GlabreneCAS# 60008-03-9 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 60008-03-9 | SDF | Download SDF |
| PubChem ID | 480774 | Appearance | Powder |
| Formula | C20H18O4 | M.Wt | 322.4 |
| Type of Compound | Flavonoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
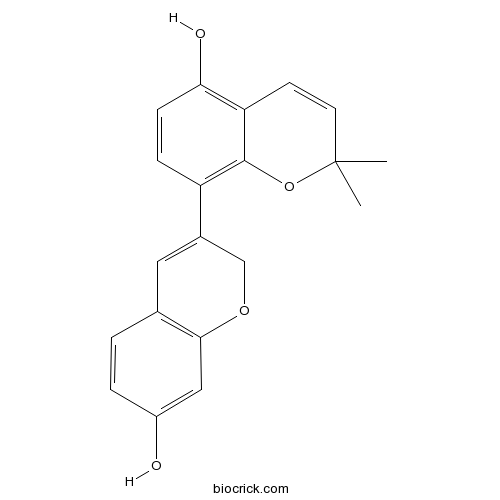
| Chemical Name | 8-(7-hydroxy-2H-chromen-3-yl)-2,2-dimethylchromen-5-ol | ||
| SMILES | CC1(C=CC2=C(C=CC(=C2O1)C3=CC4=C(C=C(C=C4)O)OC3)O)C | ||
| Standard InChIKey | NGGYSPUAKQMTNP-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C20H18O4/c1-20(2)8-7-16-17(22)6-5-15(19(16)24-20)13-9-12-3-4-14(21)10-18(12)23-11-13/h3-10,21-22H,11H2,1-2H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Glabrene, Liquiritin apioside, neolicuroside, and 18β-glycyrrhetic acid are the predominant phenolic derivatives partitioning at the interface and most likely the major contributors to the notable synergistic antioxidant activity when coupled with pea protein hydrolysates (PPHs).Glabrene has estrogen-like activity, it can stimulate DNA synthesis in human endothelial cells (ECV-304; E304) and has a bi-phasic effect on proliferation of human vascular smooth muscle cells (VSMC). Glabrene and isoliquiritigenin may serve as candidates for skin-lightening agents, they exert varying degrees of inhibition on tyrosinase-dependent melanin biosynthesis. |
| Targets | Tyrosinase | Estrogen receptor | Progestogen receptor |
| In vitro | Glabrene and isoliquiritigenin as tyrosinase inhibitors from licorice roots.[Pubmed: 12590456 ]J Agric Food Chem. 2003 Feb 26;51(5):1201-7.Tyrosinase is known to be a key enzyme in melanin biosynthesis, involved in determining the color of mammalian skin and hair. Various dermatological disorders, such as melasama, age spots, and sites of actinic damage, arise from the accumulation of an excessive level of epidermal pigmentation. The inadequacy of current therapies to treat these conditions as well as high cytotoxicity and mutagenicity, poor skin penetration, and low stability of formulations led us to seek new whitening agents to meet the medical requirements for depigmenting agents.
|
| In vivo | Estrogen-like activity of licorice root constituents: glabridin and glabrene, in vascular tissues in vitro and in vivo.[Pubmed: 15276622 ]J Steroid Biochem Mol Biol. 2004 Jul;91(3):147-55.Post-menopausal women have higher incidence of heart diseases compared to pre-menopausal women, suggesting a protective role for estrogen. The recently Women's Health Initiative (WHI) randomized controlled trial concluded that the overall heart risk exceeded benefits from use of combined estrogen and progestin as hormone replacement therapy for an average of five years among healthy postmenopausal US women. Therefore, there is an urgent need for new agents with tissue-selective activity with no deleterious effects.
|
| Structure Identification | J Agric Food Chem. 2014 Aug 13;62(32):8204-13.Synergy of licorice extract and pea protein hydrolysate for oxidative stability of soybean oil-in-water emulsions.[Pubmed: 25058384 ]Previously developed radical-scavenging pea protein hydrolysates (PPHs) prepared with Flavourzyme (Fla-PPH) and Protamex (Pro-PPH) were used as cosurfactants with Tween 20 to produce soybean oil-in-water (O/W) emulsions, and the suppression of lipid oxidation was investigated.
|

Glabrene Dilution Calculator

Glabrene Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.1017 mL | 15.5087 mL | 31.0174 mL | 62.0347 mL | 77.5434 mL |
| 5 mM | 0.6203 mL | 3.1017 mL | 6.2035 mL | 12.4069 mL | 15.5087 mL |
| 10 mM | 0.3102 mL | 1.5509 mL | 3.1017 mL | 6.2035 mL | 7.7543 mL |
| 50 mM | 0.062 mL | 0.3102 mL | 0.6203 mL | 1.2407 mL | 1.5509 mL |
| 100 mM | 0.031 mL | 0.1551 mL | 0.3102 mL | 0.6203 mL | 0.7754 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Oblongine
Catalog No.:BCN4103
CAS No.:60008-01-7
- 11Beta-hydroxyprogesterone
Catalog No.:BCN2211
CAS No.:600-57-7
- N-Me-DL-Ala-OH.HCl
Catalog No.:BCC2618
CAS No.:600-21-5
- Adenosine cyclophosphate
Catalog No.:BCN2190
CAS No.:60-92-4
- Phloretin
Catalog No.:BCN4128
CAS No.:60-82-2
- Phlorizin
Catalog No.:BCN4126
CAS No.:60-81-1
- Antipyrine
Catalog No.:BCC8834
CAS No.:60-80-0
- Veratramine
Catalog No.:BCN2965
CAS No.:60-70-8
- Methimazole
Catalog No.:BCC3812
CAS No.:60-56-0
- Tetracycline
Catalog No.:BCC9176
CAS No.:60-54-8
- Acetyl-Strophanthidin
Catalog No.:BCC8113
CAS No.:60-38-8
- Acetamide
Catalog No.:BCN4114
CAS No.:60-35-5
- Chlormethiazole hydrochloride
Catalog No.:BCC6830
CAS No.:6001-74-7
- BOC-L-6-HYDROXYNORLEUCINE
Catalog No.:BCN2360
CAS No.:77611-37-1
- Alizarin 2-methyl ether
Catalog No.:BCN3480
CAS No.:6003-11-8
- GL3
Catalog No.:BCN8180
CAS No.:60037-39-0
- N-Demethylechitamine
Catalog No.:BCN4104
CAS No.:60048-88-6
- Bay 65-1942 free base
Catalog No.:BCC1408
CAS No.:600734-02-9
- Bay 65-1942 HCl salt
Catalog No.:BCC1409
CAS No.:600734-06-3
- Secologanic acid
Catalog No.:BCN8214
CAS No.:60077-46-5
- Vogeloside
Catalog No.:BCN6737
CAS No.:60077-47-6
- (1)Benzopyrano(3,4-b)furo(2,3-h)(1)benzopyran-6(12H)-one, 1,2,-dihydro-5-hydroxy-8,9-dimethoxy-2-(1-methylethenyl)-, (R)-
Catalog No.:BCN8538
CAS No.:60077-62-5
- Confluentic acid
Catalog No.:BCN4105
CAS No.:6009-12-7
- Sodium taurochenodeoxycholate
Catalog No.:BCN8152
CAS No.:6009-98-9
Synergy of licorice extract and pea protein hydrolysate for oxidative stability of soybean oil-in-water emulsions.[Pubmed:25058384]
J Agric Food Chem. 2014 Aug 13;62(32):8204-13.
Previously developed radical-scavenging pea protein hydrolysates (PPHs) prepared with Flavourzyme (Fla-PPH) and Protamex (Pro-PPH) were used as cosurfactants with Tween 20 to produce soybean oil-in-water (O/W) emulsions, and the suppression of lipid oxidation was investigated. Both PPHs significantly retarded oxidation (P < 0.05) of the emulsions when stored at 37 degrees C for 14 days. Electron microscopy revealed an interfacial peptidyl membrane around oil droplets, which afforded steric restrictions to oxidation initiators. When licorice extract (LE) was also used in emulsion preparation, a remarkable synergistic oxidation inhibition was observed with both PPHs. LE adsorbed onto oil droplets either directly or through associating with PPH to produce a thick and compact interfacial membrane enabling the defense against oxygen species. Liquiritin apioside, neolicuroside, Glabrene, and 18beta-glycyrrhetic acid were the predominant phenolic derivatives partitioning at the interface and most likely the major contributors to the notable synergistic antioxidant activity when coupled with PPHs.
Glabrene and isoliquiritigenin as tyrosinase inhibitors from licorice roots.[Pubmed:12590456]
J Agric Food Chem. 2003 Feb 26;51(5):1201-7.
Tyrosinase is known to be a key enzyme in melanin biosynthesis, involved in determining the color of mammalian skin and hair. Various dermatological disorders, such as melasama, age spots, and sites of actinic damage, arise from the accumulation of an excessive level of epidermal pigmentation. The inadequacy of current therapies to treat these conditions as well as high cytotoxicity and mutagenicity, poor skin penetration, and low stability of formulations led us to seek new whitening agents to meet the medical requirements for depigmenting agents. The inhibitory effect of licorice extract on tyrosinase activity was higher than that expected from the level of glabridin in the extract. This led us to test for other components that may contribute to this strong inhibitory activity. Results indicated that Glabrene and isoliquiritigenin (2',4',4-trihydroxychalcone) in the licorice extract can inhibit both mono- and diphenolase tyrosinase activities. The IC(50) values for Glabrene and isoliquiritigenin were 3.5 and 8.1 microM, respectively, when tyrosine was used as substrate. The effects of Glabrene and isoliquiritigenin on tyrosinase activity were dose-dependent and correlated to their ability to inhibit melanin formation in melanocytes. This is the first study indicating that Glabrene and isoliquiritigenin exert varying degrees of inhibition on tyrosinase-dependent melanin biosynthesis, suggesting that isoflavenes and chalcones may serve as candidates for skin-lightening agents.
Estrogen-like activity of licorice root constituents: glabridin and glabrene, in vascular tissues in vitro and in vivo.[Pubmed:15276622]
J Steroid Biochem Mol Biol. 2004 Jul;91(3):147-55.
Post-menopausal women have higher incidence of heart diseases compared to pre-menopausal women, suggesting a protective role for estrogen. The recently Women's Health Initiative (WHI) randomized controlled trial concluded that the overall heart risk exceeded benefits from use of combined estrogen and progestin as hormone replacement therapy for an average of five years among healthy postmenopausal US women. Therefore, there is an urgent need for new agents with tissue-selective activity with no deleterious effects. In the present study, we tested the effects on vascular tissues in vitro and in vivo of two natural compounds derived from licorice root: glabridin, the major isoflavan, and Glabrene, an isoflavene, both demonstrated estrogen-like activities. Similar to estradiol-17beta (E2), glabridin (gla) stimulated DNA synthesis in human endothelial cells (ECV-304; E304) and had a bi-phasic effect on proliferation of human vascular smooth muscle cells (VSMC). Raloxifene inhibited gla as well as E2 activities. In animal studies, both intact females or after ovariectomy, gla similar to E2 stimulated the specific activity of creatine kinase (CK) in aorta (Ao) and in left ventricle of the heart (Lv). Glabrene (glb), on the other hand, had only the stimulatory effect on DNA synthesis in vascular cells, with no inhibition by raloxifene, suggesting a different mechanism of action. To further elucidate the mechanism of action of glb, cells were pre-incubated with glb and then exposed to either E2 or to gla; the DNA stimulation at low doses was unchanged but there was abolishment of the inhibition of VSMC cell proliferation at high doses as well as inhibition of CK stimulation by both E2 and by gla. We conclude that glb behaved differently than E2 or gla, but similarly to raloxifene, being a partial agonist/antagonist of E2. Glabridin, on the other hand, demonstrated only estrogenic activity. Therefore, we suggest the use of glb with or without E2 as a new agent for modulation of vascular injury and atherogenesis for the prevention of cardiovascular diseases in post-menopausal women.


