9-ACCAS# 723-62-6 |
- Dihydroberberine
Catalog No.:BCN2573
CAS No.:483-15-8
- Sesamolin
Catalog No.:BCN1289
CAS No.:526-07-8
- Carnosol
Catalog No.:BCN1055
CAS No.:5957-80-2
- Harpagide
Catalog No.:BCN4996
CAS No.:6926-08-5
- Levistilide A
Catalog No.:BCN1197
CAS No.:88182-33-6
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 723-62-6 | SDF | Download SDF |
| PubChem ID | 2201 | Appearance | Powder |
| Formula | C15H10O2 | M.Wt | 222.24 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble in DMSO > 10 mM | ||
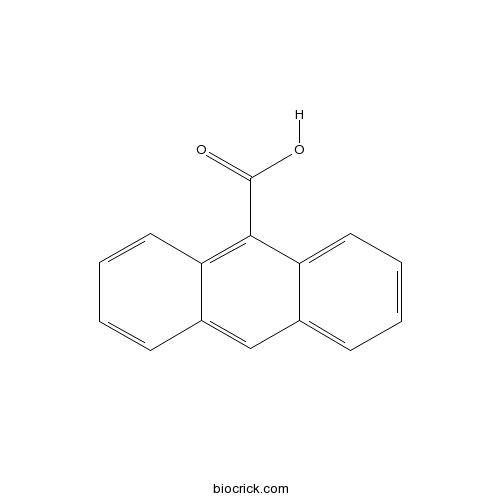
| Chemical Name | anthracene-9-carboxylic acid | ||
| SMILES | C1=CC=C2C(=C1)C=C3C=CC=CC3=C2C(=O)O | ||
| Standard InChIKey | XGWFJBFNAQHLEF-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C15H10O2/c16-15(17)14-12-7-3-1-5-10(12)9-11-6-2-4-8-13(11)14/h1-9H,(H,16,17) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Cl- transport inhibitor with a moderate to strong inhibitory action on PKA activated cardiac IcI. |

9-AC Dilution Calculator

9-AC Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 4.4996 mL | 22.4982 mL | 44.9964 mL | 89.9928 mL | 112.491 mL |
| 5 mM | 0.8999 mL | 4.4996 mL | 8.9993 mL | 17.9986 mL | 22.4982 mL |
| 10 mM | 0.45 mL | 2.2498 mL | 4.4996 mL | 8.9993 mL | 11.2491 mL |
| 50 mM | 0.09 mL | 0.45 mL | 0.8999 mL | 1.7999 mL | 2.2498 mL |
| 100 mM | 0.045 mL | 0.225 mL | 0.45 mL | 0.8999 mL | 1.1249 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Sulfamethoxazole
Catalog No.:BCC4857
CAS No.:723-46-6
- Streptolydigin
Catalog No.:BCN1858
CAS No.:7229-50-7
- Malvidin-3-O-glucoside chloride
Catalog No.:BCN3031
CAS No.:7228-78-6
- Barasertib (AZD1152-HQPA)
Catalog No.:BCC2168
CAS No.:722544-51-6
- AZD1152
Catalog No.:BCC1393
CAS No.:722543-31-9
- Org 24598 lithium salt
Catalog No.:BCC7845
CAS No.:722456-08-8
- Laurifoline
Catalog No.:BCN4278
CAS No.:7224-61-5
- 2alpha-Hydroxyeupatolide 8-O-angelate
Catalog No.:BCN7340
CAS No.:72229-39-1
- 2-Hydroxyeupatolide
Catalog No.:BCN2490
CAS No.:72229-33-5
- Echitovenidine
Catalog No.:BCN7482
CAS No.:7222-35-7
- Parsonsine
Catalog No.:BCN2111
CAS No.:72213-98-0
- Calcitetrol
Catalog No.:BCC1446
CAS No.:72203-93-1
- Rivularine
Catalog No.:BCN2038
CAS No.:723-78-4
- ZLN024
Catalog No.:BCC5527
CAS No.:723249-01-2
- ZJ 43
Catalog No.:BCC2355
CAS No.:723331-20-2
- Beta-Carotene
Catalog No.:BCN4965
CAS No.:7235-40-7
- Licoflavone C
Catalog No.:BCN3256
CAS No.:72357-31-4
- Isobellendine
Catalog No.:BCN1916
CAS No.:72362-45-9
- 2,3-Dihydrobellendine
Catalog No.:BCN1896
CAS No.:72362-47-1
- Yuheinoside
Catalog No.:BCN4279
CAS No.:72396-01-1
- Oxacillin sodium monohydrate
Catalog No.:BCC4816
CAS No.:7240-38-2
- X-Gal
Catalog No.:BCC1211
CAS No.:7240-90-6
- 6-Methoxydihydrosanguinarine
Catalog No.:BCN7581
CAS No.:72401-54-8
- Meglumine Metrizoate
Catalog No.:BCC5631
CAS No.:7241-11-4
Synthesis and structure of dimeric anthracene-9-carboxylato bridged dinuclear erbium(III) complex, [Er(2)(9-AC)(6)(DMF)(2)(H(2)O)(2)].[Pubmed:19167268]
Spectrochim Acta A Mol Biomol Spectrosc. 2009 May;72(4):884-9.
We study the influence of the bulky aromatic rings, e.g. anthracence-9-carboxylic acid (9-ACA) with a large conjugated pi-system on the structure and spectroscopic properties of [Er(2)(9-AC)(6)(DMF)(2)(H(2)O)(2)] complex where 9-AC=anthracence-9-carboxylato and DMF=N,N'-dimethylformamide. The complex has been prepared from the erbium chloride and 9-ACA in the mixture of H(2)O:DMF solution (4:1, v/v) followed by pH adjustment to 6. The complex is crystallized in a monoclinic system with space group P2(1)/n. The two Er(III) ions are double bridged by the deprotonated carboxyl groups of two 9-AC anions (O1 and O1A), forming an eight-coordination number. The chelating bidentate (O,O), chelating-bridging tridentate (O,O,O') and monodentate of 9-AC anions are observed in the dinuclear [Er(2)(9-AC)(6)(DMF)(2)(H(2)O)(2)] complex. The Er-Er distance is 4.015A in the dimeric unit. Intramolecular O-Hcdots, three dots, centeredO and C-Hcdots, three dots, centeredO hydrogen bonds as well as numerous of intermolecular C-Hcdots, three dots, centeredpi interactions between the anthracene rings by edge-to-face interactions linked the dinuclear dimeric units into two-dimensional supramolecular network in a propeller-arrangement. Electronic absorption spectra of the Er(III) complex and its salt were measured. The emission spectrum of the complex is composed of a broad band due to the emission of intraligand pi*-->pi transition from the 9-AC anions and a shoulder peak originating from the 4f-4f emission transition of the Er(III) ions. The complex has a high thermal stability which can be attributed to the effectively increase the rigidity of the 9-AC anions.
Pharmacokinetic modeling of absorption behavior of 9-aminocamptothecin (9-AC) released from colon-specific HPMA copolymer-9-AC conjugate in rats.[Pubmed:17929146]
Pharm Res. 2008 Jan;25(1):218-26.
PURPOSE: To quantitate and predict colon-specific 9-aminocamptothecin (9-AC) release from the N-(2-hydroxypropyl)methacrylamide (HPMA) copolymer-9-AC conjugate and its absorption behavior after oral administration in rats. METHODS: Drug distribution in the gastrointestinal (GI) tract and the plasma concentration-time profile of 9-AC released from the HPMA copolymer conjugate were predicted using the degradation, transit, and absorption rate constants in cecum. The fate of 9-AC in cecum and liver was measured by in-situ cecum absorption and liver perfusion. RESULTS: Following oral administration of the conjugate, 9-AC was released rapidly in cecum. Based on the pharmacokinetic model, up to 60% of the dose was in the cecum at ~6 h, and 7% of the dose still remained there at 24 h. The predicted plasma concentration curve for released 9-AC after an oral dose of 3 mg/kg of 9-AC equivalent increased gradually and reached a peak of 98 nM at 7 h, then started decreasing slowly to 16 nM at 24 h. The bioavailability value was estimated as 0.31 after the first-pass elimination. CONCLUSIONS: A pharmacokinetic model delineated the impact of GI transit, drug absorption rate, and first-pass metabolism on drug disposition following oral administration of HPMA copolymer-9-AC conjugate in rats.
Phase II evaluation of 9-aminocamptothecin (9-AC, NSC #603071) in platinum-resistant ovarian and primary peritoneal carcinoma: a Gynecologic Oncology Group Study.[Pubmed:15589582]
Gynecol Oncol. 2005 Jan;96(1):67-71.
OBJECTIVE: To estimate the antitumor activity of 9-aminocamptothecin (9-AC) in patients with recurrent platinum-"resistant" ovarian cancer; and to determine the nature and degree of toxicity of 9-AC in this cohort of patients. METHODS: A multicenter phase II study was conducted by the Gynecologic Oncology Group (GOG). Patients were to receive 9-AC (colloidal dispersion) 25 microg/m(2)/h (600 microg/m(2)/day) IV over 120 h (5 days) beginning days 1 and 8. Dose adjustment was permitted for toxicity. This schedule was repeated every 21 days until disease progression or unacceptable adverse events. Hematopoietic growth factor support was used as necessary. RESULTS: From January 1999 to December 2000, 29 member institutions of the GOG enrolled 58 patients. Two patients received no therapy; thus, 56 (97%) were evaluable. Median age was 61 (range: 33-81) years. A median of four (range: 1-32) courses of 9-AC was administered. The most frequent grade 3 or 4 toxicities were neutropenia in 46%, leukopenia in 37%, gastrointestinal in 29%, anemia in 25%, and thrombocytopenia in 21%. There was one possible treatment-related death. There were four (7%) complete and four (7%) partial responses, for an overall response rate of 14%. Eighteen (32%) patients had stable disease, 22 (39%) progressed, and response could not be assessed in 8 (14%). CONCLUSION: The 9-AC at this dose and schedule showed limited activity comparable to that seen with other agents in platinum-resistant ovarian or primary peritoneal cancer.
Pre-irradiation 9-amino [20s] camptothecin (9-AC) in patients with newly diagnosed glioblastoma multiforme.[Pubmed:16086097]
Invest New Drugs. 2006 May;24(3):177-80.
PURPOSE: To evaluate the efficacy of 9-amino [20s] camptothecin (9-AC) given before radiation therapy to patients with newly diagnosed glioblastoma multiforme (GBM). METHODS: Eligible patients had newly diagnosed GBM who had residual measurable contrast-enhancing tumor. The trial was a phase 2 trial of 9-AC at 1100 microg/m2 /24 h infused over 72 h every two weeks for up to six cycles in patients with newly diagnosed GBM before radiation therapy. RESULTS: Fourteen patients entered the study and all were evaluable. All of the patients had progressive disease by imaging criteria after at least two cycles of 9-AC (1 month). The median overall survival was 7.5 months (range 1.5-18 months). The most common adverse event was transient lymphopenia (grade 3-4). One patient developed grade 4 neutropenic fever that resolved after three days of diagnosis. CONCLUSIONS: 9-AC lacks activity against glioblastoma multiforme (GBM). Further studies looking at the efficacy of 9-AC in GBM may be futile.


