Coronarin ACAS# 119188-33-9 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 119188-33-9 | SDF | Download SDF |
| PubChem ID | 24851535 | Appearance | Powder |
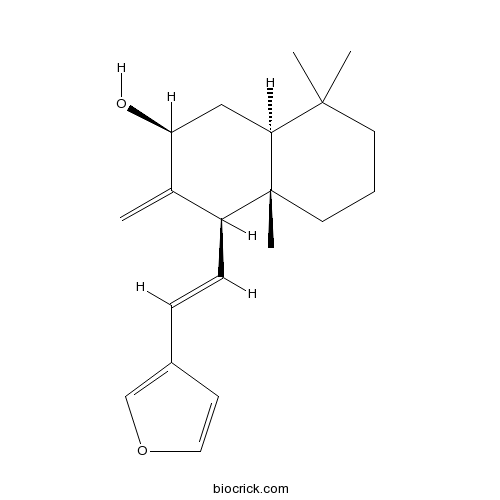
| Formula | C20H28O2 | M.Wt | 300.4 |
| Type of Compound | Diterpenoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | (2S,4R,4aS,8aS)-4-[(E)-2-(furan-3-yl)ethenyl]-4a,8,8-trimethyl-3-methylidene-2,4,5,6,7,8a-hexahydro-1H-naphthalen-2-ol | ||
| SMILES | CC1(CCCC2(C1CC(C(=C)C2C=CC3=COC=C3)O)C)C | ||
| Standard InChIKey | RHCBUXSXDFNUAG-UDMCIFMYSA-N | ||
| Standard InChI | InChI=1S/C20H28O2/c1-14-16(7-6-15-8-11-22-13-15)20(4)10-5-9-19(2,3)18(20)12-17(14)21/h6-8,11,13,16-18,21H,1,5,9-10,12H2,2-4H3/b7-6+/t16-,17-,18-,20+/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. Coronarin A exhibits good growth inhibition activities on HUVEC proliferation, it effectively suppresses the growth factor induced tube formation of HUVEC at the concentration of 10 microg/mL. 2. Coronarin, A, B, C, and D are cytotoxic prinicples from the rhizomes of Hedychium coronarium, Zingiberaceae. 3. Coronarin A has anti-inflammatory activity, it exhibits potent inhibition (IC50 <6.1708g/mL) against fMLP-induced superoxide production and elastase release. |
| Targets | Immunology & Inflammation related |

Coronarin A Dilution Calculator

Coronarin A Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.3289 mL | 16.6445 mL | 33.2889 mL | 66.5779 mL | 83.2224 mL |
| 5 mM | 0.6658 mL | 3.3289 mL | 6.6578 mL | 13.3156 mL | 16.6445 mL |
| 10 mM | 0.3329 mL | 1.6644 mL | 3.3289 mL | 6.6578 mL | 8.3222 mL |
| 50 mM | 0.0666 mL | 0.3329 mL | 0.6658 mL | 1.3316 mL | 1.6644 mL |
| 100 mM | 0.0333 mL | 0.1664 mL | 0.3329 mL | 0.6658 mL | 0.8322 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Glychionide A
Catalog No.:BCN3250
CAS No.:119152-50-0
- 3-Oxosapriparaquinone
Catalog No.:BCN3153
CAS No.:119139-56-9
- B-Raf inhibitor 1 dihydrochloride
Catalog No.:BCC4183
CAS No.:1191385-19-9
- Ganomycin I
Catalog No.:BCN3504
CAS No.:1191255-15-8
- IOWH-032
Catalog No.:BCC3922
CAS No.:1191252-49-9
- 1-Acetoxy-5-deacetylbaccatin I
Catalog No.:BCN6357
CAS No.:119120-27-3
- Linolenic acid ethyl ester
Catalog No.:BCN8333
CAS No.:1191-41-9
- Phellolactone
Catalog No.:BCN3467
CAS No.:1190897-23-4
- Fluorobexarotene
Catalog No.:BCC6110
CAS No.:1190848-23-7
- Fmoc-Asp-OH
Catalog No.:BCC3085
CAS No.:119062-05-4
- Euchrenone B1
Catalog No.:BCN3575
CAS No.:119061-09-5
- MRT67307
Catalog No.:BCC1779
CAS No.:1190378-57-4
- Coronarin D
Catalog No.:BCN6076
CAS No.:119188-37-3
- Coronarin B
Catalog No.:BCN6077
CAS No.:119188-38-4
- 10-Hydroxyscandine
Catalog No.:BCN6078
CAS No.:119188-47-5
- CZC-25146
Catalog No.:BCC5371
CAS No.:1191911-26-8
- CZC 54252 hydrochloride
Catalog No.:BCC6218
CAS No.:1191911-27-9
- 4-(1H-1,2,4-Triazol-1-ylmethyl)aniline
Catalog No.:BCC8645
CAS No.:119192-10-8
- PHT-427
Catalog No.:BCC2554
CAS No.:1191951-57-1
- LX7101 HCL
Catalog No.:BCC6414
CAS No.:1192189-69-7
- Gomisin S
Catalog No.:BCN3622
CAS No.:119239-49-5
- Avibactam
Catalog No.:BCC1384
CAS No.:1192500-31-4
- Scutellarin methylester
Catalog No.:BCN2828
CAS No.:119262-68-9
- PE 154
Catalog No.:BCC7858
CAS No.:1192750-33-6
A study on the synthesis of antiangiogenic (+)-coronarin A and congeners from (+)-sclareolide.[Pubmed:12781184]
Bioorg Med Chem Lett. 2003 Jun 16;13(12):2009-12.
Coronarin A 1, epi-Coronarin A 2 and some synthetic intermediates 14a and 14b synthesized from sclareolide exhibit good growth inhibition activities on HUVEC proliferation. In particular, Coronarin A 1 and epi-Coronarin A 2 effectively suppressed the growth factor induced tube formation of HUVEC at the concentration of 10 micro g/mL.
Labdane-type diterpenoids from the rhizomes of Hedychium coronarium inhibit lipopolysaccharide-stimulated production of pro-inflammatory cytokines in bone marrow-derived dendritic cells.[Pubmed:22293485]
Chem Pharm Bull (Tokyo). 2012;60(2):246-50.
The rhizomes of Hedychium coronarium have been used for the treatment of inflammation, skin diseases, headache, and sharp pain due to rheumatism in traditional medicine. From this plant, two new labdanes, 15-methoxylabda-8(17),11E,13-trien-16,15-olide (1) and 16-methoxylabda-8(17),11E,13-trien-15,16-olide (3), named hedycoronens A and B, as well as four known, labda-8(17),11,13-trien-16,15-olide (2), 16-hydroxylabda-8(17),11,13-trien-15,16-olide (4), Coronarin A (5), and corronarin E (6) were isolated. Their chemical structures were elucidated by mass, 1D- and 2D-nuclear magnetic resonance (NMR) spectroscopy. They were evaluated for inhibitory effects on the lipopolysaccharide (LPS)-stimulated production of pro-inflammatory cytokines in bone marrow-derived dendritic cells. Among of them, compounds 1-3 were potent inhibitors of LPS-stimulated interleukin-6 (IL-6) and IL-12 p40, with IC(50) ranging from 4.1+/-0.2 to 9.1+/-0.3 muM. Compounds 1 and 3 showed moderate inhibitory activity on the tumor necrosis factor-alpha (TNF-alpha) production with IC(50) values of 46.0+/-1.3 and 12.7+/-0.3 muM. The remains of compounds showed inactivity. These results warrant further studies concerning the potential anti-inflammatory benefits of labdane-diterpenes from H. coronarium.
Concise syntheses of coronarin A, coronarin E, austrochaparol and pacovatinin A.[Pubmed:18310958]
Chem Pharm Bull (Tokyo). 2008 Mar;56(3):398-403.
Total syntheses of (+)-Coronarin A (1), (+)-coronarin E (2), (+)-austrochaparol (3) and (+)-pacovatinin A (4) were achieved from the synthetic (+)-albicanyl acetate (6). Dess-Martin oxidation of (+)-albicanol (5) derived from the chemoenzymatic product (6) gave an aldehyde (7), which was subjected to Julia one-pot olefination using beta-furylmethyl-heteroaromatic sulfones (8 or 9 ) gave (+)-trans coronarin E (2) and (+)-cis coronarin E (12) with high cis-selectivity. The synthesis of (+)-Coronarin A (1) from (+)-trans coronarin E (2) was achiev-ed, while (+)-cis coronarin E (12) was converted to the natural products (+)-(5S,9S,10S)-15,16-epoxy-8(17),13(16),14-labdatriene (13) and (+)-austrochaparol (3). By the asymmetric synthesis of (+)-3, the absolute structure of (+)-3 was determined to be 5S, 7R, 9R, 10S configurations. Homologation of (+)-albicanol (5) followed by allylic oxidation gave (7 alpha)-hydroxy nitrile (17), which was finally converted to the natural (+)-pacovatinin A (4) in 8 steps from (+)-albicanol (5).
Labdane-type diterpenes from Hedychium gardnerianum with potent cytotoxicity against human small cell lung cancer cells.[Pubmed:19960422]
Phytother Res. 2010 Jul;24(7):1009-13.
Seven labdane-type diterpenes, coronarin E, Coronarin A, yunnanCoronarin A, yunnancoronarin B, hedyforrestin B, villosin, and hedyforrestin C were isolated from the rhizome of Hedychium gardnerianum and evaluated for cytotoxic activity against human small cell lung cancer (NCI-H187) and non-cancerous Vero cells. The results showed that villosin exhibited potent cytotoxic activity with IC(50) of 0.40 microM, which was higher than that of the drug ellipticine (IC(50) 1.79 microM). Moreover, ellipticine was very toxic to Vero cells (IC(50) 7.47 microM) whereas the toxicity of villosin was undetectable at concentration lower than 166.42 microM. The results have indicated that the lactone ring is essential for high cytotoxic activity and that the presence of a hydroxyl group at the 6 or 7 position causes decrease in activity. The very high cytotoxicity against the NCI-H187 cells and the exceptionally high selectivity index (>416) of villosin suggested that this compound may be used as a potential lead molecule for antitumor therapeutic development.


