N1,N11-Diethylnorspermine tetrahydrochlorideSpermine and spermidine acetyltransferase potentiator CAS# 156886-85-0 |
- SU14813
Catalog No.:BCC1971
CAS No.:627908-92-3
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 156886-85-0 | SDF | Download SDF |
| PubChem ID | 11501988 | Appearance | Powder |
| Formula | C13H36Cl4N4 | M.Wt | 390.27 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 100 mM in water | ||
| Chemical Name | N-ethyl-N'-[3-[3-(ethylamino)propylamino]propyl]propane-1,3-diamine;tetrahydrochloride | ||
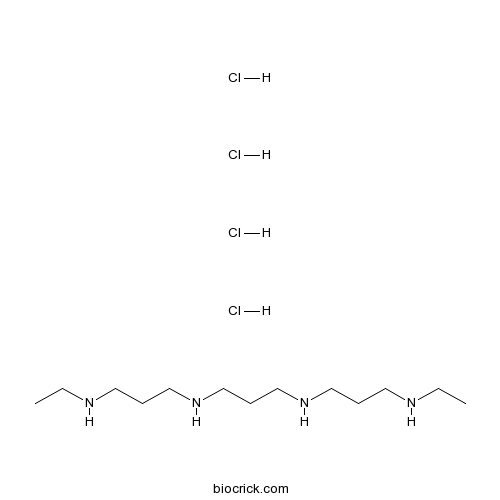
| SMILES | CCNCCCNCCCNCCCNCC.Cl.Cl.Cl.Cl | ||
| Standard InChIKey | LAWBSOKBIROCQP-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C13H32N4.4ClH/c1-3-14-8-5-10-16-12-7-13-17-11-6-9-15-4-2;;;;/h14-17H,3-13H2,1-2H3;4*1H | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Highly potent inducer of spermidine/spermine N1-acetyltransferase, able to increase enzyme activity 200-1000 fold. |

N1,N11-Diethylnorspermine tetrahydrochloride Dilution Calculator

N1,N11-Diethylnorspermine tetrahydrochloride Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.5623 mL | 12.8116 mL | 25.6233 mL | 51.2466 mL | 64.0582 mL |
| 5 mM | 0.5125 mL | 2.5623 mL | 5.1247 mL | 10.2493 mL | 12.8116 mL |
| 10 mM | 0.2562 mL | 1.2812 mL | 2.5623 mL | 5.1247 mL | 6.4058 mL |
| 50 mM | 0.0512 mL | 0.2562 mL | 0.5125 mL | 1.0249 mL | 1.2812 mL |
| 100 mM | 0.0256 mL | 0.1281 mL | 0.2562 mL | 0.5125 mL | 0.6406 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Ibuprofen
Catalog No.:BCC3791
CAS No.:15687-27-1
- Cephalexin
Catalog No.:BCC4646
CAS No.:15686-71-2
- Hosenkoside A
Catalog No.:BCN4962
CAS No.:156791-82-1
- Iristectorene B
Catalog No.:BCN7695
CAS No.:156791-81-0
- Agroastragaloside I
Catalog No.:BCC8294
CAS No.:156769-94-7
- 3-beta-Hydroxyergost-5-en-7-one
Catalog No.:BCN1704
CAS No.:156767-69-0
- Hosenkoside C
Catalog No.:BCN2632
CAS No.:156764-83-9
- Hosenkoside B
Catalog No.:BCN4584
CAS No.:156764-82-8
- (RS)-(±)-Sulpiride
Catalog No.:BCC6835
CAS No.:15676-16-1
- Delphinidin-3-O-rutinoside chloride
Catalog No.:BCN3115
CAS No.:15674-58-5
- SNC 80
Catalog No.:BCC6785
CAS No.:156727-74-1
- Rostafuroxin (PST 2238)
Catalog No.:BCC6431
CAS No.:156722-18-8
- Licofelone
Catalog No.:BCC4432
CAS No.:156897-06-2
- OSU 6162 hydrochloride
Catalog No.:BCC7424
CAS No.:156907-84-5
- Isosalicifolin
Catalog No.:BCN6502
CAS No.:156974-99-1
- Grosvenorin
Catalog No.:BCN1262
CAS No.:156980-60-8
- Wedeliatrilolactone A
Catalog No.:BCN6732
CAS No.:156993-29-2
- H-D-Arg-OH
Catalog No.:BCC2868
CAS No.:157-06-2
- 5,7-Dihydroxy-3,4',8-trimethoxyflavone
Catalog No.:BCN1551
CAS No.:1570-09-8
- U 89843A
Catalog No.:BCC7466
CAS No.:157013-85-9
- SNAP 5089
Catalog No.:BCC7350
CAS No.:157066-77-8
- Lipedoside B-III
Catalog No.:BCC8201
CAS No.:157085-48-8
- 3-Amino-4-hydroxybenzoic acid
Catalog No.:BCC8610
CAS No.:1571-72-8
- Ajuforrestin A
Catalog No.:BCN8008
CAS No.:157110-18-4
Structural basis for differential induction of spermidine/spermine N1-acetyltransferase activity by novel spermine analogs.[Pubmed:9224814]
Mol Pharmacol. 1997 Jul;52(1):69-74.
The spermine analog N1,N11-diethylnorspermine (DE-333, also known as DENSPM or BENSPM) is regarded as the most potent known inducer of the polyamine catabolic enzyme, spermidine/spermine N1-acetyltransferase (SSAT), increasing activity by more than 200- to 1000-fold in certain cell types. The relative ability of a series of eight systematically modified DE-333 analogs to affect SSAT expression was examined in Malme-3M human melanoma cells, one of several cell lines known to be especially responsive to induction of this enzyme. In particular, we examined the relative contribution of induction of enzyme mRNA and prolongation of enzyme half-life to analog-mediated increases in enzyme activity. Induction of enzyme mRNA was most influenced by intra-amine carbon distances; relative effectiveness was found to be proportional to the number of three-carbon units. Stabilization of enzyme was most determined by the terminal N-alkyl substituent size; among methyl, ethyl and propyl groups, methyl was least effective. Thus, DE-333, which most potently induces SSAT mRNA and effectively stabilizes SSAT enzyme activity, produces the greatest increase in enzyme activity. Although other contributing mechanisms may be involved, the relative abilities of the various analogs to induce enzyme activity is at least partially attributable to their combined effects on enzyme mRNA and protein half-life. These data reveal the highly sensitive structure-activity relationships that underlie and control spermine analog induction of SSAT activity. Pending further definition of the relationship between SSAT induction and antitumor growth and toxicity in vivo, these relationships may be used to optimize therapeutic efficacy.
Antitumor activity of N1,N11-bis(ethyl)norspermine against human melanoma xenografts and possible biochemical correlates of drug action.[Pubmed:8425191]
Cancer Res. 1993 Feb 1;53(3):581-6.
In in vitro systems, the spermine analogue, N1,N11-bis(ethyl)norspermine (BENSPM), suppresses the polyamine biosynthetic enzymes, ornithine and S-adenosylmethionine decarboxylase (ornithine decarboxylase and S-adenosylmethionine decarboxylase, respectively), greatly induces the polyamine catabolic enzyme, spermidine/spermine N1-acetyltransferase (SSAT), depletes polyamine pools, and inhibits cell growth. Against MALME-3 M human melanoma xenografts, BENSPM and related homologues demonstrate potent antitumor activity that has been found to correlate positively with their ability to induce SSAT activity in vitro. Herein, we further evaluate the antitumor activity of BENSPM and at the same time characterize the biochemical effects of BENSPM treatment on polyamine metabolism of selected normal and tumor tissues. At 40 mg/kg 3 times/day for 6 days i.p., BENSPM suppressed growth of MALME-3 M human melanoma xenografts during treatment and for 65 days afterwards. Similar antitumor activity was obtained with 120 mg/kg once daily for 6 days and 40 mg/kg once daily for 6 days, indicating that against this tumor model, the dosing schedule can be relaxed up to sixfold without compromising antitumor activity. When MALME-3 M tumor-bearing mice were retreated with BENSPM 2 weeks after the first treatment at 40 mg/kg 3 times/day for 6 days, initial tumor volumes of 85 mm3 were reduced to < 10 mm3. Analysis of melanoma, liver, and kidney tissues from mice treated with 40 mg/kg 3 times/day for 6 days revealed relatively similar accumulations of BENSPM in all tissues at levels greater than the original total content of polyamine pools. By 2 weeks following treatment, BENSPM pools in normal tissues were almost gone, whereas in tumor tissues significant amounts (40%) were still retained. The biosynthetic enzymes, ornithine decarboxylase and S-adenosylmethionine decarboxylase, gave no indication of enzyme suppression (or increase) by the analogue as typically occurs in vitro. By contrast, SSAT was induced from an average of < 50 pmol/min/mg in control tissues to 320 pmol/min/mg in liver, 1255 pmol/min/mg in kidney, and 13,710 pmol/min/mg in MALME-3M tumor. Two weeks later, SSAT activity was still 12 times higher in tumor than in kidney. Polyamine pools (putrescine, spermidine, and spermine) were reduced after treatment in all tissues and approached near-total depletion in the tumor. Good antitumor activity and even more potent induction of SSAT (i.e., 26,680 pmol/min/mg) was also observed in PANUT-3 human melanoma xenografts. Overall, the findings reveal meaningful antitumor activity by BENSPM against 2 human melanoma xenografts and provide in vivo evidence consistent with SSAT-induced polyamine depletion playing a determining role in at least the initial phase of the antitumor response.
Antitumor activity of N,N'-bis(ethyl)spermine homologues against human MALME-3 melanoma xenografts.[Pubmed:1568212]
Cancer Res. 1992 May 1;52(9):2424-30.
The spermine analogues, N1,N12-bis(ethyl)spermine (BESPM), N1,N11-bis(ethyl)norspermine (BENSPM), and N1,N14-bis(ethyl)-homospermine (BEHSPM) behave similarly in down-regulating the key polyamine biosynthetic enzymes, ornithine and S-adenosylmethionine decarboxylase, but differ distinctly in their abilities to induce the polyamine catabolic enzyme, spermidine/spermine-N1-acetyltransferase; BENSPM is 6-fold more effective than BESPM in increasing spermidine/spermine-N1-acetyltransferase activity and BEHSPM is 10-fold less effective. Since MALME-3 human melanoma cells are extremely responsive to spermidine/spermine-N1-acetyltransferase induction (i.e., increases greater than 200-fold) and since this induction correlates with growth inhibition among melanoma cell lines, the ability of these homologues to inhibit the growth of MALME-3 xenografts was examined. Analogues were administered i.p. three times per day (i.e., every 8 h) for 6 days at the following doses per injection: BEHSPM, 1.5, 3, or 6 mg/kg; BESPM, 10, 20, or 40 mg/kg; BENSPM, 20, 40, or 80 mg/kg. At the highest tolerated doses, all of the analogues fully suppressed growth of established (100-200 mm3) MALME-3 tumor during treatment and sustained tumor growth inhibition following treatment as follows: BEHSPM, 14 days; BESPM, 27 days, and BENSPM, 37 days. The tumor delay (to reach 1000 mm3 relative to control) at the highest tolerated doses was as follows: BEHSPM, 20 days; BESPM, 34 days, and BENSPM, 63 days. The rank order of analogue host toxicity as indicated by weight loss was opposite that for antitumor activity, BEHSPM was most toxic, BESPM, intermediate, and BENSPM, least toxic. Thus, the most effective of the three homologues, BENSPM, was best tolerated, and produced an initial tumor regression, full suppression of tumor regrowth during treatment, and sustained inhibition of tumor regrowth for 37 days after treatment stopped. Owing to its potent antitumor activity, mild host toxicity, and novel apparent mechanism of action, BENSPM is being considered for further development toward clinical trial.
Characterization of human spermidine/spermine N1-acetyltransferase purified from cultured melanoma cells.[Pubmed:1989509]
Arch Biochem Biophys. 1991 Feb 1;284(2):238-44.
Extreme inducibility of spermidine/spermine acetyltransferase (SSAT) by bis-ethyl derivatives of spermine in human large cell lung carcinoma and melanoma cells has prompted biochemical characterization of the purified enzyme. Treatment of human MALME-3 melanoma cells with 10 microM N1,N11-bis(ethyl)norspermine (BENSPM) for 48-72 h increased SSAT activity by some 1000- to 4000-fold and enabled purification of the enzyme by established procedures--binding on immobilized spermine and elution with spermine followed by binding on Matrex Blue A and elution with coenzyme A. The enzyme showed a single band by sodium dodecyl sulfate-polyacrylamide gel electrophoresis with a single subunit species and molecular weight of approximately 20,300 Da. By gel permeation chromatography, the holoenzyme was found to have a molecular weight of 80,000 Da, suggesting a total of four identical subunits. Purified SSAT had a specific activity of 285 mumol/min/mg for spermidine and Km values of 5.9 microM for acetylcoenzyme A, 55 microM for spermidine, 5 microM for spermine, 36 microM for N1-acetylspermine, 1.6 microM for norspermidine, and 4 microM for norspermine. Homologs of BENSPM were found to be competitive inhibitors of spermidine acetylation, with Ki values of 0.8 microM for BENSPM, 1.9 microM for N1,N12-bis-(ethyl)spermine and 17 microM for N1,N14-bis-(ethyl)-homospermine. Correlation of these values with the relative abilities of the homologs to increase SSAT in intact cells suggests that formation of an enzyme inhibitor complex may play a contributing role in enzyme induction.


