6-DemethoxytangeretinCAS# 6601-66-7 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 6601-66-7 | SDF | Download SDF |
| PubChem ID | 629964 | Appearance | Yellow powder |
| Formula | C19H18O6 | M.Wt | 342.4 |
| Type of Compound | Flavonoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
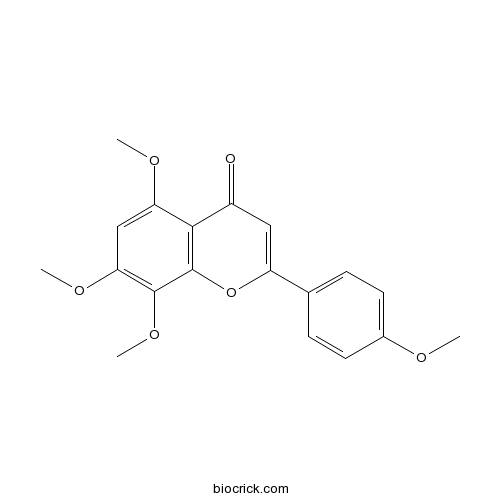
| Chemical Name | 5,7,8-trimethoxy-2-(4-methoxyphenyl)chromen-4-one | ||
| SMILES | COC1=CC=C(C=C1)C2=CC(=O)C3=C(O2)C(=C(C=C3OC)OC)OC | ||
| Standard InChIKey | DDGJUTBQQURRGE-UHFFFAOYSA-N | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. 6-Demethoxytangeretin has anti-inflammatory activity, it also can suppress production and gene expression of interleukin-6 in human mast cell-1 via anaplastic lymphoma kinase and mitogen-activated protein kinase pathways. 2. 6-Demethoxytangeretin can suppress the interleukin 1 (IL-1) induced production of proMMP-9/progelatinase B in rabbit synovial cells in a dose dependent manner (<64 microM). |
| Targets | ALK | IL Receptor | MMP(e.g.TIMP) | ERK | Beta Amyloid | MAPK | PGE |

6-Demethoxytangeretin Dilution Calculator

6-Demethoxytangeretin Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.9206 mL | 14.6028 mL | 29.2056 mL | 58.4112 mL | 73.014 mL |
| 5 mM | 0.5841 mL | 2.9206 mL | 5.8411 mL | 11.6822 mL | 14.6028 mL |
| 10 mM | 0.2921 mL | 1.4603 mL | 2.9206 mL | 5.8411 mL | 7.3014 mL |
| 50 mM | 0.0584 mL | 0.2921 mL | 0.5841 mL | 1.1682 mL | 1.4603 mL |
| 100 mM | 0.0292 mL | 0.146 mL | 0.2921 mL | 0.5841 mL | 0.7301 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Cirsimaritin
Catalog No.:BCN4206
CAS No.:6601-62-3
- H-Nva-OH
Catalog No.:BCC2643
CAS No.:6600-40-4
- Psoralen
Catalog No.:BCN4219
CAS No.:66-97-7
- D-Glucosamine hydrochloride
Catalog No.:BCN5982
CAS No.:66-84-2
- Cycloheximide
Catalog No.:BCC3653
CAS No.:66-81-9
- Dicoumarol
Catalog No.:BCC9225
CAS No.:66-76-2
- Strophantidin
Catalog No.:BCC8255
CAS No.:66-28-4
- Uracil
Catalog No.:BCN4211
CAS No.:66-22-8
- Punicalin
Catalog No.:BCN4961
CAS No.:65995-64-4
- Punicalagin
Catalog No.:BCN1037
CAS No.:65995-63-3
- Isogarciniaxanthone E
Catalog No.:BCN4205
CAS No.:659747-28-1
- Micranoic acid A
Catalog No.:BCN4204
CAS No.:659738-08-6
- (5Z)-7-Oxozeaenol
Catalog No.:BCC7724
CAS No.:66018-38-0
- H-D-Arg(NO2)-OH
Catalog No.:BCC2871
CAS No.:66036-77-9
- Glycyrin
Catalog No.:BCN7681
CAS No.:66056-18-6
- Licoisoflavone A
Catalog No.:BCN2929
CAS No.:66056-19-7
- Gomisin H
Catalog No.:BCN3902
CAS No.:66056-20-0
- Angeloylgomisin H
Catalog No.:BCN2843
CAS No.:66056-22-2
- Benzoylgomisin H
Catalog No.:BCN7242
CAS No.:66056-23-3
- Licoisoflavone B
Catalog No.:BCN6695
CAS No.:66056-30-2
- Licoisoflavanone
Catalog No.:BCN6856
CAS No.:66067-26-3
- Tigloylgomisin H
Catalog No.:BCN6927
CAS No.:66069-55-4
- GW 542573X
Catalog No.:BCC7914
CAS No.:660846-41-3
- Nimodipine
Catalog No.:BCC3823
CAS No.:66085-59-4
A citrus flavonoid, 6-demethoxytangeretin, suppresses production and gene expression of interleukin-6 in human mast cell-1 via anaplastic lymphoma kinase and mitogen-activated protein kinase pathways.[Pubmed:24500009]
Biol Pharm Bull. 2014;37(5):871-6. Epub 2014 Feb 5.
Citrus species has been traditionally used in Korea for the treatment of coughing, sputum and dyspepsia. Of the known citrus flavonoids, 6-Demethoxytangeretin was reported to exert anti-inflammatory activity. In order to determine the anti-allergic activity of 6-Demethoxytangeretin, we examined whether or not 6-Demethoxytangeretin was able to suppress activation of the human mast cell line, HMC-1, induced by phorbol 12-myristate 13-acetate (PMA) plus A23187. Interleukin-6 production and relevant gene expression in activated HMC-1 cells were determined by enzyme-linked immunosorbent assay (ELISA) and quantitative reverse transcription-polymerase chain reaction (RT-PCR) analysis. Also, the involvement of the anaplastic lymphoma kinase (ALK) and mitogen-activated protein kinases (MAPKs) in activated HMC-1 cells were studied. 6-Demethoxytangeretin suppresses interleukin-6 production, tumor necrosis factor-alpha gene expression, ALK and MAPKs in HMC-1 cells stimulated by PMA plus A23187. Therefore, it was evident that 6-Demethoxytangeretin suppressed activation of HMC-1 cells by PMA plus A23187 by inhibiting the activity of ALK and MAPKs and subsequently suppressing gene expression, which suggest that 6-Demethoxytangeretin may be involved in the regulation of mast cell-mediated inflammatory responses.
6-demethoxynobiletin, a nobiletin-analog citrus flavonoid, enhances extracellular signal-regulated kinase phosphorylation in PC12D cells.[Pubmed:23934345]
Biol Pharm Bull. 2013;36(10):1646-9. Epub 2013 Aug 9.
We previously demonstrated that nobiletin, a polymethoxylated flavone isolated from citrus peels, has the potential to improve cognitive dysfunction in patients with Alzheimer's disease (AD). Recent studies suggest that the generation of intraneuronal amyloid-beta (Abeta) oligomers is an early event in the pathogenesis of AD. Abeta oligomers cause deficits in the regulation of the extracellular signal-regulated kinase (ERK) signaling which is critical for consolidation of the memory. Our previous studies revealed that nobiletin activated ERK signaling and subsequent cyclic AMP response element-dependent transcription. In this study, the effects of five nobiletin analogs, 6-demethoxynobiletin, tangeretin, 5-demethylnobiletin, sinensetin, and 6-Demethoxytangeretin, isolated from citrus peels were assessed on ERK phosphorylation in PC12D cells, and the structure-activity relationships were examined. PC12D cells were treated with nobiletin or its analogs, and the cell extracts were analyzed by Western blotting using an antibody specific to phosphorylated ERK. 6-Demethoxynobiletin markedly enhanced ERK phosphorylation in a concentration-dependent manner. These results may be useful in developing drugs and functional foods using citrus peels for the treatment of dementia including AD.
A citrus flavonoid, nobiletin, suppresses production and gene expression of matrix metalloproteinase 9/gelatinase B in rabbit synovial fibroblasts.[Pubmed:10648013]
J Rheumatol. 2000 Jan;27(1):20-5.
OBJECTIVE: Flavonoids including nobiletin are known to exert many biological actions in vitro. We investigated the chondroprotective effect of citrus flavonoids, especially nobiletin, using cultured rabbit synovial fibroblasts and articular chondrocytes. METHODS: We examined the effects of citrus flavonoids on the production and gene expression of matrix metalloproteinases (MMP) and prostaglandin E2 (PGE2)production in rabbit synovial fibroblasts. RESULTS: Six flavonoids isolated from Citrus depressa Rutaceae including tangeretin, 6-Demethoxytangeretin, nobiletin, 5-demethylnobiletin, 6-demethoxynobiletin, and sinensetin suppressed the interleukin 1 (IL-1) induced production of proMMP-9/progelatinase B in rabbit synovial cells in a dose dependent manner (<64 microM); nobiletin most effectively suppressed proMMP-9 production along with the decrease in its mRNA. Nobiletin also reduced IL-1 induced production of PGE2 in the synovial cells, but did not modify the synthesis of total protein. These suppressive effects of nobiletin were also observed in rabbit articular chondrocytes. Nobiletin inhibited proliferation of rabbit synovial fibroblasts in the growth phase. CONCLUSION: These results suggest nobiletin is a novel antiinflammatory candidate that has the potential to inhibit PGE2 production, matrix degradation of the articular cartilage, and pannus formation in osteoarthritis and rheumatoid arthritis.


