BerbamineCAS# 478-61-5 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 478-61-5 | SDF | Download SDF |
| PubChem ID | 275182 | Appearance | Powder |
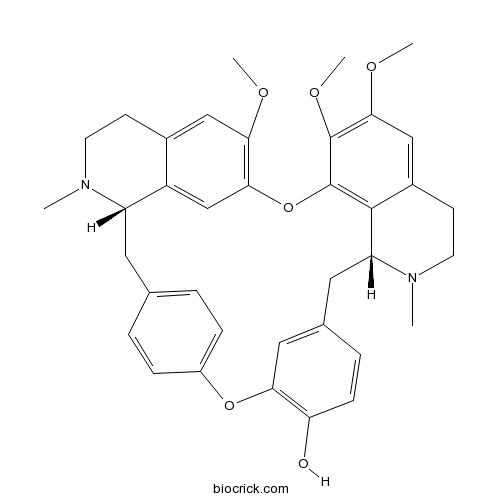
| Formula | C37H40N2O6 | M.Wt | 608.7 |
| Type of Compound | Alkaloids | Storage | Desiccate at -20°C |
| Solubility | DMSO : 125 mg/mL (205.35 mM; Need ultrasonic) | ||
| SMILES | CN1CCC2=CC(=C3C=C2C1CC4=CC=C(C=C4)OC5=C(C=CC(=C5)CC6C7=C(O3)C(=C(C=C7CCN6C)OC)OC)O)OC | ||
| Standard InChIKey | DFOCUWZXJBAUSQ-URLMMPGGSA-N | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Berbamine is a novel inhibitor of bcr/abl fusion gene with potent anti-leukemia activity and also an inhibitor of NF-κB. Berbamine may be the first ATP-competitive inhibitor of CaMKII γ, could be as a new type of molecular targeted agent through inhibition of the CaMKII γ activity for treatment of leukemia.Berbamine confers cardioprotection against I/R injury by attenuating [Ca(2+)inf(i) overloading and preventing calpain activation through the activation of the PI3K-Akt-GSK3β pathway and, subsequently, opening of the mitoK(ATP) channel. |
| Targets | Bcl-2/Bax | TGF-β/Smad | p21 | c-Myc | Calcium Channel | ATPase | Akt | GSK-3 | PI3K |
| In vitro | Berbamine enhances the antineoplastic activity of gemcitabine in pancreatic cancer cells by activating transforming growth factor-β/Smad signaling.[Pubmed: 24619961]Anat Rec (Hoboken). 2014 May;297(5):802-9.Drug-resistance to gemcitabine chemotherapy in pancreatic cancer is still an unsolved problem. Combinations of other chemotherapy drugs with gemcitabine have been shown to increase the efficacy of gemcitabine-based treatment.
Berbamine protects the heart from ischemia/reperfusion injury by maintaining cytosolic Ca(2+) homeostasis and preventing calpain activation.[Pubmed: 22664727]Circ J. 2012;76(8):1993-2002. Epub 2012 May 15.Berbamine, a natural compound from Barberry, was reported to protect myocardium from ischemia/reperfusion (I/R) injury, but the underlying mechanisms are largely unknown.
|
| In vivo | Berbamine inhibits the growth of liver cancer cells and cancer-initiating cells by targeting Ca2⁺/calmodulin-dependent protein kinase II.[Pubmed: 23960096 ]Mol Cancer Ther. 2013 Oct;12(10):2067-77.Liver cancer is the third leading cause of cancer deaths worldwide but no effective treatment toward liver cancer is available so far. Therefore, there is an unmet medical need to identify novel therapies to efficiently treat liver cancer and improve the prognosis of this disease.
|
| Kinase Assay | CaMKII γ, a critical regulator of CML stem/progenitor cells, is a target of the natural product berbamine.CaMKII γ, a critical regulator of CML stem/progenitor cells, is a target of the natural product berbamine.[Pubmed: 23074277]Blood. 2012 Dec 6;120(24):4829-39.Bcr-Abl tyrosine kinase inhibitors (TKIs) have been a remarkable success for the treatment of Ph(+) chronic myeloid leukemia (CML). However, a significant proportion of patients treated with TKIs develop resistance because of leukemia stem cells (LSCs) and T315I mutant Bcr-Abl.
|
| Structure Identification | Rapid Commun Mass Spectrom. 2014 Jan 15;28(1):143-7.Electrospray ionization mass spectrometry probing of binding affinity of berbamine, a flexible cyclic alkaloid from traditional Chinese medicine, with G-quadruplex DNA.[Pubmed: 24285399]Classic G-quadruplex binders typically have a large aromatic core and interact with G-quadruplexes through π-π stacking with the quartets. There are rather few reports on natural flexible cyclic molecule from traditional Chinese medicine which has high binding affinity with G-quadruplex.
|

Berbamine Dilution Calculator

Berbamine Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.6428 mL | 8.2142 mL | 16.4285 mL | 32.8569 mL | 41.0711 mL |
| 5 mM | 0.3286 mL | 1.6428 mL | 3.2857 mL | 6.5714 mL | 8.2142 mL |
| 10 mM | 0.1643 mL | 0.8214 mL | 1.6428 mL | 3.2857 mL | 4.1071 mL |
| 50 mM | 0.0329 mL | 0.1643 mL | 0.3286 mL | 0.6571 mL | 0.8214 mL |
| 100 mM | 0.0164 mL | 0.0821 mL | 0.1643 mL | 0.3286 mL | 0.4107 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Rhein
Catalog No.:BCN5947
CAS No.:478-43-3
- Droserone
Catalog No.:BCN7985
CAS No.:478-40-0
- Eleutherin
Catalog No.:BCN8475
CAS No.:478-36-4
- Pseudoaspidin
Catalog No.:BCN6386
CAS No.:478-28-4
- Lucidin
Catalog No.:BCC1709
CAS No.:478-08-0
- Nobiletin
Catalog No.:BCN5542
CAS No.:478-01-3
- Neostenine
Catalog No.:BCN5541
CAS No.:477953-07-4
- 3-(2-Glucosyloxy-4-methoxyphenyl)propanoic acid
Catalog No.:BCN7068
CAS No.:477873-63-5
- PIM-1 Inhibitor 2
Catalog No.:BCC2446
CAS No.:477845-12-8
- Tofacitinib (CP-690550,Tasocitinib)
Catalog No.:BCC2192
CAS No.:477600-75-2
- PHA-665752
Catalog No.:BCC1181
CAS No.:477575-56-7
- Musellarin A
Catalog No.:BCN7186
CAS No.:477565-36-9
- Pseudococaine
Catalog No.:BCN1902
CAS No.:478-73-9
- PHA 543613 hydrochloride
Catalog No.:BCC5972
CAS No.:478149-53-0
- Isoerysenegalensein E
Catalog No.:BCN3978
CAS No.:478158-77-9
- R-1479
Catalog No.:BCC1878
CAS No.:478182-28-4
- Gabapentin enacarbil
Catalog No.:BCC4239
CAS No.:478296-72-9
- 4-Hydroxymethylphenol 1-O-rhamnoside
Catalog No.:BCN7750
CAS No.:478314-67-9
- ISO-1
Catalog No.:BCC5427
CAS No.:478336-92-4
- CP 339818 hydrochloride
Catalog No.:BCC7048
CAS No.:478341-55-8
- Kisspeptin 10 (rat)
Catalog No.:BCC6132
CAS No.:478507-53-8
- Zopfiellamide A
Catalog No.:BCN1865
CAS No.:478945-64-1
- Angiotensin 1/2 (1-6)
Catalog No.:BCC1036
CAS No.:47896-63-9
- Calcium-Sensing Receptor Antagonists I
Catalog No.:BCC1448
CAS No.:478963-79-0
CaMKII gamma, a critical regulator of CML stem/progenitor cells, is a target of the natural product berbamine.[Pubmed:23074277]
Blood. 2012 Dec 6;120(24):4829-39.
Bcr-Abl tyrosine kinase inhibitors (TKIs) have been a remarkable success for the treatment of Ph(+) chronic myeloid leukemia (CML). However, a significant proportion of patients treated with TKIs develop resistance because of leukemia stem cells (LSCs) and T315I mutant Bcr-Abl. Here we describe the unknown activity of the natural product Berbamine that efficiently eradicates LSCs and T315I mutant Bcr-Abl clones. Unexpectedly, we identify CaMKII gamma as a specific and critical target of Berbamine for its antileukemia activity. Berbamine specifically binds to the ATP-binding pocket of CaMKII gamma, inhibits its phosphorylation and triggers apoptosis of leukemia cells. More importantly, CaMKII gamma is highly activated in LSCs but not in normal hematopoietic stem cells and coactivates LSC-related beta-catenin and Stat3 signaling networks. The identification of CaMKII gamma as a specific target of Berbamine and as a critical molecular switch regulating multiple LSC-related signaling pathways can explain the unique antileukemia activity of Berbamine. These findings also suggest that Berbamine may be the first ATP-competitive inhibitor of CaMKII gamma, and potentially, can serve as a new type of molecular targeted agent through inhibition of the CaMKII gamma activity for treatment of leukemia.
Electrospray ionization mass spectrometry probing of binding affinity of berbamine, a flexible cyclic alkaloid from traditional Chinese medicine, with G-quadruplex DNA.[Pubmed:24285399]
Rapid Commun Mass Spectrom. 2014 Jan 15;28(1):143-7.
RATIONALE: Classic G-quadruplex binders typically have a large aromatic core and interact with G-quadruplexes through pi-pi stacking with the quartets. There are rather few reports on natural flexible cyclic molecule from traditional Chinese medicine which has high binding affinity with G-quadruplex. METHODS: Electrospray ionization mass spectrometry (ESI-MS) experiments were performed to evaluate the binding affinities of a natural alkaloid, Berbamine, with seven G-quadruplexes. Furthermore, we utilized autodock4 analysis to uncover the binding mode of Berbamine with the G-quadruplex. RESULTS: ESI-MS experiments showed that Berbamine has the best binding affinity toward the (GGA)8 G-quadruplex compared with the other six G-quadruplexes. Autodock4 analysis indicated that Berbamine interacted with the (GGA)8 G-quadruplex through hydrogen bonding and van der Waals forces with a binding site at the lateral groove formed by DG8-DA9-DA15-DG16. CONCLUSIONS: In this study, we discovered a novel G-quadruplex binder, Berbamine, which has high binding affinity toward the (GGA)8 G-quadruplex. This study provided important clues regarding the probing of small molecule from traditional Chinese medicine which can target the G-quadruplex with high affinity.
Berbamine enhances the antineoplastic activity of gemcitabine in pancreatic cancer cells by activating transforming growth factor-beta/Smad signaling.[Pubmed:24619961]
Anat Rec (Hoboken). 2014 May;297(5):802-9.
Drug-resistance to gemcitabine chemotherapy in pancreatic cancer is still an unsolved problem. Combinations of other chemotherapy drugs with gemcitabine have been shown to increase the efficacy of gemcitabine-based treatment. In this study, the effect of Berbamine on the antitumor activity of gemcitabine was evaluated in human pancreatic cancer cell lines Bxpc-3 and Panc-1, and the underlying mechanisms were explored. Our results demonstrated that Berbamine exhibited a time- and dose-dependent inhibitory effect in the pancreatic cancer cell lines. Berbamine enhanced gemcitabine-induced cell growth inhibition and apoptosis in these cells. Combined treatment of Berbamine and gemcitabine resulted in down-regulation of anti-apoptotic proteins (Bcl-2, Bcl-xL) and up-regulation of pro-apoptotic proteins (Bax, Bid). More importantly, Berbamine treatment in combination with gemcitabine activated the transforming growth factor-beta/Smad (TGF-beta/Smad) signaling pathway, as a result of a decrease in Smad7 and an increase in transforming growth factor-beta receptor II (TbetaRII) expression. Changes in downstream targets of Smad7, such as up-regulation of p21 and down-regulation of c-Myc and Cyclin D1 were also observed. Therefore, Berbamine could enhance the antitumor activity of gemcitabine by inhibiting cell growth and inducing apoptosis, possibly through the regulation of the expression of apoptosis-related proteins and the activation of TGF-beta/Smad signaling pathway. Our study indicates that Berbamine may be a promising candidate to be used in combination with gemcitabine for pancreatic cancer treatment.
Berbamine protects the heart from ischemia/reperfusion injury by maintaining cytosolic Ca(2+) homeostasis and preventing calpain activation.[Pubmed:22664727]
Circ J. 2012;76(8):1993-2002. Epub 2012 May 15.
BACKGROUND: Berbamine, a natural compound from Barberry, was reported to protect myocardium from ischemia/reperfusion (I/R) injury, but the underlying mechanisms are largely unknown. METHODS AND RESULTS: Berbamine pretreatment from 10 to 100nmol/L concentration-dependently improved post-ischemic myocardial function. Similar protection was confirmed in isolated cardiomyocytes characterized by the attenuation of I/R-induced intracellular free Ca(2+) concentration ([Ca(2+)](i)) overloading and the depression of cell shortening and Ca(2+) transients, which were partially mimicked but not augmented by calpain inhibitor calpeptin and abolished by mitochondrial ATP-sensitive potassium (mitoK(ATP) channel inhibitor 5-hydroxydecanoate (5-HD) and phosphoinositide 3-kinase (PI3K) inhibitor wortmannin. Consistently, I/R-induced increase of calpain activity and decrease of sarcoplasmic reticulum Ca(2+) ATPase (SERCA2) activity; and protein expression of SERCA2a, desmin, calpastatin and Akt was significantly attenuated by Berbamine. In addition, I/R-decreased Akt protein was reversed by calpeptin. Moreover, Berbamine further increased I/R-enhanced phosphorylation of Akt and glycogen synthase kinase-3beta (GSK3beta). These protections were abolished by wortmannin. Furthermore, Berbamine significantly attenuated I/R-induced lactate dehydrogenase release, infarct size and contractile dysfunction, and such cardioprotective actions were abolished by wortmannin and 5-HD or mimicked by glycogen synthase kinase-3beta (GSK3beta) inhibitor SB216763 but without additive effect. CONCLUSIONS: These findings suggest that Berbamine confers cardioprotection against I/R injury by attenuating [Ca(2+)inf(i) overloading and preventing calpain activation through the activation of the PI3K-Akt-GSK3beta pathway and, subsequently, opening of the mitoK(ATP) channel.
Berbamine inhibits the growth of liver cancer cells and cancer-initiating cells by targeting Ca(2)(+)/calmodulin-dependent protein kinase II.[Pubmed:23960096]
Mol Cancer Ther. 2013 Oct;12(10):2067-77.
Liver cancer is the third leading cause of cancer deaths worldwide but no effective treatment toward liver cancer is available so far. Therefore, there is an unmet medical need to identify novel therapies to efficiently treat liver cancer and improve the prognosis of this disease. Here, we report that Berbamine and one of its derivatives, bbd24, potently suppressed liver cancer cell proliferation and induced cancer cell death by targeting Ca(2+)/calmodulin-dependent protein kinase II (CAMKII). Furthermore, Berbamine inhibited the in vivo tumorigenicity of liver cancer cells in NOD/SCID mice and downregulated the self-renewal abilities of liver cancer-initiating cells. Chemical inhibition or short hairpin RNA-mediated knockdown of CAMKII recapitulated the effects of Berbamine, whereas overexpression of CAMKII promoted cancer cell proliferation and increased the resistance of liver cancer cells to Berbamine treatments. Western blot analyses of human liver cancer specimens showed that CAMKII was hyperphosphorylated in liver tumors compared with the paired peritumor tissues, which supports a role of CAMKII in promoting human liver cancer progression and the potential clinical use of Berbamine for liver cancer therapies. Our data suggest that Berbamine and its derivatives are promising agents to suppress liver cancer growth by targeting CAMKII. Mol Cancer Ther; 12(10); 2067-77. (c)2013 AACR.


