TembetarineCAS# 18446-73-6 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 18446-73-6 | SDF | Download SDF |
| PubChem ID | 167718 | Appearance | Powder |
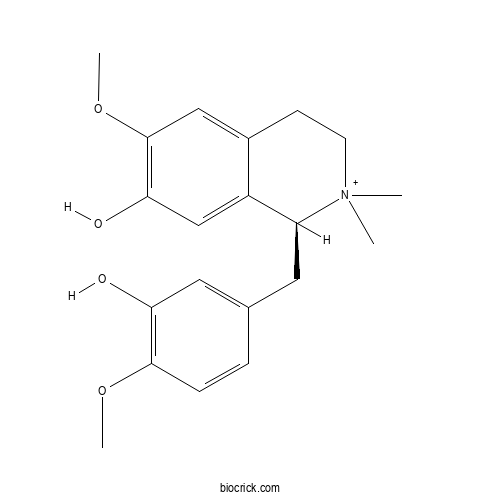
| Formula | C20H26NO4 | M.Wt | 344.4 |
| Type of Compound | Alkaloids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | (1S)-1-[(3-hydroxy-4-methoxyphenyl)methyl]-6-methoxy-2,2-dimethyl-3,4-dihydro-1H-isoquinolin-2-ium-7-ol | ||
| SMILES | C[N+]1(CCC2=CC(=C(C=C2C1CC3=CC(=C(C=C3)OC)O)O)OC)C | ||
| Standard InChIKey | ABSDACFLIMOXJY-INIZCTEOSA-O | ||
| Standard InChI | InChI=1S/C20H25NO4/c1-21(2)8-7-14-11-20(25-4)18(23)12-15(14)16(21)9-13-5-6-19(24-3)17(22)10-13/h5-6,10-12,16H,7-9H2,1-4H3,(H-,22,23)/p+1/t16-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Tembetarine Dilution Calculator

Tembetarine Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.9036 mL | 14.518 mL | 29.036 mL | 58.072 mL | 72.59 mL |
| 5 mM | 0.5807 mL | 2.9036 mL | 5.8072 mL | 11.6144 mL | 14.518 mL |
| 10 mM | 0.2904 mL | 1.4518 mL | 2.9036 mL | 5.8072 mL | 7.259 mL |
| 50 mM | 0.0581 mL | 0.2904 mL | 0.5807 mL | 1.1614 mL | 1.4518 mL |
| 100 mM | 0.029 mL | 0.1452 mL | 0.2904 mL | 0.5807 mL | 0.7259 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Apigenin-6-C-beta-D-glucopyranosyl-8-C-[alpha-L-rhamnopyranosyl-(1->2)]-beta-glucopyranoside
Catalog No.:BCX0033
CAS No.:1646598-06-2
- Ganolucidic acid E
Catalog No.:BCX0032
CAS No.:114567-50-9
- Caffeoylcalleryanin
Catalog No.:BCX0031
CAS No.:20300-49-6
- Odontoside
Catalog No.:BCX0030
CAS No.:20300-50-9
- Catechin 7-O-beta-D-glucopyranoside
Catalog No.:BCX0029
CAS No.:65597-47-9
- 5-O-Coumaroylquinic acid
Catalog No.:BCX0028
CAS No.:32451-86-8
- Gossypetin 3-sophoroside-8-glucoside
Catalog No.:BCX0027
CAS No.:77306-93-5
- Alopecurone A
Catalog No.:BCX0026
CAS No.:162558-89-6
- Kidjolanin
Catalog No.:BCX0025
CAS No.:38395-01-6
- Hainanic acid B
Catalog No.:BCX0024
CAS No.:1637737-46-2
- 5-Hydroxy-4a,8-dimethyl-3-methylen-decahydroazuleno[6,5-b]furan-2(3H)-on
Catalog No.:BCX0023
CAS No.:114579-31-6
- Ganoderic acid GS-3
Catalog No.:BCX0022
CAS No.:1206781-66-9
- Scoparin
Catalog No.:BCX0035
CAS No.:301-16-6
- (Z)-Ferulic acid 4-O-beta-D-glucoside
Catalog No.:BCX0036
CAS No.:94942-20-8
- Methyl ganoderate A
Catalog No.:BCX0037
CAS No.:105742-78-7
- (3,4-Dihydroxyphenyl)methyl 3-(beta-D-glucopyranosyloxy)-4-hydroxybenzoate
Catalog No.:BCX0038
CAS No.:877461-90-0
- Bayin
Catalog No.:BCX0039
CAS No.:3681-96-7
- Ganoderic acid GS-2
Catalog No.:BCX0040
CAS No.:1206781-65-8
- Ephedrannin D1
Catalog No.:BCX0041
CAS No.:1592431-55-4
- Ganoderic acid beta
Catalog No.:BCX0042
CAS No.:217476-76-1
- Methyl ganoderate B
Catalog No.:BCX0043
CAS No.:81907-65-5
- Oblongaroside B
Catalog No.:BCX0044
CAS No.:1000889-11-1
- Symplocoside
Catalog No.:BCX0045
CAS No.:76502-76-6
- Mahuannin B
Catalog No.:BCX0046
CAS No.:82796-37-0
An in silico study on pulmonary fibrosis inhibitors from Tinospora cordifolia and Curcuma longa targeting TGF-beta RI.[Pubmed:35068362]
J Biomol Struct Dyn. 2022 Jan 24:1-17.
Pulmonary fibrosis is characterized by damage to the epithelial cells and alveolar-capillary basement membrane. The increased expression levels of transforming growth factor beta (TGF-beta) and TGF-beta-receptor-1 induced differentiation of lung fibroblasts to myofibroblasts, an alarming sign and considered the hallmark event development of pulmonary fibrosis. In the current study, the stability of phytochemicals of Curcuma longa and Tinospora cordifolia as inhibitors of transforming growth factor beta RI (TGF-beta RI) were evaluated using molecular docking and molecular dynamics studies. A total of 108 Curcuma longa and 16 Tinospora cordifolia constituents were screened against TGF-beta RI as the target. Further, their ADMET properties were evaluated using the pkCSM online server. The compounds Tembetarine, magnoflorine from T. cordiolia, and 2-(Hydroxymethyl) anthraquinone and quercetin in C. longa showed significant binding affinities bonding interactions with the target, TGF-beta RI, and the study was compared with the known inhibitors from the literature. The MD simulations study also supported that the selected compounds show a close affinity with the binding site and maintained stable behavior throughout the simulation time. The pharmacophore feature analysis of the selected compounds and inhibitors were analyzed using the pharmagist web server, and the common features like H-bond donor and aromatic ring were mapped.Communicated by Ramaswamy H. Sarma.
In silico analysis of phytoconstituents from Tinospora cordifolia with targets related to diabetes and obesity.[Pubmed:33442530]
In Silico Pharmacol. 2021 Jan 2;9(1):3.
Traditionally, Tinospora cordifolia is commonly used in the treatment of diabetes and obesity; has been evaluated for their anti-diabetic and anti-obese potency in experimental animal models. However, the binding affinity of multiple bioactives with various proteins involved in the pathogenesis of diabetes and obesity has not been reported yet. Hence, the present study aimed to assess the binding affinity of multiple bioactives from T. cordifolia with various targets involved in the pathogenesis of diabetes and obesity. The ligands and targets were retrieved from the PubChem and Protein Data Bank respectively and docked using autodock4.0. Druglikeness and absorption, distribution, metabolism, excretion, and toxicity profile were predicted using Molsoft and admetSAR1 respectively. The multiple bioactives from T. cordifolia were identified to interact with multiple proteins involved in the pathogenesis of diabetes/obesity, i.e., isocolumbin (- 9 kcal/mol) with adiponectin (PDB: 4DOU), beta-sitosterol (- 10.9 kcal/mol) with cholesteryl ester transfer protein (PDB: 2OBD), tinocordiside (- 6.9 kcal/mol) with lamin A/C (PDB: 3GEF), berberine (- 9.5 kcal/mol) with JNK1 (PDB:3ELJ), beta-sitosterol & isocolumbin (- 10.1 kcal/mol) with peroxisome proliferator-activated receptor-gamma (PDB:4CI5), berberine (- 7.5 kcal/mol) with suppressor of cytokine signaling 3 (PDB: 2BBU), isocolumbin (- 9.6 kcal/mol) with pancreatic alpha-amylase (PDB: 1B2Y), isocolumbin (- 9 kcal/mol) with alpha-glucosidase (PDB: 3TOP), and beta-sitosterol (- 10.8 kcal/mol) with aldose reductase (PDB: 3RX2). Similarly, among the selected bioactives, Tembetarine scored highest druglikeness score, i.e., 1.21. In contrast, isocolumbin scored lowest drug-likeness character i.e. - 0.52. The predicted result of phytochemicals from T. cordifolia for acute oral toxicity, rat acute toxicity, fish toxicity, drug-likeness score, and aqueous solubility showed the probability of lower side/adverse effects in human consumption. The study suggests processing for bioactives from T. cordifolia against diabetes and obesity via in-vitro and in-vivo approaches.
Screening of phytochemical compounds of Tinospora cordifolia for their inhibitory activity on SARS-CoV-2: an in silico study.[Pubmed:32627715]
J Biomol Struct Dyn. 2021 Sep;39(15):5799-5803.
In the present study, we explored phytochemical constituents of Tinospora cordifolia in terms of its binding affinity targeting the active site pocket of the main protease (3CL pro) of SARS-CoV-2 using molecular docking study and assessed the stability of top docking complex of tinosponone and 3CL pro using molecular dynamics simulations with GROMACS 2020.2 version. Out of 11 curated screened compounds, we found the significant docking score for tinosponone, xanosporic acid, cardiofolioside B, Tembetarine and berberine in Tinospora cordifolia. Based on the findings of the docking study, it was confirmed that tinosponone is the potent inhibitor of main protease of SARS-CoV-2 with the best binding affinity of -7.7 kcal/mol. Further, ADME along with toxicity analysis was studied to predict the pharmacokinetics and drug-likeness properties of five top hits compounds. The molecular dynamics simulation analysis confirmed the stability of tinosponone and 3CL pro complex with a random mean square deviation (RMSD) value of 0.1 nm. The computer-aided drug design approach proved that the compound tinosponone from T. cordifolia is a potent inhibitor of 3CL main protease of SARS-CoV-2. Further, the in vitro and in vivo-based testing will be required to confirm its inhibitory effect on SARS-CoV-2.Communicated by Ramaswamy H. Sarma.
Anti-inflammatory and antinociceptive activities of Croton urucurana Baillon bark.[Pubmed:26944237]
J Ethnopharmacol. 2016 May 13;183:128-135.
ETHNOPHARMACOLOGICAL RELEVANCE: Croton urucurana (Euphorbiaceae) is popularly used in Brazil to treat inflammatory processes, pain, and gastric ulcers. AIM OF STUDY: To evaluate the anti-inflammatory and antinociceptive properties of the methanol extract from the bark of C. urucurana (MECu) in mice and identify its chemical constituents. MATERIALS AND METHODS: The extract was characterized by UHPLC-DAD-ESI-Q-TOF-MS/MS. Extract doses of 25, 100, and 400mg/kg were employed in the biological assays. Evaluation of anti-inflammatory activity was based on paw edema and leukocyte recruitment into the peritoneal cavity of mice, both induced by carrageenan. Abdominal writhing caused by acetic acid and duration of formalin-induced paw-licking were the models employed to evaluate antinociceptive activity. RESULTS: Ten compounds were identified in the extract: (+)-gallocatechin (1), procyanidin B3 (2), (+)-catechin (3), (-)-epicatechin (4), Tembetarine (5), magnoflorine (6), taspine (7), methyl-3-oxo-12-epi-barbascoate (8), methyl-12-epi-barbascoate (9), and hardwickiic acid (10). This is the first report of compounds 2, 4, 6, 7, and 10 in C. urucurana and compound 5 in the genus Croton. In addition to inhibiting paw edema and leukocyte recruitment (particularly of polymorphonuclear cells) into the peritoneal cavity of mice, MECu reduced the number of abdominal writhings induced by acetic acid and the duration of formalin-induced paw licking. CONCLUSIONS: The methanol extract of C. urucurana bark exhibited anti-inflammatory and antinociceptive properties, corroborating its use in folk medicine. These effects may be related to the presence of diterpenes, alkaloids, and flavonoids.
Studies on the alkaloids of the bark of Magnolia officinalis: isolation and on-line analysis by HPLC-ESI-MS(n).[Pubmed:23823874]
Molecules. 2013 Jul 3;18(7):7739-50.
The bark of Magnolia officinalis is a well-known Traditional Chinese Medicine. In the present study, two new alkaloids, named (S)-4-keto-magnoflorine (6) and (R)-3,4-dehydromagnocurarine (11), together with seven known alkaloids: (S)-magnoflorine (5), trans/cis N-feruloylputrescine (7/8), (R)-magnocurarine (10), (S)-Tembetarine (12), (R)-oblongine (14), and (R)-asimilobine (17) were isolated and their structures elucidated by spectroscopic methods, including 1D, 2D NMR, and HRESI-MS. The absolute configurations of the isoquinoline alkaloids 5, 6, 10-12, 14, and 17 were determined by CD. In vitro inhibitory activities against aldose reductase, lipase, alpha-glucosidase, DPP-IV and three cancer cell lines (A549, Bel-7402, and HCT-8) were evaluated for all isolated compounds. However, all compounds showed weak activities in all tests at the same concentration as the positive control drugs. An HPLC-ESI-MS(n) method has been established for screening of alkaloids in the bark of M. officinalis. A total of 23 alkaloids were identified or tentatively characterized; including 13 aporphines, eight benzylisoquinolines and two amides. Plausible fragmentation pathways of the representative compounds 6, 7/8, 11, and 17 were proposed in the present study.
[Study on chemical constituents from branches and leaves of Polyalthia nemoralis].[Pubmed:21809577]
Zhongguo Zhong Yao Za Zhi. 2011 Apr;36(8):1024-7.
OBJECTIVE: To investigate the chemical constituents of the branches and leaves of Polyalthia nemoralis. METHOD: The compounds were isolated and purified by silica gel, macroporous adsorption resin and Sephadex LH-20 column chromatographic methods. Their chemical structures were elucidated on the basis of physicochemical properties and spectral data. RESULT: Fourteen compounds were isolated and identified as syringic acid (1), 3-methoxy-4-hydroxycinnamic acid (2), vanillic acid (3), 4-hydroxybenzoic acid (4), mauritianin (5), (+)-xylopinidine (6), (+)-oblongine(7), (+)-Tembetarine (8), eythritol (9), D-mannitol (10), ethyl-beta-D-glucopyranoside (11), (+)-magnoflorine (12), stepharanine (13), (2S, 4R)-4-hydroxy-2-piperidine-carboxylic acid (14), respectively. CONCLUSION: All the compounds were isolated from the genus Polyalthia for the first time; compounds 6 and 13 showed inhibitation activities against multi tumor cell lines.
Quaternary isoquinoline alkaloids from Xylopia parviflora.[Pubmed:15081299]
Phytochemistry. 2004 Apr;65(7):939-44.
From the quaternary alkaloidal fraction of the bark and the root of Xylopia parviflora (Annonaceae), four isoquinoline alkaloids, xylopinidine, dehydrocoreximine, N, N-dimethylanomurine and N-methylphoebine were isolated along with the known compounds, pycnarrhine, lotusine, 6,7-dimethoxy-2-methyl-isoquinolinium salt, 1,2-dehydroreticuline, (-)-phellodendrine, (+)-Tembetarine, (-)-litcubine, (+)-magnoflorine, tetradehydroreticuline, (-)-oblongine, (+)-menisperine, (+)-N-methylcorydine, stepharanine, (+)-xanthoplanine, dehydrodiscretine, jatrorrhizine and palmatine. 3,4-Dihydro-6,7-dimethoxy-2-methyl-isoquinolinium and N-methylpurpuerine were isolated as natural products for the first time. Their structures were determined on the basis of spectroscopic evidence.
Antifeedant constituents from Fagara macrophylla.[Pubmed:11429249]
Fitoterapia. 2001 Jun;72(5):538-43.
Analysis of the polar fractions of an EtOH extract obtained from the bark of the African medicinal plant Fagara macrophylla led to the isolation and identification of the alkaloids oblongine (6), Tembetarine (7) and magnoflorine (8) and the flavonoid hesperidin (9). These compounds, together with other metabolites (1--5) previously isolated from F. macrophylla, were tested for antifeedant activity in a binary-choice bioassay. The acridone alkaloid xanthoxoline (4) was found to have a potent antifeedant activity against larvae of both Spodoptera frugiperda and S. littoralis. 1-Hydroxy-3-methoxy-N-methyl-acridone (2), arborinine (3), Tembetarine (7) and magnoflorine (8) were antifeedant against S. frugiperda.
Further alkaloids from bark of Fagara mayu.[Pubmed:17404954]
Planta Med. 1983 Jun;48(2):77-80.
In addition to the alkaloids canthin-6-one; dictamnine, chelerythrine, gamma-fagarine, skimmianine, magnoflorine and 11-(2'-ketobutane)-dihydrochelerythrine, four further alkaloids were isolated from the bark of Fagara mayu (Bert. ex Hook et Arn.) Engler and edulinine, bocconoline, Tembetarine were identified. A new base, N-methylpseudolaudanine, was also isolated.


