K02288ALK inhibitor CAS# 1431985-92-0 |
- SB 431542
Catalog No.:BCC3658
CAS No.:301836-41-9
- SB-505124 hydrochloride
Catalog No.:BCC1930
CAS No.:356559-13-2
- SB525334
Catalog No.:BCC2531
CAS No.:356559-20-1
- A 77-01
Catalog No.:BCC1318
CAS No.:607737-87-1
- LY2109761
Catalog No.:BCC3806
CAS No.:700874-71-1
- LY2157299
Catalog No.:BCC3709
CAS No.:700874-72-2
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 1431985-92-0 | SDF | Download SDF |
| PubChem ID | 46173038 | Appearance | Powder |
| Formula | C20H20N2O4 | M.Wt | 352.38 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | K 02288a | ||
| Solubility | DMSO : ≥ 58.6 mg/mL (166.30 mM) *"≥" means soluble, but saturation unknown. | ||
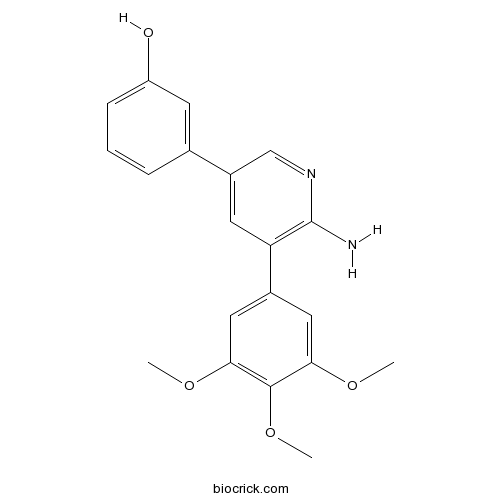
| Chemical Name | 3-[6-amino-5-(3,4,5-trimethoxyphenyl)pyridin-3-yl]phenol | ||
| SMILES | COC1=CC(=CC(=C1OC)OC)C2=C(N=CC(=C2)C3=CC(=CC=C3)O)N | ||
| Standard InChIKey | CJLMANFTWLNAKC-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C20H20N2O4/c1-24-17-9-13(10-18(25-2)19(17)26-3)16-8-14(11-22-20(16)21)12-5-4-6-15(23)7-12/h4-11,23H,1-3H3,(H2,21,22) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Potent and selective inhibitor of type I bone morphogenic protein (BMP) receptors (IC50 values are 1.1, 1.8, 6.4, 34.4, 220, 302 and 321 nM for ALK2, ALK1, ALK6, ALK3, ActRIIA, ALK4 and ALK5 respectively). Reduces BMP4-induced Smad1/5/8 phosphorylation in vitro (IC50 = 100 nM) and induces dorsalization of zebrafish embryos. |

K02288 Dilution Calculator

K02288 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.8378 mL | 14.1892 mL | 28.3785 mL | 56.7569 mL | 70.9461 mL |
| 5 mM | 0.5676 mL | 2.8378 mL | 5.6757 mL | 11.3514 mL | 14.1892 mL |
| 10 mM | 0.2838 mL | 1.4189 mL | 2.8378 mL | 5.6757 mL | 7.0946 mL |
| 50 mM | 0.0568 mL | 0.2838 mL | 0.5676 mL | 1.1351 mL | 1.4189 mL |
| 100 mM | 0.0284 mL | 0.1419 mL | 0.2838 mL | 0.5676 mL | 0.7095 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
K02288 is a highly selective inhibitor of ALK2 with IC50 value of 1.1nM [1].
K02288 is a 2-aminopyridine inhibitor of ALK2. It shows high selectivity for the homologous kinases ALK1-6 and ActRIIA. K02288 shows potency against the type I BMP receptors ALK1, ALK2 (IC50s range from 1–2nM), ALK3 and ALK6 (IC50s of 5–34nM), ALK4 (IC50 of 302nM) and ALK5 (IC50 of 321nM). K02288 has a favorable kinome-wide selectivity. In a panel of 200 human kinases, it shows more than 50% inhibition against only ABL and ARG at 0.1μM and only a further 6 kinases at 1μM [1].
K02288 also affects ALK2 related BMP signaling. In C2C12 cells, it reduces the phosphorylation of Smad dose-dependently when using BMP4 or BMP6 as ligands. Furthermore, K02288 induces a dorsalized phenotype in Tg(BRE:mRFP) transgenic zebrafish embryos [1].
References:
[1] Sanvitale CE, Kerr G, Chaikuad A, Ramel MC, Mohedas AH, Reichert S, Wang Y, Triffitt JT, Cuny GD, Yu PB, Hill CS, Bullock AN. A new class of small molecule inhibitor of BMP signaling. PLoS One. 2013 Apr 30;8(4):e62721.
- WEHI-539
Catalog No.:BCC2055
CAS No.:1431866-33-9
- (S)-Tedizolid
Catalog No.:BCC1294
CAS No.:1431699-67-0
- KN-92 hydrochloride
Catalog No.:BCC1681
CAS No.:1431698-47-3
- CCT241533 hydrochloride
Catalog No.:BCC1463
CAS No.:1431697-96-9
- SB-408124 Hydrochloride
Catalog No.:BCC1929
CAS No.:1431697-90-3
- OTSSP167
Catalog No.:BCC4314
CAS No.:1431697-89-0
- AT7867 dihydrochloride
Catalog No.:BCC1378
CAS No.:1431697-86-7
- AT7519 trifluoroacetate
Catalog No.:BCC1377
CAS No.:1431697-85-6
- gamma-secretase modulator 3
Catalog No.:BCC1585
CAS No.:1431697-84-5
- CAL-130 Hydrochloride
Catalog No.:BCC1441
CAS No.:1431697-78-7
- CAL-130
Catalog No.:BCC1440
CAS No.:1431697-74-3
- UNC1999
Catalog No.:BCC4552
CAS No.:1431612-23-5
- 3'-O-Galloylmyricitrin
Catalog No.:BCN8268
CAS No.:143202-36-2
- WAY 267464 dihydrochloride
Catalog No.:BCC7813
CAS No.:1432043-31-6
- Nyasicol 1,2-acetonide
Catalog No.:BCN6879
CAS No.:1432057-64-1
- Catalponol methylthiomethyl ether
Catalog No.:BCC8905
CAS No.:1432057-74-3
- 10-Acetoxyscandine
Catalog No.:BCN7035
CAS No.:1432058-90-6
- 3-(3-Hydroxy-3-methylbutanyl)-2,4,6-trihydroxybenzophenone
Catalog No.:BCC8588
CAS No.:1432062-53-7
- 8-Isomulberrin hydrate
Catalog No.:BCC8789
CAS No.:1432063-35-8
- 6-O-Acetylcoriatin
Catalog No.:BCN7048
CAS No.:1432063-63-2
- 10-O-Acetylisocalamendiol
Catalog No.:BCN7071
CAS No.:1432064-69-1
- (Z)-3-Hydroxy-5-methoxystilbene
Catalog No.:BCN6688
CAS No.:143207-76-5
- 7-Geranyloxy-5-methoxycoumarin
Catalog No.:BCN5806
CAS No.:1432075-68-7
- Dehydroadynerigenin beta-neritrioside
Catalog No.:BCN4706
CAS No.:143212-60-6
Polydatin Increases Radiosensitivity by Inducing Apoptosis of Stem Cells in Colorectal Cancer.[Pubmed:30745832]
Int J Biol Sci. 2019 Jan 1;15(2):430-440.
This study aimed to investigate the radiosensitizing effect of polydatin (PD) on colorectal cancer (CRC) and its underlying mechanism. The C57BL/6 mouse model of CRC was induced by treatment with azoxymethane (AOM)/dextran sodium sulfate (DSS) and then divided into four groups: control, PD alone, IR alone, and combination of PD and IR. Radiation therapy (200 cGy/min, 10Gy) was performed in mice in the experimental groups for once a week with a total of four times. Thirty minutes before IR, mice were intraperitoneally injected with PD at the dose of 25mg/kg. The number and volume of CRC xenografts were calculated. Immunohistochemical staining was performed to detect the expression of Ki67 and cleaved caspase-3 in tumor tissues samples. The effects of PD on proliferation and apoptosis were evaluated in CT26 and HCT116 colon tumor cells. Leucine-rich repeat-containing G-protein coupled receptor 5 positive (Lgr5(+)) cancer stem cells (CSCs) were sorted from CT26 cells and the effects of PD on their proliferation and apoptosis were observed to elucidate the radiosensitizing mechanism of PD in CRC cells. Combined therapy with PD and IR significantly decreased tumor volume, inhibited proliferation and induced apoptosis of tumor cells in the mouse model of CRC compared to other three groups. Compared to the IR group, in vitro assay showed that PD combined with IR inhibited proliferation and promoted apoptosis of CT26 and HCT116 colon tumor cells as well as Lgr5(+) CSCs. However, addition of the bone morphogenetic protein (BMP) type I receptor inhibitor K02288 (6.4nM) dramatically increased proliferation of Lgr5(+) CSCs and abolished the cytotoxic effect of PD combined with IR on Lgr5(+) CSCs. The in vivo and in vitro experiments demonstrated that IR combined treatment with PD could inhibit proliferation and promote apoptosis of CRC cells and Lgr5(+) CSCs, and BMP signaling pathway was involved in the radiosensitizing effect of PD.
Transforming growth factor beta-inhibitor Repsox downregulates collagen expression of scleroderma dermal fibroblasts and prevents bleomycin-induced mice skin fibrosis.[Pubmed:28418584]
Exp Dermatol. 2017 Nov;26(11):1139-1143.
Inhibition of transforming growth factor (TGF)-beta1 signalling may be one of the most reliable approaches to treat skin fibrosis of scleroderma. Although there have been many basic researches of TGF-beta blockade reagents, few of them were proved to have inhibitory effects on fibrosis both in vitro and in vivo. In this study, we randomly chose four commercially available low molecular weight compounds (Repsox, LY2109761, LY364947 and K02288) from TGF-beta1 inhibitor library, and compared their antifibrotic effects in vitro and in vivo. We demonstrated that Repsox has the most potent inhibitory effects on TGF-beta-induced expression of CTGF and collagen of cultured normal dermal fibroblasts in vitro and their constitutive overexpression of scleroderma fibroblast in vitro. In addition, Repsox could attenuate skin fibrosis by bleomycin in vivo, via the downregulation of CTGF or collagen. Our results may facilitate clinical trial of Repsox against fibrotic diseases in future.
[Bushen Tiaojing Recipe Regulated Expressions of BMPR I[/ALK6-Smads in Mouse Oocytes Cul- ture in vitro].[Pubmed:30641014]
Zhongguo Zhong Xi Yi Jie He Za Zhi. 2016 Oct;36(10):1241-1246.
Objective To observe the effects of Bushen Tiaojing Recipe (BTR) on the counts of survival preantral follicles and the bone morphogenetic protein receptor II (BMPR II )/activin receptor- like kinase 6-drosophila mothers against decapentaplegic proteins (ALK6-Smads) signal pathway in oocytes cultured in vitro, and to study its mechanism for improving the quality of oocytes. Methods Prean- tral follicles were mechanically isolated from 65 female 12-day old healthy Kunming mice, which were inoculated by normal rats' serum (as the control group) , high, medium, low dose BTR containing serums (as Shen-supplementing groups) , high dose BTR containing serum + K02288 (as the inhibitor group) , respectively. All were cultured by common method in vitro. On the 6th day the counts of survival preantral follicles were compared between each Shen-supplementing group and the control group respectively. mR- NA expressions of BMPR II, ALK6, Smad1 , Smad5, and Smad8 were detected by Real-time fluorescence quantitative PCR. The protein expressions of indices mentioned above and phospho-Smadl/5/8 (p- Smadl/5/8) were detected by cellular immunofluorescence test. Results Compared with the control group, the quantity of survival preantral follicles increased in the high dose BTR containing serum group; mRNA expressions of BMPR II, ALK6, Smad5, and Smad8 were elevated, protein expressions of indi- ces mentioned above and p-Smadl/5/8 were increased in the 3 Shen-supplementing groups (P <0. 05) ; mRNA and protein expressions of Smad1 were increased in high and medium dose BTR containing serum groups (P<0.05). Compared with the high dose BTR containing serum group, protein expressions of Smad1/5/8 were reduced in the inhibitor group (P <0.05). Conclusion BTR could elevate the quantity of survival preantral follicles cultured in vitroand improve the quality of oocytes, which might be possibly as- sociated to regulating the BMPR II/ALK6-Smads signal pathway in oocytes.
Rac1 promotes chondrogenesis by regulating STAT3 signaling pathway.[Pubmed:27306109]
Cell Biol Int. 2016 Sep;40(9):976-83.
The small GTPase protein Rac1 is involved in a wide range of biological processes including cell differentiation. Previously, Rac1 was shown to promote chondrogenesis in micromass cultures of limb mesenchyme. However, the pathways mediating Rac1's role in chondrogenesis are not fully understood. This study aimed to explore the molecular mechanisms by which Rac1 regulates chondrogenic differentiation. Phosphorylation of signal transducer and activator of transcription 3 (STAT3) was increased as chondrogenesis proceeded in micromass cultures of chick wing bud mesenchyme. Inhibition of Rac1 with NSC23766, janus kinase 2 (JAK2) with AG490, or STAT3 with stattic inhibited chondrogenesis and reduced phosphorylation of STAT3. Conversely, overexpression of constitutively active Rac1 (Rac L61) increased phosphorylation of STAT3. Rac L61 expression resulted in increased expression of interleukin 6 (IL-6), and treatment with IL-6 increased phosphorylation of STAT3. NSC23766, AG490, and stattic prohibited cell aggregation, whereas expression of Rac L61 increased cell aggregation, which was reduced by stattic treatment. Our studies indicate that Rac1 induces STAT3 activation through expression and action of IL-6. Overexpression of Rac L61 increased expression of bone morphogenic protein 4 (BMP4). BMP4 promoted chondrogenesis, which was inhibited by K02288, an activin receptor-like kinase-2 inhibitor, and increased phosphorylation of p38 MAP kinase. Overexpression of Rac L61 also increased phosphorylation of p38 MAPK, which was reduced by K02288. These results suggest that Rac1 activates STAT3 by expression of IL-6, which in turn increases expression and activity of BMP4, leading to the promotion of chondrogenesis.
A small molecule targeting ALK1 prevents Notch cooperativity and inhibits functional angiogenesis.[Pubmed:25557927]
Angiogenesis. 2015 Apr;18(2):209-17.
Activin receptor-like kinase 1 (ALK1, encoded by the gene ACVRL1) is a type I BMP/TGF-beta receptor that mediates signalling in endothelial cells via phosphorylation of SMAD1/5/8. During angiogenesis, sprouting endothelial cells specialise into tip cells and stalk cells. ALK1 synergises with Notch in stalk cells to induce expression of the Notch targets HEY1 and HEY2 and thereby represses tip cell formation and angiogenic sprouting. The ALK1-Fc soluble protein fusion has entered clinic trials as a therapeutic strategy to sequester the high-affinity extracellular ligand BMP9. Here, we determined the crystal structure of the ALK1 intracellular kinase domain and explored the effects of a small molecule kinase inhibitor K02288 on angiogenesis. K02288 inhibited BMP9-induced phosphorylation of SMAD1/5/8 in human umbilical vein endothelial cells to reduce both the SMAD and the Notch-dependent transcriptional responses. In endothelial sprouting assays, K02288 treatment induced a hypersprouting phenotype reminiscent of Notch inhibition. Furthermore, K02288 caused dysfunctional vessel formation in a chick chorioallantoic membrane assay of angiogenesis. Such activity may be advantageous for small molecule inhibitors currently in preclinical development for specific BMP gain of function conditions, including diffuse intrinsic pontine glioma and fibrodysplasia ossificans progressiva, as well as more generally for other applications in tumour biology.
Structure-activity relationship of 3,5-diaryl-2-aminopyridine ALK2 inhibitors reveals unaltered binding affinity for fibrodysplasia ossificans progressiva causing mutants.[Pubmed:25101911]
J Med Chem. 2014 Oct 9;57(19):7900-15.
There are currently no effective therapies for fibrodysplasia ossificans progressiva (FOP), a debilitating and progressive heterotopic ossification disease caused by activating mutations of ACVR1 encoding the BMP type I receptor kinase ALK2. Recently, a subset of these same mutations of ACVR1 have been identified in diffuse intrinsic pontine glioma (DIPG) tumors. Here we describe the structure-activity relationship for a series of novel ALK2 inhibitors based on the 2-aminopyridine compound K02288. Several modifications increased potency in kinase, thermal shift, or cell-based assays of BMP signaling and transcription, as well as selectivity for ALK2 versus closely related BMP and TGF-beta type I receptor kinases. Compounds in this series exhibited a wide range of in vitro cytotoxicity that was not correlated with potency or selectivity, suggesting mechanisms independent of BMP or TGF-beta inhibition. The study also highlights a potent 2-methylpyridine derivative 10 (LDN-214117) with a high degree of selectivity for ALK2 and low cytotoxicity that could provide a template for preclinical development. Contrary to the notion that activating mutations of ALK2 might alter inhibitor efficacy due to potential conformational changes in the ATP-binding site, the compounds demonstrated consistent binding to a panel of mutant and wild-type ALK2 proteins. Thus, BMP inhibitors identified via activity against wild-type ALK2 signaling are likely to be of clinical relevance for the diverse ALK2 mutant proteins associated with FOP and DIPG.
A new class of small molecule inhibitor of BMP signaling.[Pubmed:23646137]
PLoS One. 2013 Apr 30;8(4):e62721.
Growth factor signaling pathways are tightly regulated by phosphorylation and include many important kinase targets of interest for drug discovery. Small molecule inhibitors of the bone morphogenetic protein (BMP) receptor kinase ALK2 (ACVR1) are needed urgently to treat the progressively debilitating musculoskeletal disease fibrodysplasia ossificans progressiva (FOP). Dorsomorphin analogues, first identified in zebrafish, remain the only BMP inhibitor chemotype reported to date. By screening an assay panel of 250 recombinant human kinases we identified a highly selective 2-aminopyridine-based inhibitor K02288 with in vitro activity against ALK2 at low nanomolar concentrations similar to the current lead compound LDN-193189. K02288 specifically inhibited the BMP-induced Smad pathway without affecting TGF-beta signaling and induced dorsalization of zebrafish embryos. Comparison of the crystal structures of ALK2 with K02288 and LDN-193189 revealed additional contacts in the K02288 complex affording improved shape complementarity and identified the exposed phenol group for further optimization of pharmacokinetics. The discovery of a new chemical series provides an independent pharmacological tool to investigate BMP signaling and offers multiple opportunities for pre-clinical development.
Development of an ALK2-biased BMP type I receptor kinase inhibitor.[Pubmed:23547776]
ACS Chem Biol. 2013;8(6):1291-302.
The bone morphogenetic protein (BMP) signaling pathway has essential functions in development, homeostasis, and the normal and pathophysiologic remodeling of tissues. Small molecule inhibitors of the BMP receptor kinase family have been useful for probing physiologic functions of BMP signaling in vitro and in vivo and may have roles in the treatment of BMP-mediated diseases. Here we describe the development of a selective and potent inhibitor of the BMP type I receptor kinases, LDN-212854, which in contrast to previously described BMP receptor kinase inhibitors exhibits nearly 4 orders of selectivity for BMP versus the closely related TGF-beta and Activin type I receptors. In vitro, LDN-212854 exhibits some selectivity for ALK2 in preference to other BMP type I receptors, ALK1 and ALK3, which may permit the interrogation of ALK2-mediated signaling, transcriptional activity, and function. LDN-212854 potently inhibits heterotopic ossification in an inducible transgenic mutant ALK2 mouse model of fibrodysplasia ossificans progressiva. These findings represent a significant step toward developing selective inhibitors targeting individual members of the highly homologous BMP type I receptor family. Such inhibitors would provide greater resolution as probes of physiologic function and improved selectivity against therapeutic targets.


