S- (+)-RolipramCAMP-specific PDE4 inhibitor CAS# 85416-73-5 |
- GSK256066 2,2,2-trifluoroacetic acid
Catalog No.:BCC1605
CAS No.:1415560-64-3
- Nortadalafil
Catalog No.:BCC1806
CAS No.:171596-36-4
- Bay 60-7550
Catalog No.:BCC1405
CAS No.:439083-90-6
- Oglemilast
Catalog No.:BCC1817
CAS No.:778576-62-8
- AN-2728
Catalog No.:BCC1361
CAS No.:906673-24-3
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 85416-73-5 | SDF | Download SDF |
| PubChem ID | 158758 | Appearance | Powder |
| Formula | C16H21NO3 | M.Wt | 275.34 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | DMSO : 100 mg/mL (363.19 mM; Need ultrasonic) | ||
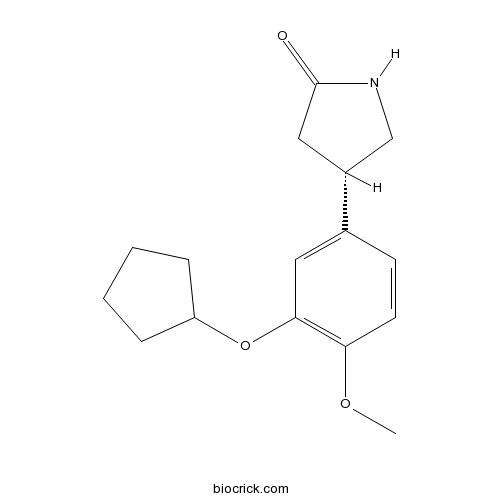
| Chemical Name | (4S)-4-(3-cyclopentyloxy-4-methoxyphenyl)pyrrolidin-2-one | ||
| SMILES | COC1=C(C=C(C=C1)C2CC(=O)NC2)OC3CCCC3 | ||
| Standard InChIKey | HJORMJIFDVBMOB-GFCCVEGCSA-N | ||
| Standard InChI | InChI=1S/C16H21NO3/c1-19-14-7-6-11(12-9-16(18)17-10-12)8-15(14)20-13-4-2-3-5-13/h6-8,12-13H,2-5,9-10H2,1H3,(H,17,18)/t12-/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Less active enantiomer of the PDE4 inhibitor rolipram. (R)-(-)-Rolipram also available. |

S- (+)-Rolipram Dilution Calculator

S- (+)-Rolipram Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.6319 mL | 18.1594 mL | 36.3187 mL | 72.6375 mL | 90.7968 mL |
| 5 mM | 0.7264 mL | 3.6319 mL | 7.2637 mL | 14.5275 mL | 18.1594 mL |
| 10 mM | 0.3632 mL | 1.8159 mL | 3.6319 mL | 7.2637 mL | 9.0797 mL |
| 50 mM | 0.0726 mL | 0.3632 mL | 0.7264 mL | 1.4527 mL | 1.8159 mL |
| 100 mM | 0.0363 mL | 0.1816 mL | 0.3632 mL | 0.7264 mL | 0.908 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
S-(+)-Rolipram inhibits human monocyte cyclic AMP-specific PDE4 with IC50 of 0.75 μM, has anti-inflammatory and anti-depressant activity in the central nervous system, less potent than its R enantiomer.
- Rilmenidine Phosphate
Catalog No.:BCC5637
CAS No.:85409-38-7
- Ropivacaine mesylate
Catalog No.:BCC9137
CAS No.:854056-07-8
- (-)-Haplomyrfolin
Catalog No.:BCN3225
CAS No.:85404-48-4
- Norcaesalpinin E
Catalog No.:BCN7006
CAS No.:854038-96-3
- Dasatinib hydrochloride
Catalog No.:BCC1517
CAS No.:854001-07-3
- OGT 2115
Catalog No.:BCC7458
CAS No.:853929-59-6
- NVP-BAG956
Catalog No.:BCC1813
CAS No.:853910-02-8
- (+)-AJ 76 hydrochloride
Catalog No.:BCC6747
CAS No.:85378-82-1
- SKF 89976A hydrochloride
Catalog No.:BCC6930
CAS No.:85375-15-1
- 5,19-Epoxy-19R,25-dimethoxycucurbita-6,23-dien-3-ol
Catalog No.:BCN1328
CAS No.:85372-72-1
- 5,19-Epoxy-19S,25-dimethoxycucurbita-6,23-dien-3-ol
Catalog No.:BCN1329
CAS No.:85372-70-9
- SecinH3
Catalog No.:BCC7503
CAS No.:853625-60-2
- (R)-(-)-Rolipram
Catalog No.:BCC5429
CAS No.:85416-75-7
- Caffeic anhydride
Catalog No.:BCN3295
CAS No.:854237-32-4
- Ajugamarin chlorohydrin
Catalog No.:BCN3664
CAS No.:85447-27-4
- 4-Chlorotestosterone acetate
Catalog No.:BCC8705
CAS No.:855-19-6
- Eupatorin
Catalog No.:BCN4405
CAS No.:855-96-9
- PK 11195
Catalog No.:BCC6745
CAS No.:85532-75-8
- Safflor Yellow A
Catalog No.:BCN2408
CAS No.:85532-77-0
- NBI-74330
Catalog No.:BCC4111
CAS No.:855527-92-3
- Rhodionin
Catalog No.:BCN1248
CAS No.:85571-15-9
- Kurarinol
Catalog No.:BCN3447
CAS No.:855746-98-4
- Zaltidine
Catalog No.:BCC2068
CAS No.:85604-00-8
- Gynuramine
Catalog No.:BCN2085
CAS No.:85611-43-4
Behaviour of [11C]R(-)- and [11C]S(+)-rolipram in vitro and in vivo, and their use as PET radiotracers for the quantificative assay of PDE4.[Pubmed:15668983]
Synapse. 2005 Mar 15;55(4):270-9.
Cyclic AMP (cAMP) is a continually produced nucleotide which is inactivated by hydrolysis to 5'AMP via phosphodiesterase 4 (PDE4) enzymes. Rolipram is a selective PDE4 inhibitor which exists in two enantiomeric forms, R(-) and S(+). Both of these enantiomers have previously been labelled with carbon-11 and used as positron emission tomography (PET) ligands for measuring PDE4 expression and function, and indirectly to explore the function of the cAMP second messenger, in vivo, using PET. The aim of these studies was to relate the in vitro affinities of the two rolipram enantiomers using standard pharmacological assays with the in vivo behaviour of the two enantiomers using PET. In vitro competition assays were performed using rat cortical membranes and [(3)H]R(-)- and [(3)H]S(+)-rolipram with increasing concentrations of either unlabelled R(-)- or S(+)-rolipram. In vivo, a series of PET studies were performed in the porcine brain using [(11)C]R(-)-rolipram with co-administration of increasing doses of either unlabelled R(-)- or S(+)-rolipram. Additional in vivo PET studies were performed using [(11)C]S(+)-rolipram with saturating doses of rolipram. In all studies, R(-)-rolipram exhibited a higher affinity for the PDE4 enzyme than S(+)-rolipram. The calculated affinity ratios were 7.97 from the in vitro studies; 12.5 from the in vivo studies using [(11)C]R(-)-rolipram; and 14.7 from the in vivo studies using [(11)C]S(+)-rolipram. To conclude, the in vitro affinities of R(-)- and S(+)-rolipram predict their apparent in vivo behaviour in the porcine brain, as measured by PET.
Multistep continuous-flow synthesis of (R)- and (S)-rolipram using heterogeneous catalysts.[Pubmed:25877201]
Nature. 2015 Apr 16;520(7547):329-32.
Chemical manufacturing is conducted using either batch systems or continuous-flow systems. Flow systems have several advantages over batch systems, particularly in terms of productivity, heat and mixing efficiency, safety, and reproducibility. However, for over half a century, pharmaceutical manufacturing has used batch systems because the synthesis of complex molecules such as drugs has been difficult to achieve with continuous-flow systems. Here we describe the continuous-flow synthesis of drugs using only columns packed with heterogeneous catalysts. Commercially available starting materials were successively passed through four columns containing achiral and chiral heterogeneous catalysts to produce (R)-rolipram, an anti-inflammatory drug and one of the family of gamma-aminobutyric acid (GABA) derivatives. In addition, simply by replacing a column packed with a chiral heterogeneous catalyst with another column packed with the opposing enantiomer, we obtained antipole (S)-rolipram. Similarly, we also synthesized (R)-phenibut, another drug belonging to the GABA family. These flow systems are simple and stable with no leaching of metal catalysts. Our results demonstrate that multistep (eight steps in this case) chemical transformations for drug synthesis can proceed smoothly under flow conditions using only heterogeneous catalysts, without the isolation of any intermediates and without the separation of any catalysts, co-products, by-products, and excess reagents. We anticipate that such syntheses will be useful in pharmaceutical manufacturing.
Use of a column-switching high-performance liquid chromatography method to assess the presence of specific binding of (R)- and (S)-[(11)C]rolipram and their labeled metabolites to the phosphodiesterase-4 enzyme in rat plasma and tissues.[Pubmed:18482689]
Nucl Med Biol. 2008 May;35(4):515-21.
INTRODUCTION: To complement recent studies using the high-affinity (11)C-labeled phosphodiesterase-4 (PDE4) inhibitor (R)-rolipram and the less active enantiomer (S)-[(11)C]rolipram for in vivo quantification of PDE4 levels, we evaluated the presence of radiolabeled metabolites and their potential binding to PDE4 in the rat plasma, brain, heart, pancreas, skeletal muscle and brown adipose tissue. METHODS: A reverse-phase capture and analytical HPLC column-switch method was used to detect (R)-[(11)C]rolipram, (S)-[(11)C]rolipram and their radiolabeled metabolites in rat plasma and tissue extracts. The relative proportion of PDE4-specific binding of the radiotracers and their labeled metabolites was analyzed following co-injections with a saturating dose of unlabeled (R)-rolipram at 45 min post-tracer injection in tissue extracts. RESULTS: Radiolabeled metabolites were found in the plasma (72-75% of total radioactive signal), and in the heart, skeletal muscle, pancreas and brown adipose tissue (44-52%), but not in the brain. In comparison to polar labeled metabolites, the proportion of unchanged (R)-[(11)C]rolipram was reduced in PDE4-rich organs by co-injection of unlabeled (R)-rolipram. Conversely, no changes were obtained in brown adipose tissue, or with (S)-[(11)C]rolipram, suggesting that radiolabeled metabolites of (R)-[(11)C]rolipram display no specific binding to PDE4. CONCLUSIONS: Radiolabeled hydrophilic metabolites are unlikely to compete with (R)-[(11)C]rolipram for PDE4-specific retention. However, due to the high proportion of the radioactive metabolites in the total radioactive signal, any kinetic modeling calculations in the peripheral tissues will need to take into account the presence of labeled metabolites.
Analysis of (R)- and (S)-[(11)C]rolipram kinetics in canine myocardium for the evaluation of phosphodiesterase-4 with PET.[Pubmed:21424298]
Mol Imaging Biol. 2012 Apr;14(2):225-36.
PURPOSE: (R)-[(11)C]rolipram and (S)-[(11)C]rolipram have been proposed to investigate phosphodiesterase-4 and, indirectly, cAMP-mediated signaling with PET. This study assessed binding of these tracers to phosphodiesterase-4 in canine myocardium. PROCEDURES: Seven dogs underwent (R)-[(11)C]rolipram and (S)-[(11)C]rolipram dynamic PET imaging at baseline and with co-injection of saturating doses of (R)-rolipram. Dual-input compartment models were applied to estimate the volumes of distribution (V(T)). RESULTS: The model comprising one compartment for unmetabolized tracer and one compartment for labeled metabolites provided excellent fits to data acquired with (S)-[(11)C]rolipram at baseline and with both enantiomers during co-injection scans. Use of two compartments for unmetabolized (R)-[(11)C]rolipram at baseline was warranted according to Akaike and Schwarz criteria. V(T) estimates obtained with these models were robust (CV
Inhibitory effects of rolipram on partially purified phosphodiesterase 4 from rat brains.[Pubmed:9681571]
Jpn J Pharmacol. 1998 Jun;77(2):147-54.
Several previous studies have demonstrated that the phosphodiesterase 4 selective inhibitor rolipram affects cellular function at a much lower concentration than the reported Ki value for phosphodiesterase 4 inhibition. In this study, we examined the inhibitory effect of rolipram on rat brain phosphodiesterase 4 to determine the heterogeneity of the enzyme activity. Partial purification of various phosphodiesterases from the rat brain was performed by anion-exchange chromatography. The eluant was pooled into four fractions, two of which manifested cAMP-selective phosphodiesterase activity that was blocked by 10 microM of rolipram, indicating the presence of phosphodiesterase 4 in these fractions. The IC50 of rolipram (racemate) of these two fractions was 492 and 79 nM, respectively. The R-(-)-enantiomer of rolipram inhibited the cAMP-phosphodiesterase activity in the latter fraction 10 times more than did S-(+)-rolipram, and the inhibition of the former fraction was less stereospecific. Dixon plot analysis revealed that the rolipram enantiomers inhibited the cAMP-phosphodiesterase in the latter fraction in a multiphasic manner, with two Ki values, one at the micromolar level and the other at the sub-micromolar level, respectively, for both of the enantiomers. These results suggest that there is a heterogeneity for phosphodiesterase 4 in the rat brain, and some of the phosphodiesterase forms are sensitive to rolipram.
Human phosphodiesterase 4A: characterization of full-length and truncated enzymes expressed in COS cells.[Pubmed:9337850]
Biochem J. 1997 Aug 15;326 ( Pt 1):53-60.
The type 4 phosphodiesterase (PDE) family comprises four enzymes (4A, 4B, 4C and 4D) that are characterized by their specificity for cAMP and selective inhibition by the anti-depressant drug rolipram (4-[3-(cyclopentoxyl)-4-methoxyphenyl]2-pyrrolidone). In common with other PDEs, they consist of a central conserved domain associated with catalytic activity in addition to two N-terminal upstream conserved regions (UCR1 and UCR2) that are unique to the type 4 enzymes. We have isolated a 2 kb cDNA encoding a full-length type 4A PDE{HSPDE4A4B[Bolger, Michaeli, Martins, St.John, Steiner, Rodgers, Riggs, Wigler and Ferguson (1993) Mol. Cell. Biol. 13, 6558-6571]} from a human frontal cortex cDNA library. Northern blot analysis showed that the major PDE4A mRNA of 4.5 kb was widely distributed in different human tissues. The recombinant PDE4A expressed in COS cells had a molecular mass of approx. 117 kDa as revealed by SDS/PAGE/Western blotting with a PDE4A-specific antibody and was specific for cAMP with a Km of 4.8 microM. The enzyme activity was potently inhibited by R-rolipram (IC50 204 nM) and showed a 2.7-fold stereoselectivity over the S enantiomer. Analysis of the kinetics of inhibition indicated that R-rolipram did not behave as a simple competitive inhibitor. Dixon replots suggested that there was more than one mode of interaction consistent with the detection in the enzyme of a high-affinity binding site for R-rolipram with a Kd of 2.3 nM. Truncation of the PDE4A enzyme by deletion mutagenesis showed that neither of the UCRs was required for catalytic activity and identified an approx. 71 kDa core enzyme with a K(m) for cAMP of 3.3 microM. In contrast with the full-length PDE4A, R-rolipram behaved as a simple competitive inhibitor of this form of the enzyme with decreased potency (IC50 1022 nM) and no stereoselectivity. In addition, no high-affinity rolipram-binding site was detected in the truncated enzyme, indicating that this interaction involves sequences upstream of the catalytic domain of the enzyme.
Stereospecific binding of the antidepressant rolipram to brain protein structures.[Pubmed:3019721]
Eur J Pharmacol. 1986 Aug 7;127(1-2):105-15.
The characteristics for the binding of the selective cAMP phosphodiesterase inhibitor and antidepressant agent rolipram to brain and peripheral organs were investigated. (+/-)-[3H]Rolipram equilibrium binding and Scatchard analysis revealed saturable, reversible, stereospecific, Mg2+-dependent and heat-sensitive binding with an apparent Hill number of 1. Binding was detected both to membrane-bound and soluble sites, with dissociation constants Kd of 1.2 and 2.4 nM, respectively, and binding site concentrations (Bmax) of 19.3 and 23.6 pmol/g rat forebrain. The (-)-enantiomer of rolipram was ca. 20 times more effective than the (+)-enantiomer in displacing (+/-)-[3H]rolipram from membranes. Rolipram bound to brain tissue of all mammalian species tested including man, while tissue from bird and fish showed less binding. Organs other than brain exhibited only negligible binding. Only specific cAMP phosphodiesterase inhibitors (ICI 63.197, Ro 20-1724) were potent competitors, while rolipram itself was inactive in a variety of receptor binding assays of neuroactive ligands. The kinetics of (-)-[3H]rolipram binding to the particulate fraction revealed a complex association and dissociation behaviour. The nature of the rolipram binding protein(s) is not clear, but the low affinity binding site evident from binding kinetics may represent a rolipram-sensitive phosphodiesterase isoenzyme also common to some peripheral organs, while the high affinity binding site(s) may be related to PDE isoenzymes more confined to the central nervous system.


