Vilanterol trifenatateβ2-AR agonist CAS# 503070-58-4 |
- Etifoxine
Catalog No.:BCC1560
CAS No.:21715-46-8
- Etomidate
Catalog No.:BCC1150
CAS No.:33125-97-2
- Acamprosate calcium
Catalog No.:BCC1327
CAS No.:77337-73-6
- Flumazenil
Catalog No.:BCC1259
CAS No.:78755-81-4
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 503070-58-4 | SDF | Download SDF |
| PubChem ID | 44482554 | Appearance | Powder |
| Formula | C44H49Cl2NO7 | M.Wt | 774.77 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | GW642444M | ||
| Solubility | DMSO : 100 mg/mL (129.07 mM; Need ultrasonic) H2O : < 0.1 mg/mL (insoluble) | ||
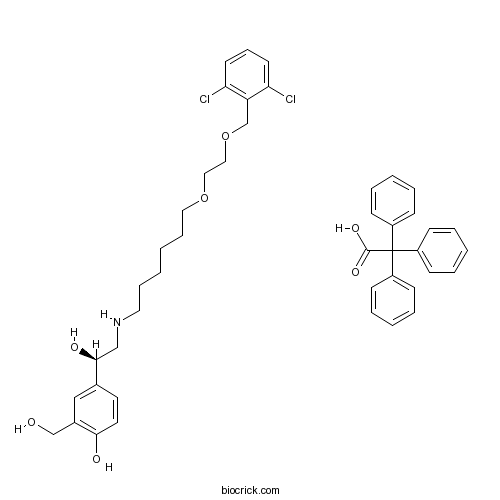
| Chemical Name | 4-[(1R)-2-[6-[2-[(2,6-dichlorophenyl)methoxy]ethoxy]hexylamino]-1-hydroxyethyl]-2-(hydroxymethyl)phenol;2,2,2-triphenylacetic acid | ||
| SMILES | C1=CC=C(C=C1)C(C2=CC=CC=C2)(C3=CC=CC=C3)C(=O)O.C1=CC(=C(C(=C1)Cl)COCCOCCCCCCNCC(C2=CC(=C(C=C2)O)CO)O)Cl | ||
| Standard InChIKey | KLOLZALDXGTNQE-JIDHJSLPSA-N | ||
| Standard InChI | InChI=1S/C24H33Cl2NO5.C20H16O2/c25-21-6-5-7-22(26)20(21)17-32-13-12-31-11-4-2-1-3-10-27-15-24(30)18-8-9-23(29)19(14-18)16-28;21-19(22)20(16-10-4-1-5-11-16,17-12-6-2-7-13-17)18-14-8-3-9-15-18/h5-9,14,24,27-30H,1-4,10-13,15-17H2;1-15H,(H,21,22)/t24-;/m0./s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Vilanterol trifenatate is a long-acting β2-adrenoceptor (β2-AR) agonist with 24 h activity. The pEC50s for β2-AR,β1-AR and β3-AR is 10.37±0.05, 6.98±0.03 and 7.36±0.03, respectively.In Vitro:The selectivity of Vilanterol trifenatate for β2-AR over the other β-AR receptor subtypes (β2 and β3) is established by testing the ability of Vilanterol to elicit concentration-dependent increases in cAMP in CHO cells expressing human β1-, β2-, and β3-AR. Vilanterol is demonstrated to be highly selective for the β2-AR with at least a 1000-fold selectivity over both β2- and β3-AR subtypes. This analysis results in a low-affinity pKD for [3H]Vilanterol of 9.44±0.07 (n=4) in the presence Gpp(NH)p and a high-affinity pKD of 10.82±0.12 (n=4) and a low-affinity pKD 9.47±0.17 (n=4) in the absence of Gpp(NH)p. In addition, a low-affinity pKD for [3H]Vilanterol of 9.52±0.24 (n=4) in the absence of Gpp(NH)p (37°C) is observed[1]. Vilanterol trifenatate is a novel inhaled long-acting β2-agonist with inherent 24 h activity in vitro in development as a combination with the inhaled corticosteroid fluticasone furoate for both COPD and asthma[2]. Vilanterol is a novel long-acting β2-agonist (LABA) with inherent 24-hour activity for once-daily clinical treatment of chronic obstructive pulmonary disease (COPD) and asthma in combination with the inhaled novel corticosteroid fluticasone furoate, also active for 24 hours[3]. References: | |||||

Vilanterol trifenatate Dilution Calculator

Vilanterol trifenatate Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.2907 mL | 6.4535 mL | 12.9071 mL | 25.8141 mL | 32.2676 mL |
| 5 mM | 0.2581 mL | 1.2907 mL | 2.5814 mL | 5.1628 mL | 6.4535 mL |
| 10 mM | 0.1291 mL | 0.6454 mL | 1.2907 mL | 2.5814 mL | 3.2268 mL |
| 50 mM | 0.0258 mL | 0.1291 mL | 0.2581 mL | 0.5163 mL | 0.6454 mL |
| 100 mM | 0.0129 mL | 0.0645 mL | 0.1291 mL | 0.2581 mL | 0.3227 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Vilanterol trifenatate is a novel and selective agonist of β2-AR with a PEC50 value of 10.37±0.05 [1].
Vilanterol trifenatate is a novel long-acting β2-AR agonist (LABA) with 24h activity in development for inhaled once daily treatment. In the radioligand binding studies, Vilanterol trifenatate has shown the binding affinity in the one-affinity site model with pKD values of 9.44±0.07 and 10.82±0.12 in the presence Gpp(NH)p and absence Gpp(NH)p, respectively. In dissociation studies, Vilanterol trifenatate has been reported to bind from the β2-AR with a dissociation t1/2 value of 3.5 min in the presence of Gpp(NH)p. Vilanterol trifenatate has been found to have a good selectivity for β2-AR over the other β-AR receptor subtypes(β1and β3) with pEC50 values of 10.37±0.05, 6.98±0.03 and 7.36±0.03, respectively. Vilanterol trifenatate has exhibited at least 1000-fold selectivity over both β1-and β3-AR subtypes [1].
References:
[1] Slack RJ1, Barrett VJ, Morrison VS, Sturton RG, Emmons AJ, Ford AJ, Knowles RG.In vitro pharmacological characterization of vilanterol, a novel long-acting β2-adrenoceptor agonist with 24-hour duration of action.J Pharmacol Exp Ther. 2013 Jan; 344(1):218-30.
- Vilanterol
Catalog No.:BCC4030
CAS No.:503068-34-6
- Boc-His(Z)-OH
Catalog No.:BCC3404
CAS No.:50305-43-6
- Cucurbitacin IIb
Catalog No.:BCN2519
CAS No.:50298-90-3
- Erythrocentaurin
Catalog No.:BCN7684
CAS No.:50276-98-7
- Oleuropeic acid
Catalog No.:BCN5611
CAS No.:5027-76-9
- Phloracetophenone 4'-O-glucoside
Catalog No.:BCN4052
CAS No.:5027-30-5
- Lonidamine
Catalog No.:BCC9012
CAS No.:50264-69-2
- SQ109
Catalog No.:BCC1962
CAS No.:502487-67-4
- NIDA 41020
Catalog No.:BCC7810
CAS No.:502486-89-7
- H-Trp-NH2.HCl
Catalog No.:BCC3112
CAS No.:5022-65-1
- HLI 373
Catalog No.:BCC2408
CAS No.:502137-98-6
- Cyclopentadecanone
Catalog No.:BCN3822
CAS No.:502-72-7
- N-Methylflindersine
Catalog No.:BCN3641
CAS No.:50333-13-6
- 3'-Galloylquercitrin
Catalog No.:BCN8254
CAS No.:503446-90-0
- NU7441 (KU-57788)
Catalog No.:BCC3679
CAS No.:503468-95-9
- glucagon receptor antagonists 1
Catalog No.:BCC1593
CAS No.:503559-84-0
- Pentamidine dihydrochloride
Catalog No.:BCC5194
CAS No.:50357-45-4
- Apixaban
Catalog No.:BCC2295
CAS No.:503612-47-3
- Cyclo(Phe-Gly)
Catalog No.:BCN2431
CAS No.:5037-75-2
- (-)-di-de-Omethylgrandisin
Catalog No.:BCN7872
CAS No.:50393-98-1
- Orcinol
Catalog No.:BCN5612
CAS No.:504-15-4
- 4-Aminopyridine
Catalog No.:BCC5267
CAS No.:504-24-5
- DL-Homocysteic acid
Catalog No.:BCN2233
CAS No.:504-33-6
- 3-Nitropropionic acid
Catalog No.:BCC6303
CAS No.:504-88-1
Fluticasone furoate and vilanterol trifenatate combination therapy for the treatment of asthma.[Pubmed:27221165]
Expert Rev Respir Med. 2016 Aug;10(8):839-47.
INTRODUCTION: Combination inhaled corticosteroids (ICS) and long acting beta2-adrenergic agonists (LABA) are used in a stepwise fashion for patients whose asthma is not controlled by low dose ICS alone. Adherence is one of the main issues facing clinicians in the control of asthma symptoms with currently available combination inhalers requiring twice-daily (BD) inhalation. Fluticasone furoate (FF) and Vilanterol trifenatate (VI) both have prolonged retention in the lung with effects on lung function over 24-hours and as such the combination has been proposed for once-daily (OD) dosing. AREAS COVERED: The stepwise pharmacologic approach to asthma management is addressed, followed by a detailed assessment of the literature pertaining to the efficacy, tolerability and safety of FF/VI combination therapy for the treatment of asthma. Expert commentary: Doses of 100/25microg and 200/25microg inhaled OD, have similar improvements in lung function, asthma control as well as rates of side effects to one of the currently available BD ICS/LABA combinations, fluticasone propionate and salmeterol (FP/SAL) but have not been compared with other commonly used combinations. It is hoped that OD dosage of FF/VI can improve adherence and hence asthma control in these patients, however evidence to support this has yet to become available.
Spotlight on fluticasone furoate/vilanterol trifenatate for the once-daily treatment of asthma: design, development and place in therapy.[Pubmed:28008228]
Drug Des Devel Ther. 2016 Dec 14;10:4047-4060.
The use of inhaled corticosteroids (ICSs) plays a key role in the treatment of asthmatic patients, and international guidelines have designated ICSs as an early maintenance therapy in controlling asthma symptoms. When asthmatic patients remain symptomatic on ICSs, one common option is to add a long-acting beta2 agonist (LABA) to the maintenance treatment. Fixed combination inhalers that contain both an ICS and a LABA have been popular for both chronic obstructive pulmonary disease (COPD) and asthma. Historically, these inhalers have been dosed twice daily. However, currently, there is a once-daily combination therapy with the ICS fluticasone furoate (FF) and the LABA Vilanterol trifenatate (VI) with indications for use in both COPD and asthma. This dry powder inhaler (DPI) comes in two doses of FF (100 or 200 mug) both combined with VI (25 mug). This article reviews the clinical trial data for FF, VI and FF/VI combination inhalers and documents the efficacy and safety of once-daily inhaled maintenance therapy by DPI in asthmatic patients.
Vilanterol trifenatate for the treatment of COPD.[Pubmed:27143334]
Expert Rev Respir Med. 2016 Jul;10(7):719-31.
INTRODUCTION: Currently the treatment of chronic obstructive pulmonary disease (COPD) has limited effectiveness and there is a need to develop new drugs. International guidelines recommend the use of long-acting bronchodilators (beta2 agonists and anti-cholinergics/muscarinics), inhaled steroids and associations between these drugs in the maintenance treatment of moderate-to-severe COPD. AREA COVERED: Vilanterol trifenate is a new once-daily highly selective beta2-agonist available in USA and Europe in association with umeclidinium bromide (a long-acting anti-muscarnic agent) and fluticasone furoate (an inhaled corticosteroid) for the once-daily maintenance treatment of COPD. Vilanterol combined in fixed-dose treatments has been tested in numerous clinical trials involving thousands of patients. Expert commentary: These new once-daily formulations have the potential to improve compliance to long-term inhaled therapy. This paper will review the clinical and experimental data regarding vilanterol use in the regular treatment of COPD as well as provide a critical discussion of possible future treatment settings.
Phamacology of fluticasone furoate and vilanterol trifenatate combination therapy for asthma.[Pubmed:27599692]
Expert Rev Respir Med. 2016 Oct;10(10):1069-78.
INTRODUCTION: Fluticasone furoate (FF) is a novel inhaled corticosteroid (ICS). Vilanterol trifenate (VI) is a new inhaled, selective, long - acting beta2 adrenergic agonist (LABA). It is now also marketed as a novel once daily combined ICS/LABA indicated for treatment of moderate and severe asthma. AREAS COVERED: FF has a highly specific, fast association and slow dissociation from the glucocorticoid receptor, with a 24 hr duration of action. This, combined with a slow transport out of respiratory cells, creates a long tissue retention period. Vilanterol trifenate (VI) is a new inhaled, selective, long - acting beta2 adrenergic agonist, also with a rapid onset of action with a maximal effect within 6 mins and prolonged lung retention with effects on lung function over 24 hours. Expert commentary: Multiple Phase I-III efficacy studies performed on FF and VI have shown an improvement in spirometry as well as symptom control in asthma. The development of once daily ICS/LABA combinations may potentially improve adherence to asthma therapy, but this has yet to be demonstrated.


