Fitoterapia. 2010 Sep;81(6):518-23.
Chemical investigation of the stems of Erythrina fusca Lour. led to the isolation of three new pterocarpans, named fuscacarpans A-C (1-3), together with fourteen known compounds, sandwicensin (4), erythribyssin A (5), erythrabissin I (6), demethylmedicarpin (7), eryvarin D (8), erypoegin I (9), hydroxycristacarpone (10), Orientanol A (11), scandenone (12), genistein (13), liquiritigenin (14), isoliquiritigenin (15), vestitone (16) and 3,7,4'-trihydroxyflavone (17). Structures 1-3 were elucidated by spectroscopic and chemical methods. The isolates were evaluated for antibacterial, antiplasmodial and cytotoxic activities.


