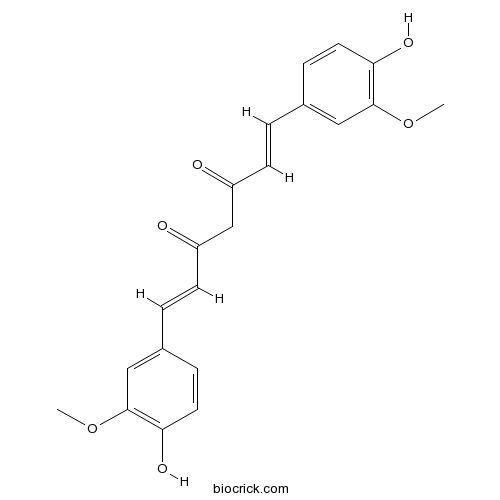
CurcuminTyrosinase inhibitor CAS# 458-37-7 |
- Celastrol
Catalog No.:BCN5986
CAS No.:34157-83-0
- Honokiol
Catalog No.:BCN1001
CAS No.:35354-74-6
- Deoxyarbutin
Catalog No.:BCC4774
CAS No.:53936-56-4
- EUK 134
Catalog No.:BCC4317
CAS No.:81065-76-1
- Edaravone
Catalog No.:BCC2480
CAS No.:89-25-8
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 458-37-7 | SDF | Download SDF |
| PubChem ID | 969516 | Appearance | Orange-yellow powder |
| Formula | C21H20O6 | M.Wt | 368.4 |
| Type of Compound | Phenols | Storage | Desiccate at -20°C |
| Synonyms | Diferuloylmethane | ||
| Solubility | DMSO : ≥ 100 mg/mL (271.46 mM) H2O : < 0.1 mg/mL (insoluble) *"≥" means soluble, but saturation unknown. | ||
| Chemical Name | (1E,6E)-1,7-bis(4-hydroxy-3-methoxyphenyl)hepta-1,6-diene-3,5-dione | ||
| SMILES | COC1=C(C=CC(=C1)C=CC(=O)CC(=O)C=CC2=CC(=C(C=C2)O)OC)O | ||
| Standard InChIKey | VFLDPWHFBUODDF-FCXRPNKRSA-N | ||
| Standard InChI | InChI=1S/C21H20O6/c1-26-20-11-14(5-9-18(20)24)3-7-16(22)13-17(23)8-4-15-6-10-19(25)21(12-15)27-2/h3-12,24-25H,13H2,1-2H3/b7-3+,8-4+ | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Curcumin is a natural phenolic compound with diverse pharmacologic effects including antitumour, anti-bacteria, anti-fungicidal ,anti-edemic, hepatoprotective, anti-inflammatory, antioxidant, antiproliferative and antiangiogenic activities. Curcumin is an inhibitor of p300 histone acetylatransferase ((HATs)) and also shows inhibitory effects on NF-κB and MAPKs.Curcumin application causes markedly fast wound closure with well-formed granulation tissue dominated by fibroblast proliferation, collagen deposition, and complete early regenerated epithelial layer. |
| Targets | DNA Methyltransferase | TNF-α | IFN-γ | IL Receptor | Nrf2 | HO-1 | GLUT | Caspase | STAT | NF-kB | HDAC | VEGFR | TGF-β/Smad |
| In vitro | Curcumin induces oxidation-dependent cell cycle arrest mediated by SIRT7 inhibition of rDNA transcription in human aortic smooth muscle cells.[Pubmed: 25644192]Toxicol Lett. 2015 Mar 18;233(3):227-38.It is widely accepted that abnormal accumulation of vascular smooth muscle cells (VSMCs) may promote atherosclerosis and post-angioplasty restenosis. The use of some plant polyphenols with potent antiproliferative activities may be considered as a therapeutic intervention to diminish/prevent the development of cardiovascular pathologies.
Potential anticancer properties and mechanisms of action of curcumin.[Pubmed: 25667441]Anticancer Res. 2015 Feb;35(2):645-51.Curcumin, a yellow substance belonging to the polyphenols superfamily, is the active component of turmeric, a common Indian spice, which is derived from the dried rhizome of the Curcuma longa plant. Numerous studies have demonstrated that Curcumin possesses anti-oxidant, anti-inflammatory and anticancerous properties.
The purpose of this review is to focus on the anti-tumor effects of Curcumin.
|
| In vivo | Curcumin enhances recovery of pancreatic islets from cellular stress induced inflammation and apoptosis in diabetic rats.[Pubmed: 25541178]Toxicol Appl Pharmacol. 2015 Feb 1;282(3):297-310.The phytochemical, Curcumin, has been reported to play many beneficial roles. However, under diabetic conditions, the detail mechanism of its beneficial action in the glucose homeostasis regulatory organ, pancreas, is poorly understood.
|
| Animal Research | Curcumin-induced angiogenesis hastens wound healing in diabetic rats.[Pubmed: 25454972]J Surg Res. 2015 Feb;193(2):978-88.Neovasculogenesis, vital for wound healing, gets compromised in diabetics patients, which consequently delayed wound healing. Previous studies have shown Curcumin as both a stimulatory and an inhibitory agent in the neovasculogenesis process. So, present study was aimed to investigate the effects of Curcumin on wound healing in diabetic rats and to explore the expressions of the various factors involved in neovasculogenesis.
|

Curcumin Dilution Calculator

Curcumin Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.7144 mL | 13.5722 mL | 27.1444 mL | 54.2888 mL | 67.861 mL |
| 5 mM | 0.5429 mL | 2.7144 mL | 5.4289 mL | 10.8578 mL | 13.5722 mL |
| 10 mM | 0.2714 mL | 1.3572 mL | 2.7144 mL | 5.4289 mL | 6.7861 mL |
| 50 mM | 0.0543 mL | 0.2714 mL | 0.5429 mL | 1.0858 mL | 1.3572 mL |
| 100 mM | 0.0271 mL | 0.1357 mL | 0.2714 mL | 0.5429 mL | 0.6786 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Curcumin is an inhibitor of tyrosinase with IC50 value of 47uM [1].
Curcumin is a natural compound with potency of anticancer. It is currently under clinical investigation for cancer chemoprevention. There is a variety of biochemical mechanisms of this anticancer function. The targets of Curcumin are involved in signaling pathways include transcription factors, growth factors,
inflammatory cytokines, receptors, and enzymes. Phase I trials have tested the toxicity and tolerability of Curcumin and found that Curcumin has no toxicities at doses up to 12 g/day. However, the bioavailability of Curcumin is quite poor. It is about ~1% after oral administration in phase I/II clinical trials and this hinders Curcumin‘s use in the clinic [2].
It is also reported that Curcumin reduces the generation of amloid beta (Aβ 40 and Aβ42) in SH-SY5Y neuroblastoma cells. It also down-regulates the expression of PS1 and GSK-3β in cells. All these cause the inhibition of Aβ formation and make Curcumin to be a potent therapeutic agent in AD [3].
References:
[1] Sachiko Shirota, Kouji Miyazaki, Ritsuo Aiyama, Minoru Ichioka and Teruo Yokokura. Tyrosinase inhibitors from crude drugs. Biol. Pharm. Bull. 1994, 17 (2): 266-269.
[2] Wungki Park, A.R.M Ruhul Amin, Zhuo Georgia Chen, and Dong M. Shin. New perspectives of curcumin in cancer prevention. Cancer Prev Res (Phila). 2013, 6(5): 387–400.
[3] Zhang Xiong, Zhang Hongmei, SiLu, LiYu. Curcumin mediates presenilin-1 activity to reduce β-amyloid production in a model of Alzheimer’s disease. Pharmacological Reports. 2013, 63: 1101-1108.
-
Scutebarbatine L
Catalog No.:BCN8369
CAS No.:960302-91-4
- BMS 470539 dihydrochloride
Catalog No.:BCC7850
CAS No.:457893-92-4
- PA 452
Catalog No.:BCC8005
CAS No.:457657-34-0
- Pyridone 6
Catalog No.:BCC1874
CAS No.:457081-03-7
- 1alpha, 24, 25-Trihydroxy VD2
Catalog No.:BCC1298
CAS No.:457048-34-9
- 5-O-Methylgenistein
Catalog No.:BCN7714
CAS No.:4569-98-6
- 3-Benzyl-5-(2-hydroxyethyl)-4-methylthiazolium chloride
Catalog No.:BCC8625
CAS No.:4568-71-2
- Neocnidilide
Catalog No.:BCN8174
CAS No.:4567-33-3
- H-Glu(OBzl)-OBzl.HCl
Catalog No.:BCC2927
CAS No.:4561-10-8
- Zaurategrast
Catalog No.:BCC2070
CAS No.:455264-31-0
- Isovouacapenol C
Catalog No.:BCN6557
CAS No.:455255-15-9
- 4-Benzoyl-3-methyl-1-phenyl-5-pyrazolone
Catalog No.:BCC8695
CAS No.:4551-69-3
- Ilicic acid
Catalog No.:BCN5505
CAS No.:4586-68-9
- Boc-Asn-ONp
Catalog No.:BCC3072
CAS No.:4587-33-1
- SW033291
Catalog No.:BCC3981
CAS No.:459147-39-8
- JNJ-7777120
Catalog No.:BCC4543
CAS No.:459168-41-3
- SB 452533
Catalog No.:BCC7620
CAS No.:459429-39-1
- Obeticholic Acid
Catalog No.:BCC5572
CAS No.:459789-99-2
- PEAQX
Catalog No.:BCC5495
CAS No.:459836-30-7
- 5-[(2R)-2-Aminopropyl]-2,3-dihydro-1-[3-(phenylmethoxy)propyl]-1H-indole-7-carbonitrile
Catalog No.:BCN1438
CAS No.:459868-73-6
- Rucaparib (AG-014699,PF-01367338)
Catalog No.:BCC2207
CAS No.:459868-92-9
- Interiotherin C
Catalog No.:BCN3636
CAS No.:460090-65-7
- Larixyl acetate
Catalog No.:BCC8195
CAS No.:4608-49-5
- Eact
Catalog No.:BCC6313
CAS No.:461000-66-8
Potential anticancer properties and mechanisms of action of curcumin.[Pubmed:25667441]
Anticancer Res. 2015 Feb;35(2):645-51.
Curcumin, a yellow substance belonging to the polyphenols superfamily, is the active component of turmeric, a common Indian spice, which is derived from the dried rhizome of the Curcuma longa plant. Numerous studies have demonstrated that Curcumin possesses anti-oxidant, anti-inflammatory and anticancerous properties. The purpose of this review is to focus on the anti-tumor effects of Curcumin. Curcumin inhibits the STAT3 and NF-kappaB signaling pathways, which play key-roles in cancer development and progression. Also, inhibition of Sp-1 and its housekeeping gene expressions may serve as an important hypothesis to prevent cancer formation, migration, and invasion. Recent data have suggested that Curcumin may act by suppressing the Sp-1 activation and its downstream genes, including ADEM10, calmodulin, EPHB2, HDAC4, and SEPP1 in a concentration-dependent manner in colorectal cancer cell lines; these results are consistent with other studies, which have reported that Curcumin could suppress the Sp-1 activity in bladder cancer and could decrease DNA binding activity of Sp-1 in non-small cell lung carcinoma cells. Recent data advocate that ER stress and autophagy may as well play a role in the apoptosis process, which is induced by the Curcumin analogue B19 in an epithelial ovarian tumor cell line and that autophagy inhibition could increase Curcumin analogue-induced apoptosis by inducing severe ER stress. The ability of Curcumin to induce apoptosis in tumor cells and its anti-angiogenic potential will be discussed in this review.
Curcumin enhances recovery of pancreatic islets from cellular stress induced inflammation and apoptosis in diabetic rats.[Pubmed:25541178]
Toxicol Appl Pharmacol. 2015 Feb 1;282(3):297-310.
The phytochemical, Curcumin, has been reported to play many beneficial roles. However, under diabetic conditions, the detail mechanism of its beneficial action in the glucose homeostasis regulatory organ, pancreas, is poorly understood. The present study has been designed and carried out to explore the role of Curcumin in the pancreatic tissue of STZ induced and cellular stress mediated diabetes in eight weeks old male Wistar rats. Diabetes was induced with a single intraperitoneal dose of STZ (65 mg/kg body weight). Post to diabetes induction, animals were treated with Curcumin at a dose of 100 mg/kg body weight for eight weeks. Underlying molecular and cellular mechanism was determined using various biochemical assays, DNA fragmentation, FACS, histology, immunoblotting and ELISA. Treatment with Curcumin reduced blood glucose level, increased plasma insulin and mitigated oxidative stress related markers. In vivo and in vitro experimental results revealed increased levels of proinflammatory cytokines (TNF-alpha, IL1-beta and IFN-gamma), reduced level of cellular defense proteins (Nrf-2 and HO-1) and glucose transporter (GLUT-2) along with enhanced levels of signaling molecules of ER stress dependent and independent apoptosis (cleaved Caspase-12/9/8/3) in STZ administered group. Treatment with Curcumin ameliorated all the adverse changes and helps the organ back to its normal physiology. Results suggest that Curcumin protects pancreatic beta-cells by attenuating inflammatory responses, and inhibiting ER/mitochondrial dependent and independent pathways of apoptosis and crosstalk between them. This uniqueness and absence of any detectable adverse effect proposes the possibility of using this molecule as an effective protector in the cellular stress mediated diabetes mellitus.
Curcumin induces oxidation-dependent cell cycle arrest mediated by SIRT7 inhibition of rDNA transcription in human aortic smooth muscle cells.[Pubmed:25644192]
Toxicol Lett. 2015 Mar 18;233(3):227-38.
It is widely accepted that abnormal accumulation of vascular smooth muscle cells (VSMCs) may promote atherosclerosis and post-angioplasty restenosis. The use of some plant polyphenols with potent antiproliferative activities may be considered as a therapeutic intervention to diminish/prevent the development of cardiovascular pathologies. In the present study, VSMC response to Curcumin treatment was evaluated. 5 muM Curcumin elicited a cytostatic effect, which was accompanied by protein carbonylation, oxidative DNA damage and changes in the nucleolar activity (the size and number of nucleoli, nucleolar protein levels and their localization). The levels of p53 and p21 were elevated. However, this was independent of DNA DSBs. Curcumin caused inhibition of rDNA transcription, which could be due to SIRT7 downregulation, site-specific methylation of RNA18S5 gene promoter or both. Curcumin-induced DNA methyltransferase 2 (DNMT2) upregulation was also shown. DNMT2-mediated RNA methylation could promote RNA stabilization upon Curcumin treatment. In conclusion, a nucleolus-focused cytostatic action of Curcumin at a low micromolar concentration range, which could be feasibly achieved through dietary means, was established in VSMCs and we propose a novel mechanism underlying this action. We believe that our results may contribute to better understanding of the biological and pharmacological effects of Curcumin on the human cardiovascular system.
Curcumin-induced angiogenesis hastens wound healing in diabetic rats.[Pubmed:25454972]
J Surg Res. 2015 Feb;193(2):978-88.
BACKGROUND: Neovasculogenesis, vital for wound healing, gets compromised in diabetics patients, which consequently delayed wound healing. Previous studies have shown Curcumin as both a stimulatory and an inhibitory agent in the neovasculogenesis process. So, present study was aimed to investigate the effects of Curcumin on wound healing in diabetic rats and to explore the expressions of the various factors involved in neovasculogenesis. MATERIALS AND METHODS: Open excisional diabetic wound was created in sixty rats and divided into three groups viz. i) control, ii) pluronic gel-treated, and iii) Curcumin-treated. The pluronic F-127 gel (25%) and Curcumin (0.3%) in the pluronic gel were topically applied once daily for 19 d. The wound healing and neovasculogenesis among these groups were evaluated by gross appearance of wounds and microscopically by hematoxylin and eosin staining, immunohistochemistry for CD31, messenger RNA expressions of vascular endothelial growth factor (VEGF), transforming growth factor (TGF)-beta1, hypoxia-inducible growth factor-1 alpha, stromal cell-derived growth factor-1 alpha, and heme oxygenase-1, and Western blotting studies of VEGF and TGF-beta1 in granulation and/or healing tissue on days 3, 7, 14, and 19. RESULTS: Curcumin application caused markedly fast wound closure with well-formed granulation tissue dominated by fibroblast proliferation, collagen deposition, and complete early regenerated epithelial layer. Immunohistochemistry for CD31 revealed well-formed blood vessels with increased microvessel density on days 3, 7, and 14 in the Curcumin-treated group. Expressions of VEGF and TGF-beta1 on days 3, 7, and 14, hypoxia-inducible growth factor-1 alpha, stromal cell-derived growth factor-1 alpha, and heme oxygenase-1 on days 3 and 7 were increased in Curcumin-treated diabetic rats, as compared with other groups. CONCLUSIONS: Curcumin enhanced the neovasculogenesis and accelerated the wound healing in diabetic rats by increased expressions of various factors.
Pharmacological basis for the role of curcumin in chronic diseases: an age-old spice with modern targets.[Pubmed:19110321]
Trends Pharmacol Sci. 2009 Feb;30(2):85-94.
Curcumin (diferuloylmethane), a yellow pigment in the spice turmeric (also called curry powder), has been used for centuries as a treatment for inflammatory diseases. Extensive research within the past two decades has shown that Curcumin mediates its anti-inflammatory effects through the downregulation of inflammatory transcription factors (such as nuclear factor kappaB), enzymes (such as cyclooxygenase 2 and 5 lipoxygenase) and cytokines (such as tumor necrosis factor, interleukin 1 and interleukin 6). Because of the crucial role of inflammation in most chronic diseases, the potential of Curcumin has been examined in neoplastic, neurological, cardiovascular, pulmonary and metabolic diseases. The pharmacodynamics and pharmacokinetics of Curcumin have been examined in animals and in humans. Various pharmacological aspects of Curcumin in vitro and in vivo are discussed in detail here.
Neuroprotective effects of curcumin.[Pubmed:17569212]
Adv Exp Med Biol. 2007;595:197-212.
Neurodegenerative diseases result in the loss of functional neurons and synapses. Although future stem cell therapies offer some hope, current treatments for most of these diseases are less than adequate and ourbest hope is to prevent these devastating diseases. Neuroprotective approaches work best prior to the initiation of damage, suggesting that some safe and effective prophylaxis would be highly desirable. Curcumin has an outstanding safety profile and a number of pleiotropic actions with potential for neuroprotective efficacy, including anti-inflammatory, antioxidant, and anti-protein-aggregate activities. These can be achieved at submicromolar levels. Curcumin's dose-response curves are strongly dose dependent and often biphasic so that in vitro data need to be cautiously interpreted; many effects might not be achievable in target tissues in vivo with oral dosing. However, despite concerns about poor oral bioavailability, Curcumin has at least 10 known neuroprotective actions and many of these might be realized in vivo. Indeed, accumulating cell culture and animal model data show that dietary Curcumin is a strong candidate for use in the prevention or treatment of major disabling age-related neurodegenerative diseases like Alzheimer's, Parkinson's, and stroke. Promising results have already led to ongoing pilot clinical trials.
Antitumor, anti-invasion, and antimetastatic effects of curcumin.[Pubmed:17569210]
Adv Exp Med Biol. 2007;595:173-84.
Curcumin was found to be cytotoxic in nature to a wide variety of tumor cell lines of different tissue origin. The action of Curcumin is dependent on with the cell type, the concentration of Curcumin (IC50: 2-40 microg/mL), and the time of the treatment. The major mechanism by which Curcumin induces cytotoxicity is the induction of apoptosis. Curcumin decreased the expression of antiapoptotic members of the Bcl-2 family and elevated the expression of p53, Bax, procaspases 3, 8, and 9. Curcumin prevents the entry of nuclear factor KB (NF-KB) into the nucleus there by decreasing the expression of cell cycle regulatory proteins and survival factors such as Bcl-2 and survivin. Curcumin arrested the cell cycle by preventing the expression of cyclin D1, cdk-1 and cdc-25. Curcumin inhibited the growth of transplantable tumors in different animal models and increased the life span of tumor-harboring animals. Curcumin inhibits metastasis of tumor cells as shown in in vitro as well as in vivo models, and the possible mechanism is the inhibition of matrix metalloproteases. Curcumin was found to suppress the expression of cyclooxygenase-2, vascular endothelial growth factor, and intercellular adhesion molecule- and elevated the expression of antimetastatic proteins, the tissue inhibitor of metalloproteases-2, nonmetastatic gene 23, and Ecadherin. These results indicate that Curcumin acts at various stages of tumor cell progression.
Molecular targets of curcumin.[Pubmed:17569214]
Adv Exp Med Biol. 2007;595:227-43.
Curcumin possesses anti-inflammatory activity and is a potent inhibitor of reactive-oxygen-generating enzymes such as lipoxygenase/cyclooxygenase, xanthine dehydrogenase/oxidase, and inducible nitric oxide synthase (iNOS); it is an effective inducer of heme oxygenase-1. Curcumin is also a potent inhibitor of protein kinase C (PKC), EGF-receptor tyrosine kinase, and IkappaB kinase. Subsequently, Curcumin inhibits the activation of NF-KB and the expressions of oncogenes including c-jun, c-fos, c-myc, NIK, MAPKs, ERK, ELK, PI3K, Akt, CDKs, and iNOS. It is considered that PKC, mTOR, and EGFR tyrosine kinase are the major upstream molecular targest for Curcumin intervention, whereas the nuclear oncogenes such as c-jun, c-fos, c-myc, CDKs, FAS, and iNOS might act as downstream molecular targets for Curcumin actions. It is proposed that Curcumin might suppress tumor promotion through blocking signal transduction pathways in the target cells. The oxidant tumor promoter TPA activates PKC by reacting with zinc thiolates present within the regulatory domain, whereas the oxidized form of cancer chemopreventive agent such as Curcumin can inactivate PKC by oxidizing the vicinal thiols present within the catalytic domain. Recent studies indicated that proteasome-mediated degradation of cell proteins play a pivotal role in the regulation of several basic cellular processes, including differentiation, proliferation, cell cycling, and apoptosis. It has been demonstrated that Curcumin-induced apoptosis is mediated through the impairment of the ubiquitin-proteasome pathway.
Cardioprotective effects of curcumin.[Pubmed:17569220]
Adv Exp Med Biol. 2007;595:359-77.
Curcumin, a major active component of turmeric, is extracted from the powdered dry rhizome of Curcuma longa Linn (Zingiberaceae) and it has been used for centuries in indigenous medicine. We have shown that Curcumin has a protective role against myocardial necrosis in rats. The antioxidant activity of Curcumin could be attributed to the phenolic and methoxy groups in conjunction with the 1,3-diketone-conjugated diene system, for scavenging of the oxygen radicals. In addition, Curcumin is shown to enhance the activities of detoxifying enzymes such as glutathione-S-transferase in vivo. We have also shown that oxygen free radicals exacerbate cardiac damage and Curcumin induces cardioprotective effect and it also inhibits free-radical generation in myocardial ischemia in rats. This chapter on the cardioprotective effects of Curcumin covers the following aspects: (1) the history of Curcumin and its discovery as a potent drug with relevance to cardiovascular diseases; (2) mechanistic role of Curcumin in vitro, emphasizing the antiplatelet and anticoagulant effects; (3) cardiovascular properties of Curcumin; (4) application of Curcumin in different animal models (viz. myocardial ischemia, myocardial infarction, cardiomyopathy, and arrhythmia in vitro and in vivo); (5) Curcumin free-radical scavenging activity, particularly against O2 radical and depletion of the oxidative stress.


