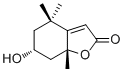
EpiloliolideCAS# 147730-52-7 |
Quality Control & MSDS
Package In Stock
Number of papers citing our products

| Cas No. | 147730-52-7 | SDF | File under preparation. |
| PubChem ID | N/A | Appearance | Oil |
| Formula | C11H16O3 | M.Wt | 196.27 |
| Type of Compound | Sesquiterpenoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Epiloliolide Dilution Calculator

Epiloliolide Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 5.095 mL | 25.4751 mL | 50.9502 mL | 101.9004 mL | 127.3756 mL |
| 5 mM | 1.019 mL | 5.095 mL | 10.19 mL | 20.3801 mL | 25.4751 mL |
| 10 mM | 0.5095 mL | 2.5475 mL | 5.095 mL | 10.19 mL | 12.7376 mL |
| 50 mM | 0.1019 mL | 0.5095 mL | 1.019 mL | 2.038 mL | 2.5475 mL |
| 100 mM | 0.051 mL | 0.2548 mL | 0.5095 mL | 1.019 mL | 1.2738 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Acutumidine
Catalog No.:BCX0300
CAS No.:18145-26-1
- Balsaminone B
Catalog No.:BCX0299
CAS No.:213271-56-8
- 2-Ethyl-3-methylmaleimide
Catalog No.:BCX0298
CAS No.:20189-42-8
- Isoorientin 3'-O-glucoside
Catalog No.:BCX0297
CAS No.:53098-95-6
- Trijugin G
Catalog No.:BCX0296
CAS No.:1233534-53-6
- Racemoflavone
Catalog No.:BCX0295
CAS No.:106055-12-3
- 24-O-Acetyllycoclavanol
Catalog No.:BCX0294
CAS No.:13956-53-1
- Trijugin H
Catalog No.:BCX0293
CAS No.:1233534-55-8
- 5α,6α-Epoxy-3β-hydroxymegastigm-7-en-9-one
Catalog No.:BCX0292
CAS No.:50281-42-0
- 5-Hydroxynoracronycine
Catalog No.:BCX0291
CAS No.:27067-70-5
- Zedoalactone A
Catalog No.:BCX0290
CAS No.:170384-82-4
- 5-Deoxytrijugin C
Catalog No.:BCX0289
CAS No.:135971-83-4
- Yunnancoronarin C
Catalog No.:BCX0302
CAS No.:260406-45-9
- Tanshinol B
Catalog No.:BCX0303
CAS No.:189290-30-0
- Spicatanol
Catalog No.:BCX0304
CAS No.:1158208-05-9
- (1S,16R)-1-Hydroxycryptotanshinone anhydride
Catalog No.:BCX0305
CAS No.:2301967-86-0
- Formononetin 7-O-β-D-apiofuranosyl-(1→6)-O-β-D-glucopyranoside
Catalog No.:BCX0306
CAS No.:857677-78-2
- Kushenol O
Catalog No.:BCX0307
CAS No.:102390-91-0
- Curzeone
Catalog No.:BCX0308
CAS No.:104068-56-6
- 16α-O-Methylneoquassin
Catalog No.:BCX0309
CAS No.:89498-94-2
- 16β-O-Ethylneoquassin
Catalog No.:BCX0310
CAS No.:343943-63-5
- 16α-O-Ethylneoquassin
Catalog No.:BCX0311
CAS No.:909272-59-9
- Methyl 2,4-dihydroxybenzoate
Catalog No.:BCX0312
CAS No.:2150-47-2
- Ethyl (10E,12E)-9-oxooctadeca-10,12-dienoate
Catalog No.:BCX0313
CAS No.:2552582-20-2
Regulation of p53 Activity by (+)-Epiloliolide Isolated from Ulva lactuca.[Pubmed:34436289]
Mar Drugs. 2021 Aug 5;19(8):450.
Ulva lactuca (U. lactuca) is a green alga distributed worldwide and used as a food and cosmetic material. In our previous study, we determined the effects of U. lactuca methanol extracts on the UVB-induced DNA repair. In the present study, we fractionated U. lactuca methanol extracts to identify the effective compound for the DNA repair. MTT assay demonstrated that (+)-Epiloliolide showed no cytotoxicity up to 100 muM in BJ-5ta human dermal fibroblast. Upon no treatment, exposure to UVB 400 J/m(2) decreased cell viability by 45%, whereas (+)-Epiloliolide treatment for 24 h after UVB exposure significantly increased the cell viability. In GO and GESA analysis, a number of differentially expressed genes were uniquely expressed in (+)-Epiloliolide treated cells, which were enriched in the p53 signaling pathway and excision repair. Immunofluorescence demonstrated that (+)-Epiloliolide increased the nuclear localization of p53. Comet assay demonstrated that (+)-Epiloliolide decreased tail moment increased by UVB. Western blot analysis demonstrated that (+)-Epiloliolide decreased the levels of p-p53, p21, Bax, and Bim, but increased that of Bcl-2. Reverse transcription PCR (RT-PCR) demonstrated that (+)-Epiloliolide decreased the levels of MMP 1, 9, and 13, but increased that of COL1A1. These results suggest that (+)-Epiloliolide regulates p53 activity and has protective effects against UVB.
The Marine-Derived Natural Product Epiloliolide Isolated from Sargassum horneri Regulates NLRP3 via PKA/CREB, Promoting Proliferation and Anti-Inflammatory Effects of Human Periodontal Ligament Cells.[Pubmed:34356813]
Mar Drugs. 2021 Jul 9;19(7):388.
Currently, periodontitis treatment relies on surgical operations, anti-inflammatory agents, or antibiotics. However, these treatments cause pain and side effects, resulting in a poor prognosis. Therefore, in this study, we evaluated the impact of the compound Epiloliolide isolated from Sargassum horneri on the recovery of inflammatory inhibitors and loss of periodontal ligaments, which are essential treatment strategies for periodontitis. Here, human periodontal ligament cells stimulated with PG-LPS were treated with the compound Epiloliolide, isolated from S. horneri. In the results of this study, Epiloliolide proved the anti-inflammatory effect, cell proliferation capacity, and differentiation potential of periodontal ligament cells into osteoblasts, through the regulation of the PKA/CREB signaling pathway. Epiloliolide effectively increased the proliferation and migration of human periodontal ligament cells without cytotoxicity and suppressed the protein expression of proinflammatory mediators and cytokines, such as iNOS, COX-2, TNF-alpha, IL-6, and IL-1beta, by downregulating NLRP3 activated by PG-LPS. Epiloliolide also upregulated the phosphorylation of PKA/CREB proteins, which play an important role in cell growth and proliferation. It was confirmed that the anti-inflammatory effect in PG-LPS-stimulated large cells was due to the regulation of PKA/CREB signaling. We suggest that Epiloliolide could serve as a potential novel therapeutic agent for periodontitis by inhibiting inflammation and restoring the loss of periodontal tissue.
Preparative separation and purification of loliolide and epiloliolide from Ascophyllum nodosum using amine-based microporous organic polymer for solid phase extraction coupled with macroporous resin and prep-HPLC.[Pubmed:33913944]
Anal Methods. 2021 Apr 28;13(16):1939-1944.
Herein, we reported a novel approach for the preparative separation and purification of loliolide and Epiloliolide from Ascophyllum nodosum. An amine-based microporous organic polymer (MOP) was used for the pretreatment of the nodosum extract via solid-phase extraction (SPE). The obtained extract was further purified using macroporous resin chromatography and preparative high-performance liquid chromatography (prep-HPLC). The loading and elution parameters of the MOP were evaluated using standard loliolide, and the optimized conditions were used during the SPE of the nodosum extract (37.5 g). After the pretreatment with MOP, the extract (2.79 g) was obtained and further purified using a D101 resin column followed by prep-HPLC. A pair of epimers were isolated and identified as loliolide (5.83 mg) and Epiloliolide (2.50 mg) using high-resolution electrospray ionization tandem mass spectrometry (HRESI-MS), 1D- and 2D-nuclear magnetic resonance (NMR) spectroscopy. This study demonstrates the potential of MOPs in the separation and purification of monoterpenoids from complex plant samples.
Neo-Aplysiatoxin A Isolated from Okinawan Cyanobacterium Moorea Producens.[Pubmed:31978978]
Molecules. 2020 Jan 22;25(3):457.
A new aplysiatoxin derivative, neo-aplysiatoxin A (1), along with seven known compounds, neo-debromoaplysiatoxin A (2), dolastatin 3 (3), lyngbic acid (4), malyngamide M (5), hermitamide A (6), (-)-loliolide (7), and (+)-Epiloliolide (8), was isolated from the Okinawan cyanobacterium Moorea producens. Their structures were elucidated on the basis of spectroscopic data, including high-resolution mass spectrometry and nuclear magnetic resonance. The compounds were evaluated for cytotoxic and diatom growth inhibition activities.
Norisoprenoids from the Brown Alga Sargassum naozhouense Tseng et Lu.[Pubmed:29414891]
Molecules. 2018 Feb 7;23(2):348.
A new C(11)-norisoprenoid derivative, sargassumone (1), has been isolated from Sargassum naozhouense together with six known norisoprenoids and a highly oxygenated cyclopentene: (2R,6S,8S,9S)-hexahydro-2,9-dihydroxy-4,4,8-trimethyl-6-acetyloxy-3(2H)-benzofuranone (2), (6S,8S,9R)-hexahydro-6,9-dihydroxy-4,4,8-trimethyl-2(2H)-benzofuranone (3), (6S,8S,9R)-hexahydro-6,9-dihydroxy-4,4,8-trimethyl-2(2H)-benzofuranone (4), loliolide (5), (+)-Epiloliolide (6), spheciospongones A (7), and (+)-kjellmanianone (8). Compound 1 was identified on the basis of nuclear magnetic resonance (NMR) and mass spectrometry (MS) analysis, and the absolute stereochemistry was defined by NOESY spectroscopy, minimizing energy calculation, and circular dichroism (CD) spectra. The known compounds 2-8, isolated from S. naozhouense for the first time, were identified by comparison of their physical and spectroscopic data with those reported in the literature. Compound 6 was tested for its inhibitory activity against protein tyrosine phosphatase 1B (PTP1B), antioxidant activity with 1,1-diphyl-2-picrylhydrazyl (DPPH) free radicals, and antimicrobial activity against resistant clinical isolates of Candida albicans, methicillin-resistant Staphylococcus aureus (MRSA), and Escherichia coli.
Inhibition of soluble epoxide hydrolase activity by compounds isolated from the aerial parts of Glycosmis stenocarpa.[Pubmed:26444316]
J Enzyme Inhib Med Chem. 2016 Aug;31(4):640-4.
The aim of this study is to search for soluble epoxide hydrolase (sEH) inhibitors from natural plants, bioassay-guided fractionation of lipophilic n-hexane and chloroform layers of an extract of the aerial parts of Glycosmis stenocarpa led to the isolation of 12 compounds (1-12) including murrayafoline-A (1), isomahanine (2), bisisomahanine (3), saropeptate (4), (24 S)-ergost-4-en-3,6-dione (5), stigmasta-4-en-3,6-dion (6), stigmast-4-en-3-one (7), beta-sitosterol (8), 24-methylpollinastanol (9), trans-phytol (10), neosarmentol III (11) and (+)-Epiloliolide (12). Their structures were elucidated on the basis of spectroscopic data. Among them, neosarmentol III (11) was isolated from nature for the first time. All the isolated compounds were evaluated for their inhibitory activity against sEH. Among isolated carbazole-type compounds, isomahanine (2) and bisisomahanine (3) were identified as a potent inhibitor of sEH, with IC50 values of 22.5 +/- 1.7 and 7.7 +/- 1.2 microM, respectively. Moreover, the inhibitory action of 2 and 3 represented mixed-type enzyme inhibition.
Anti-melanogenesis constituents from the seaweed Dictyota coriacea.[Pubmed:23738441]
Nat Prod Commun. 2013 Apr;8(4):427-8.
This study was conducted to identify the anti-melanogenesis constituents from a seaweed Dictyota coriacea (Holmes). Three known compounds, viz. 1,9-dihydroxycrenulide (1), Epiloliolide (2) and D-mannitol (3), were isolated from the ethanol extract. The melanin synthesis inhibition activities were evaluated using B16F10 melanoma cells for the isolates. Compared with the positive control, arbutin, compounds 1 and 2 exhibited more potency, showing 27.8 and 22.6% inhibition activities at a substrate concentration of 30 microg/mL. Our studies also indicate that these compounds are not cytotoxic. Hence, they might prove to be useful therapeutic agents for treating hyperpigmentation and effective components of whitening cosmetics.
Sesquiterpene lactones from Inula helianthus-aquatica.[Pubmed:22993986]
Zhongguo Zhong Yao Za Zhi. 2012 Jun;37(11):1586-9.
OBJECTIVE: To investigate the sesquiterpene lactones of the aerial parts of Inula helianthus-aquatica. METHOD: Compounds were isolated and purified by silica gel, Sephadex LH-20 and preparative HPLC. On the basis of physicochemical properties and spectroscopic data, their structures were identified. RESULT: Seven sesquiterpene lactones and four other compounds were obtained and identified as 2-desoxy-4-epi-pulchellin (1), 6-acetoxy-4-hydroxy-1, 10H-pseudoguaia-11 (13)-en-12,8-olide (2), 4-acetoxy-6-hydroxy-1, 10H-pseudoguaia-11(13)-en-12,8-olide (3), 8-epi-inuviscolide (4), 2,3,11,13-tetrahydroaromaticin (5), 11,13-dihydro-ergolide (6), 4-epipulchellin-2-O-acetate (7), 7-Epiloliolide (8), loliolide (9), beta-sitosterol (10) and daucosterol (11). CONCLUSION: All the compounds were isolated from this plant for the first time.
[Study on chemical constituents of the Ixeris chinensis].[Pubmed:22506392]
Zhong Yao Cai. 2011 Nov;34(11):1706-8.
OBJECTIVE: To study the chemical constituents of the Ixeris chinensis . METHODS: The constituents were isolated by silica gel column chromatography, HPLC and recrystallization and their structures were elucidated on the basis of spectral analysis. RESULTS: Fifteen compounds were isolated and identified as Methyl-4-hydroxyphenylacetate (1), Daucosterol (2), Sitosterol (3), Luteolin-7-O-beta-D-glucoside (4), 15-hydroxy-2-oxoguaia-1 (10), 3,11 (13)-triene-12,6-lactone (5), Chinensiolide B (6), Chinensiolide E (7), Ixerochinoside (8), Chinensiolide C (9), 10alpha-hydroxy-guaia-4(15)-ene-12,6-lactone (10), 10alpha-hydroxy-guaia-4 (15), 11 (13)-diene-12,6-lactone-3beta-O-beta-D-(6'-p-hydroxyphenylacetyl) glucopyranoside (11), Epiloliolide (12), Apigenin-O-beta-D-glucopyranoside (13), Luteolin (14), Lutein (15). CONCLUSION: Compounds 1,10,11,12 and 15 are isolated from this plant for the first time.
Isolation of antibabesial compounds from Brucea javanica, Curcuma xanthorrhiza, and Excoecaria cochinchinensis.[Pubmed:19270401]
Biosci Biotechnol Biochem. 2009 Mar 23;73(3):776-80.
One new curcuminoid, 3'-demethoxycyclocurcumin (1), was isolated from Curcuma xanthorrhiza as an antibabesial compound, together with p-hydroxybenzaldehyde (2) and cleomiscosin A (3) from Brucea javanica and (+)-Epiloliolide (4) from Excoecaria cochinchinensis. The antibabesial activities were examined in vitro, and compounds 1-4, and diminazene aceturate were studied with IC(50) values of 16.6, 7.6, 15.6, 10.0, and 0.6 microg/ml, respectively.
Bioactive constituents from Rollinia emarginata (Annonaceae).[Pubmed:17365716]
Nat Prod Res. 2007 Mar;21(3):254-9.
From an Argentine collection of the tropical tree Rollinia emarginata (Annonaceae), vomifoliol, dehydrovomifoliol, blumenol C, loliolide, 7-Epiloliolide, vanillin, dihydroactinolide, as well as other common plant constituents were obtained, and identified by their NMR and MS features compared with authentic samples. Antifeedant and toxic actions were exerted to the polyphagous moth Spodoptera frugiperda by the ethanol extract of the plant, at 250 ppm in the larval diet. Additionally, in greenhouse studies, a 200 ppm aqueous solution of the extract produced a post-emergency herbicidal effect on the annual weed common lambsquarter (Chenopodium album).


