(±)-Sigmoidin ACAS# 176046-04-1 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 176046-04-1 | SDF | Download SDF |
| PubChem ID | 4303225 | Appearance | Cryst. |
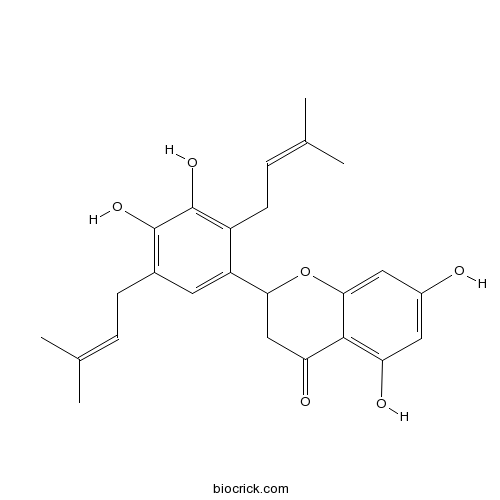
| Formula | C25H28O6 | M.Wt | 424.5 |
| Type of Compound | Flavonoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | 2-[3,4-dihydroxy-2,5-bis(3-methylbut-2-enyl)phenyl]-5,7-dihydroxy-2,3-dihydrochromen-4-one | ||
| SMILES | CC(=CCC1=C(C(=C(C(=C1)C2CC(=O)C3=C(C=C(C=C3O2)O)O)CC=C(C)C)O)O)C | ||
| Standard InChIKey | BVHLNRAYBCPKOY-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C25H28O6/c1-13(2)5-7-15-9-18(17(8-6-14(3)4)25(30)24(15)29)21-12-20(28)23-19(27)10-16(26)11-22(23)31-21/h5-6,9-11,21,26-27,29-30H,7-8,12H2,1-4H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Sigmoidin A has moderate antioxidant, antimicrobial and anti-inflammatory activity. |
| Targets | 5-lipoxygenase | Antifection |
| In vitro | Comparative Antioxidant, Prooxidant and Cytotoxic Activity of Sigmoidin A and Eriodictyol.[Reference: WebLink]Planta Medica, 2009, 76(6):589-594.Sigmoidin A (SGN) is a prenylated flavanone derivative of eriodictyol (ERD) with reported moderate antioxidant, antimicrobial and anti-inflammatory activity.
|
| Animal Research | Anti-Inflammatory Activities of Two Flavanones, Sigmoidin A and Sigmoidin B, from Erythrina sigmoidea[Reference: WebLink]Planta Medica, 2004, 70(02):104-107.Two prenylated flavanones isolated from Erythrina sigmoidea Hua (sigmoidin A and sigmoidin B) were studied for their ability to inhibit the stable 1,1-diphenyl-2-picrylhydrazyl (DPPH) free radical and arachidonic acid metabolism. In addition, the compounds were studied in two experimental models of inflammation induced in mouse ears by 12- O-tetradecanoylphorbol 13-acetate (TPA) and the phospholipase A (2)-induced mouse paw oedema. |

(±)-Sigmoidin A Dilution Calculator

(±)-Sigmoidin A Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.3557 mL | 11.7786 mL | 23.5571 mL | 47.1143 mL | 58.8928 mL |
| 5 mM | 0.4711 mL | 2.3557 mL | 4.7114 mL | 9.4229 mL | 11.7786 mL |
| 10 mM | 0.2356 mL | 1.1779 mL | 2.3557 mL | 4.7114 mL | 5.8893 mL |
| 50 mM | 0.0471 mL | 0.2356 mL | 0.4711 mL | 0.9423 mL | 1.1779 mL |
| 100 mM | 0.0236 mL | 0.1178 mL | 0.2356 mL | 0.4711 mL | 0.5889 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Valganciclovir HCl
Catalog No.:BCC4745
CAS No.:175865-59-5
- Wedelialactone A
Catalog No.:BCN6733
CAS No.:175862-40-5
- H-Dab.HBr
Catalog No.:BCC3184
CAS No.:1758-80-1
- PPAHV
Catalog No.:BCC7077
CAS No.:175796-50-6
- Lanatoside C
Catalog No.:BCN6457
CAS No.:17575-22-3
- Lanatoside B
Catalog No.:BCN6544
CAS No.:17575-21-2
- α-Conotoxin MII
Catalog No.:BCC5743
CAS No.:175735-93-0
- Tapentadol Hydrochloride
Catalog No.:BCC9159
CAS No.:175591-09-0
- Longifloroside A
Catalog No.:BCN1123
CAS No.:175556-08-8
- Voreloxin Hydrochloride
Catalog No.:BCC2045
CAS No.:175519-16-1
- Fmoc-Phe(4-Cl)-OH
Catalog No.:BCC3173
CAS No.:175453-08-4
- Fmoc-3-Pal-OH
Catalog No.:BCC2653
CAS No.:175453-07-3
- Fucosterol
Catalog No.:BCN6427
CAS No.:17605-67-3
- Zerumin A
Catalog No.:BCN3684
CAS No.:176050-48-9
- H-Val-OEt.HCl
Catalog No.:BCC3141
CAS No.:17609-47-1
- Maribavir
Catalog No.:BCC5259
CAS No.:176161-24-3
- Depressine
Catalog No.:BCN7851
CAS No.:176182-06-2
- LY 354740
Catalog No.:BCC7614
CAS No.:176199-48-7
- Nervogenic acid
Catalog No.:BCN1124
CAS No.:17622-86-5
- D-Raffinose Pentahydrate
Catalog No.:BCN2567
CAS No.:17629-30-0
- Dihydropinosylvin methyl ether
Catalog No.:BCN1125
CAS No.:17635-59-5
- Fmoc-D-Thr-ol
Catalog No.:BCC2575
CAS No.:176380-53-3
- 16alpha-Hydroxydehydrotrametenolic acid
Catalog No.:BCN1523
CAS No.:176390-66-2
- 16 alpha-Hydroxytrametenolic acid
Catalog No.:BCN2917
CAS No.:176390-68-4
The anti-obesity potential of sigmoidin A.[Pubmed:22978690]
Pharm Biol. 2012 Dec;50(12):1519-22.
CONTEXT: During the last few decades, the prevalence of obesity in the western world has dramatically increased with epidemic proportions. Hand in hand with this statistic, the incidences of obesity-linked diseases such as diabetes are increasing with pandemic rate. The search for novel drugs and nutritional intervention approaches for obesity is now of significant importance. OBJECTIVE: The anti-obesity potential of eriodictyol (ERD) and its close structural analogue, sigmoidin A (SGN), were evaluated. SGN was isolated from Erythrina abyssinica Lam. ex DC. (Fabaceae). MATERIALS AND METHODS: Concentrations between 300 and 0.1 microM of test samples and reference drugs made in three-fold dilutions were tested for enzyme inhibitory effects. The major obesity target, pancreatic lipase, was used to test the anti-obesity potential while the selective effects of the compounds were determined through assessments of effects on alpha-glucosidase. RESULTS: The inhibitory effect of SGN on pancreatic lipase (IC(5)(0), 4.5 +/- 0.87 microM) was 30-times greater than that of ERD (IC(5)(0), 134 +/- 19.39 microM) while their effect on alpha-glucosidase enzyme was comparable (IC(5)(0) value of 62.5 +/- 9.47 and 57.5 +/- 13.15 microM). The anti-obesity drug, orlistat, inhibited pancreatic lipase with an IC(5)(0) value of 0.3 +/- 0.04 microM, while the anti-diabetic drug, acarbose, inhibited alpha-glucosidase with an IC(5)(0) value of 190.6 +/- 16.05 microM. DISCUSSION: Although less active than the standard anti-obesity drug, orlistat, the observed activity indicated that prenylation of the flavonoid skeleton potently enhances anti-lipase activity. CONCLUSION: Such groups of flavonoids need to be further investigated for their therapeutic and nutritional benefit in combating obesity problems.
Sigmoidin F, a new prenylated flavonoid from Erythrina sigmoidea.[Pubmed:2614423]
J Nat Prod. 1989 Nov-Dec;52(6):1316-8.
Sigmoidin F, a new prenylated flavanone, as well as abyssinone IV and 5,7,4'-trihydroxy-3'-methoxy-5'-(1"-prenyl) flavonone have been isolated from the stem and bark of Erythrina sigmoidea. The structure of 1 has been confirmed by a combination of 1H-nmr and other spectroscopic techniques.
Activity of three cytotoxic isoflavonoids from Erythrina excelsa and Erythrina senegalensis (neobavaisoflavone, sigmoidin H and isoneorautenol) toward multi-factorial drug resistant cancer cells.[Pubmed:24252341]
Phytomedicine. 2014 Apr 15;21(5):682-8.
INTRODUCTION: Resistance of cancer cells to chemotherapy has become a worldwide concern. Naturally occuring isoflavonoids possess a variety of biological activities including anti-cancer effects. The present study was aimed at investigating the cytotoxicity and the modes of action of three naturally occuring isoflavonoids, neobavaisoflavone (1), sigmoidin H (2) and a pterocarpan that is a special type of isoflavonoid, isoneorautenol (3) against a panel of nine cancer cell lines, including various sensitive and drug-resistant phenotypes. METHODS: The cytotoxicity of the compounds was determined using a resazurin reduction assay, whereas the caspase-Glo assay was used to detect the activation of caspases 3/7, caspase 8 and caspase 9 in cells treated with compounds 3. Flow cytometry was used for cell cycle analysis and detection of apoptotic cells, analysis of mitochondrial membrane potential (MMP) as well as measurement of reactive oxygen species (ROS). RESULTS: Compounds 3 showed significant cytotoxicity toward sensitive and drug-resistant cancer cell lines. Compounds 1 and 2 were selectively active, and IC50 values below 115 muM were obtained on 6/9 and 4/9 cell lines respectively with values ranging from 42.93 muM (toward CCRF-CEM cells) to 114.64 muM [against HCT116 (p53(+/+)) cells] for 1 and 25.59 muM (toward U87MG) to 110.51 muM [against HCT116 (p53(+/+)) cells] for 2. IC50 values ranging from 2.67 muM (against MDA-MB 237BCRP cells) to 21.84 (toward U87MG) were measured for compound 3 and between 0.20 muM (toward CCRF-CEM cells) and 195.12 muM (toward CEM/ADR5000 cells) for doxorubicin as control drug. BCRP-transfected MDA-MB-231 cells, HCT116 (p53(+/+)) and U87MG.DeltaEGFR cells were hypersensitive (collateral sensitive) to compound 3 as compared to their counterpart cell lines. Compound 3 induced apoptosis in CCRF-CEM cells via activation of caspases 3/7, 8 and 9 as well as the loss of MMP and increased ROS production. CONCLUSIONS: The cytotoxicity of the studied isoflavonoids and especially the pterocarpan 3 deserve more detailed exploration in the future to develop novel anticancer drugs against sensitive and otherwise drug-resistant phenotypes.
Comparative antioxidant, prooxidant and cytotoxic activity of sigmoidin A and eriodictyol.[Pubmed:19941260]
Planta Med. 2010 Apr;76(6):589-94.
Sigmoidin A (SGN) is a prenylated flavanone derivative of eriodictyol (ERD) with reported moderate antioxidant, antimicrobial and anti-inflammatory activity. Since ERD and other structurally similar antioxidant phenolic compounds have been shown to induce prooxidative macromolecular damage and cytotoxicity in cancer cells, the comparative IN VITRO effects of these structural analogues on cancer cell viability and Cu(II)-dependent DNA damage were studied. In the presence of Cu(II) ions, both SGN and ERD (7.4-236 microM) caused comparable concentration-dependent pBR322 plasmid DNA strand scission. The DNA damage induced by SGN and ERD could be abolished by ROS scavengers, glutathione (GSH) and catalase as well as EDTA and a specific Cu(I) chelator neocuproine. Both ERD and SGN readily reduce Cu(II) to Cu(I) suggesting a prooxidative mechanism of DNA damage. In a cell free system, ERD and SGN did also show comparable radical scavenging activity. SGN was, however, by an order of magnitude more cytotoxic to cancer cells than ERD and this effect was significantly attenuated by GSH suggesting a prooxidative mechanism of cell death. A depletion of intracellular GSH level by SGN in cancer cells is also demonstrated.
Anti-inflammatory activities of two flavanones, sigmoidin A and sigmoidin B, from Erythrina sigmoidea.[Pubmed:14994185]
Planta Med. 2004 Feb;70(2):104-7.
Two prenylated flavanones isolated from Erythrina sigmoidea Hua (sigmoidin A and sigmoidin B) were studied for their ability to inhibit the stable 1,1-diphenyl-2-picrylhydrazyl (DPPH) free radical and arachidonic acid metabolism. In addition, the compounds were studied in two experimental models of inflammation induced in mouse ears by 12- O-tetradecanoylphorbol 13-acetate (TPA) and the phospholipase A (2)-induced mouse paw oedema. Both sigmoidins A and B proved to be potent scavengers of the DPPH radical, while the study of the inhibition of arachidonic acid metabolism demonstrated that these same compounds were selective inhibitors of 5-lipoxygenase, with no effect on cyclooxygenase-1 activity. Dose-response inhibitor potency was established for sigmoidin A (IC (50) = 31 microM). In the assay of phospholipase A (2)-induced mouse paw oedema, only the sigmoidin B derivative inhibited oedema formation at 60 min, showing a percentage of inhibition below that obtained with cyproheptadine (59 % vs. 74 %). In the TPA test, sigmoidins A and B decreased the induced oedema by 89 % and 83 %, respectively. This is the first time that the anti-inflammatory activity and antioxidant properties of these prenylflavanones have been reported. The results indicate that the compounds have different mechanisms of action depending on whether one or two prenyl groups are present in ring B.


