ToddaculineCAS# 4335-12-0 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 4335-12-0 | SDF | Download SDF |
| PubChem ID | 5321960 | Appearance | Cryst. |
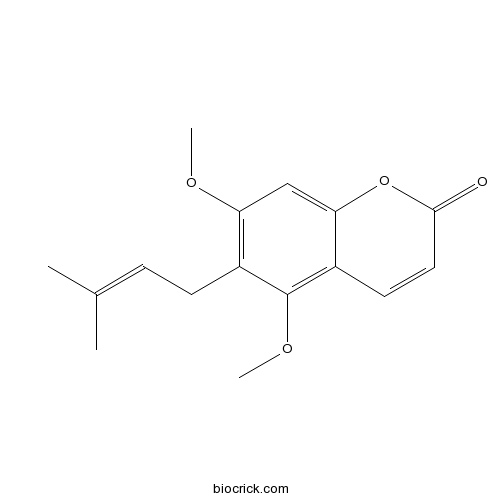
| Formula | C16H18O4 | M.Wt | 274.3 |
| Type of Compound | Coumarins | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | 5,7-dimethoxy-6-(3-methylbut-2-enyl)chromen-2-one | ||
| SMILES | CC(=CCC1=C(C=C2C(=C1OC)C=CC(=O)O2)OC)C | ||
| Standard InChIKey | KRQHZFHWEAJPNO-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C16H18O4/c1-10(2)5-6-11-13(18-3)9-14-12(16(11)19-4)7-8-15(17)20-14/h5,7-9H,6H2,1-4H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. Toddaculine can not only inhibit the differentiation of osteoclasts via activation of the NF-κB, ERK 1/2, and p38 MAPK signaling pathways, but can induce differentiation and mineralization of osteoblasts by regulating differentiation factors. 2. Toddaculine may be beneficial for the prevention and treatment of osteoporosis. 3. Toddaculine displays a dual effect as a cell differentiating agent and apoptosis inducer in U-937 cells, suggesting it may serve as a pharmacological prototype for the development of novel anti-leukemic agents. |
| Targets | NF-kB | p38MAPK | ERK | Akt |

Toddaculine Dilution Calculator

Toddaculine Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.6456 mL | 18.2282 mL | 36.4564 mL | 72.9129 mL | 91.1411 mL |
| 5 mM | 0.7291 mL | 3.6456 mL | 7.2913 mL | 14.5826 mL | 18.2282 mL |
| 10 mM | 0.3646 mL | 1.8228 mL | 3.6456 mL | 7.2913 mL | 9.1141 mL |
| 50 mM | 0.0729 mL | 0.3646 mL | 0.7291 mL | 1.4583 mL | 1.8228 mL |
| 100 mM | 0.0365 mL | 0.1823 mL | 0.3646 mL | 0.7291 mL | 0.9114 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Boc-Tyr-OMe
Catalog No.:BCC3459
CAS No.:4326-36-7
- 6-Hydroxykaempferol
Catalog No.:BCN3334
CAS No.:4324-55-4
- Formoterol Hemifumarate
Catalog No.:BCC4349
CAS No.:43229-80-7
- Fenbendazole
Catalog No.:BCC1236
CAS No.:43210-67-9
- Skp2 Inhibitor C1
Catalog No.:BCC6298
CAS No.:432001-69-9
- 6-(5-Chloropyridin-2-yl)-7-hydroxy-6,7-dihydro-5H-pyrrolo[3,4-b]pyrazin-5-one
Catalog No.:BCC8753
CAS No.:43200-81-3
- Zopiclone
Catalog No.:BCC9195
CAS No.:43200-80-2
- Allylestrenol
Catalog No.:BCC8814
CAS No.:432-60-0
- SJ 172550
Catalog No.:BCC2416
CAS No.:431979-47-4
- 7'-O-Ethylmarmin
Catalog No.:BCC8274
CAS No.:
- Necrostatin-1
Catalog No.:BCC2247
CAS No.:4311-88-0
- DPO-1
Catalog No.:BCC7398
CAS No.:43077-30-1
- 3-O-Acetyloleanolic acid
Catalog No.:BCN5486
CAS No.:4339-72-4
- VU 0357121
Catalog No.:BCC4595
CAS No.:433967-28-3
- Ethisterone
Catalog No.:BCC4478
CAS No.:434-03-7
- Methenolone acetate
Catalog No.:BCC9028
CAS No.:434-05-9
- Oxymetholone
Catalog No.:BCC4692
CAS No.:434-07-1
- Lithocholic Acid
Catalog No.:BCC3805
CAS No.:434-13-9
- Nandrolone
Catalog No.:BCC9086
CAS No.:434-22-0
- Dacarbazine
Catalog No.:BCC1174
CAS No.:4342-03-4
- K 41498
Catalog No.:BCC5867
CAS No.:434938-41-7
- L-5-Hydroxytryptophan
Catalog No.:BCC8106
CAS No.:4350-09-8
- H-Arg(Tos)-OH
Catalog No.:BCC2867
CAS No.:4353-32-6
- 5-Hydroxy-9-(3,4,5-trimethoxyphenyl)-5a,6,8a,9-tetrahydro-5H-[2]benzofuro[5,6-f][1,3]benzodioxol-8-one
Catalog No.:BCC8350
CAS No.:4354-76-1
Toddaculin, a natural coumarin from Toddalia asiatica, induces differentiation and apoptosis in U-937 leukemic cells.[Pubmed:22537907]
Phytomedicine. 2012 Jun 15;19(8-9):737-46.
Chemotherapeutics represent the main approach for the treatment of leukemia. However, the occurrence of adverse side effects and the complete lack of effectiveness in some cases make it necessary to develop new drugs. As part of our screening program to evaluate the potential chemotherapeutic effect of natural coumarins, we investigated the anti-leukemic activities of a series of six prenylated coumarins isolated from the stem bark of Toddalia asiatica (Rutaceae). Among these, 6-(3-methyl-2-butenyl)-5,7-dimethoxycoumarin (toddaculin) displayed the most potent cytotoxic and anti-proliferative effects in U-937 cells. To determine whether these effects resulted from induction of cell death or differentiation, we further evaluated the expression of several apoptosis and maturation markers. Interestingly, while toddaculin at 250 muM was able to induce apoptosis in U-937 cells, involving decreased phosphorylation levels of ERK and Akt, 50 muM toddaculin exerted differentiating effects, inducing both the capacity of U-937 cells to reduce NBT and the expression of differentiation markers CD88 and CD11b, but no change in p-Akt or p-ERK levels. Taken together, these findings indicate that toddaculin displays a dual effect as a cell differentiating agent and apoptosis inducer in U-937 cells, suggesting it may serve as a pharmacological prototype for the development of novel anti-leukemic agents.
Toddaculin, Isolated from of Toddalia asiatica (L.) Lam., Inhibited Osteoclastogenesis in RAW 264 Cells and Enhanced Osteoblastogenesis in MC3T3-E1 Cells.[Pubmed:25993011]
PLoS One. 2015 May 18;10(5):e0127158.
Osteoporosis with bone loss is widely recognized as a major health problem. Bone homeostasis is maintained by balancing bone formation and bone resorption. The imbalance caused by increased bone resorption over bone formation can lead to various bone-related diseases such as osteoporosis and rheumatoid arthritis. Osteoclasts are the principal cells responsible for bone resorption and the main targets of anti-resorptive therapies. However, excessive inhibition of osteoclast differentiation may lead to inhibition of osteoblast differentiation. Therefore, it is important to screen for new compounds capable of inhibiting bone resorption and enhancing bone formation. Toddalia asiatica (L.) Lam. has been utilized traditionally for medicinal purposes such as the treatment of rheumatism. Currently, the extract is considered to be a good source of pharmacological agents for the treatment of bone-related diseases, but the active compounds have yet to be identified. We investigated whether toddaculin, derived from Toddalia asiatica (L.) Lam., affects both processes by inhibiting bone resorption and enhancing bone formation. Towards this end, we used pre-osteoclastic RAW 264 cells and pre-osteoblastic MC3T3-E1 cells. We found that toddaculin not only inhibited the differentiation of osteoclasts via activation of the NF-kappaB, ERK 1/2, and p38 MAPK signaling pathways, but it also induced differentiation and mineralization of osteoblasts by regulating differentiation factors. Thus, toddaculin might be beneficial for the prevention and treatment of osteoporosis.


