EpiaschantinCAS# 41689-50-3 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

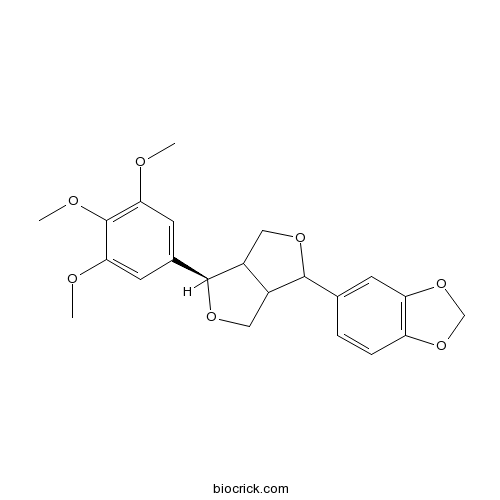
| Cas No. | 41689-50-3 | SDF | Download SDF |
| PubChem ID | 374872 | Appearance | Powder |
| Formula | C22H24O7 | M.Wt | 400.42 |
| Type of Compound | Lignans | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | 5-[(6R)-6-(3,4,5-trimethoxyphenyl)-1,3,3a,4,6,6a-hexahydrofuro[3,4-c]furan-3-yl]-1,3-benzodioxole | ||
| SMILES | COC1=CC(=CC(=C1OC)OC)C2C3COC(C3CO2)C4=CC5=C(C=C4)OCO5 | ||
| Standard InChIKey | ONDWGDNAFRAXCN-HMRZPLOASA-N | ||
| Standard InChI | InChI=1S/C22H24O7/c1-23-18-7-13(8-19(24-2)22(18)25-3)21-15-10-26-20(14(15)9-27-21)12-4-5-16-17(6-12)29-11-28-16/h4-8,14-15,20-21H,9-11H2,1-3H3/t14?,15?,20?,21-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. (+)-Epiaschantin shows marginal cancer cell line inhibitory activities. 2. (+)-Epiaschantin has anti-platelet aggregation activity. |

Epiaschantin Dilution Calculator

Epiaschantin Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.4974 mL | 12.4869 mL | 24.9738 mL | 49.9476 mL | 62.4344 mL |
| 5 mM | 0.4995 mL | 2.4974 mL | 4.9948 mL | 9.9895 mL | 12.4869 mL |
| 10 mM | 0.2497 mL | 1.2487 mL | 2.4974 mL | 4.9948 mL | 6.2434 mL |
| 50 mM | 0.0499 mL | 0.2497 mL | 0.4995 mL | 0.999 mL | 1.2487 mL |
| 100 mM | 0.025 mL | 0.1249 mL | 0.2497 mL | 0.4995 mL | 0.6243 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 8-Acetoxypentadeca-1,9Z-diene-4,6-diyn-3-ol
Catalog No.:BCN1444
CAS No.:41682-30-8
- Arctigenin 4'-O-beta-gentiobioside
Catalog No.:BCN2847
CAS No.:41682-24-0
- 8-Methyleugenitol
Catalog No.:BCN6459
CAS No.:41682-21-7
- 4',7-Dihydroxyflavanone
Catalog No.:BCC8333
CAS No.:41680-09-5
- 2H-1-Benzopyran-7-yloxy
Catalog No.:BCN3580
CAS No.:41680-08-4
- Catharanthine hemitartrate
Catalog No.:BCN8463
CAS No.:4168-17-6
- 7-O-Methyl morroniside
Catalog No.:BCN3882
CAS No.:41679-97-4
- Koaburaside
Catalog No.:BCN5475
CAS No.:41653-73-0
- Lacinilene C
Catalog No.:BCN5474
CAS No.:41653-72-9
- Ciclopirox ethanolamine
Catalog No.:BCC4372
CAS No.:41621-49-2
- Ylangenol
Catalog No.:BCN6705
CAS No.:41610-69-9
- (-)-Pinoresinol 4-O-glucoside
Catalog No.:BCN7251
CAS No.:41607-20-9
- Epimagnolin A
Catalog No.:BCN7831
CAS No.:41689-51-4
- Indicine N-oxide
Catalog No.:BCN1996
CAS No.:41708-76-3
- Croalbidine
Catalog No.:BCN2068
CAS No.:41714-30-1
- Bavachromene
Catalog No.:BCN3191
CAS No.:41743-38-8
- Procyanidin A2
Catalog No.:BCN6805
CAS No.:41743-41-3
- Luteone
Catalog No.:BCN5476
CAS No.:41743-56-0
- Sugeroside
Catalog No.:BCN6961
CAS No.:41743-57-1
- Pyrochamissanthin
Catalog No.:BCN3612
CAS No.:41743-60-6
- Irisflorentin
Catalog No.:BCN1278
CAS No.:41743-73-1
- (-)-Sativan
Catalog No.:BCN7752
CAS No.:41743-86-6
- Acuminatin
Catalog No.:BCN5477
CAS No.:41744-39-2
- Ginsenoside Rb1
Catalog No.:BCN1063
CAS No.:41753-43-9
Anti-platelet aggregation alkaloids and lignans from Hernandia nymphaeifolia.[Pubmed:10821052]
Planta Med. 2000 Apr;66(3):251-6.
A new aporphine, N-(N-methylcarbamoyl)-O-methyl-bulbocapnine (1), together with seven known compounds, (-)-5'-methoxypodorhizol (2), a mixture of beta-sitosterone (3) and stigmasta-4,22-dien-3-one (4), a mixture of 3 beta-hydroxystigmast-5-en-7-one (5) and 3 beta-hydroxystigmasta-5,22-dien-7-one (6), and a mixture of 6 alpha-hydroxystigmast-4-en-3-one (7) and 6 alpha-hydroxystigmasta-4,22-dien-3-one (8), were isolated in continuing studies on the trunk bark of Formosan Hernandia nymphaeifolia. The structures of these compounds were determined through spectral analyses. In addition, the previously reported six alkaloids, laurotetanine, oxohernagine, thalicarpine, reticuline, (+)-vateamine-2'-beta-N-oxide, (+)-hernandaline and six lignans, (+)-Epiaschantin, (+)-epimagnolin, (+)-epiyangambin, (-)-hernone, (-)-yatein, (-)-deoxypodophyllotoxin were demonstrated to have anti-platelet aggregation activity.
New dimeric aporphine alkaloids and cytotoxic constituents of Hernandia nymphaeifolia.[Pubmed:9000885]
Planta Med. 1996 Dec;62(6):528-33.
Three minor new dimeric aporphine alkaloids, oviisocorydine (1), ovihernangerine (2), and oxohernandaline (3), along with four known alkaloids, (+)-hernandaline, (+)-thallcarpine, (+)-N-methylovigerine, and N-methylcorydaldine, and five known lignans, (+)-epimagnolin, (+)-Epiaschantin, (+)-epiyangambin, (-)-deoxypodophyllotoxin, and (-)-yatein, have been additionally isolated from the trunk bark of Hernandia nymphaeifolia. The structures of these compounds were elucidated by spectroscopic methods. Among forty-four isolates obtained till now, nine compounds, hernandonine (4), hernanymphine (5), demethylsonodione (6), (+)-ovigerine (7), (+)-N-methylovigerine (8), N-formyldehydroovigerine (9), 4-methoxyoxohernandaline (10), (-)-deoxypodophyllotoxin (11), and (-)-yatein (12) showed significant cytotoxic activities (ED50 values < 1 microgram/ml) against P-388, KB16, A549, and HT-29 cell lines.
Antineoplastic agents. 522. Hernandia peltata (Malaysia) and Hernandia nymphaeifolia (Republic of Maldives).[Pubmed:14987061]
J Nat Prod. 2004 Feb;67(2):214-20.
Bioassay (P388 lymphocytic leukemia cell line and human tumor cell lines)-guided separation of the extracts prepared from the tropical and coastal trees Hernandia peltata (Malaysia) and Hernandianymphaeifolia (Republic of Maldives) led to the isolation of a new lignan designated as hernanol (1) and 12 previously known lignans: (-)-deoxypodophyllotoxin (2), deoxypicropodophyllin (3), (+)-Epiaschantin (4), (+)-epieudesmin (5), praderin (6), 5'-methoxyyatein (7), podorhizol (8), deoxypodorhizone (9), bursehernin (10), kusunokinol (11), clusin (12), and (-)-maculatin (13). The oxidative cyclization (with VOF(3)) of lignans 8, 9, and 10 resulted in a new and unusual benzopyran (14), isostegane (15), and a new dibenzocyclooctadiene lactone (16), respectively. The structure and relative stereochemistry of hernanol (1) and lignans 3, 7, 8, 9, 10, 11, and 12 were determined by 1D and 2DNMR and HRMS analyses. The structures and absolute stereochemistry of structures 2, 4, 5, 6, 13, 14, 15, and 16 were unequivocally determined by single-crystal X-ray diffraction analyses. Evaluation against the murine P388 lymphocytic leukemia cell line and human tumor cell lines showed podophyllotoxin derivatives 2 and 3 to be strong cancer cell line growth inhibitors and substances 4, 5, 8, and 15 to have marginal cancer cell line inhibitory activities. Seven of the lignans and one of the synthetic modifications (14) inhibited growth of the pathogenic bacterium Neisseria gonorrhoeae.


