RepaglinideKir6 (KATP) channel blocker CAS# 135062-02-1 |
- Dofetilide
Catalog No.:BCC3770
CAS No.:115256-11-6
- Dronedarone
Catalog No.:BCN2176
CAS No.:141626-36-0
- NS309
Catalog No.:BCC1809
CAS No.:18711-16-5
- TRAM-34
Catalog No.:BCC1122
CAS No.:289905-88-0
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 135062-02-1 | SDF | Download SDF |
| PubChem ID | 65981 | Appearance | Powder |
| Formula | C27H36N2O4 | M.Wt | 452.59 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | AG-EE 623ZW | ||
| Solubility | DMSO : ≥ 50 mg/mL (110.48 mM) *"≥" means soluble, but saturation unknown. | ||
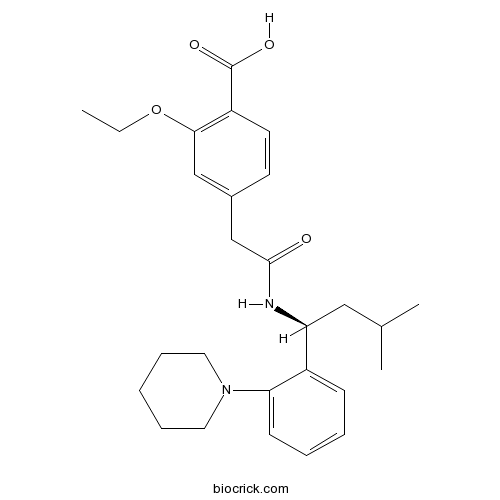
| Chemical Name | 2-ethoxy-4-[2-[[(1S)-3-methyl-1-(2-piperidin-1-ylphenyl)butyl]amino]-2-oxoethyl]benzoic acid | ||
| SMILES | CCOC1=C(C=CC(=C1)CC(=O)NC(CC(C)C)C2=CC=CC=C2N3CCCCC3)C(=O)O | ||
| Standard InChIKey | FAEKWTJYAYMJKF-QHCPKHFHSA-N | ||
| Standard InChI | InChI=1S/C27H36N2O4/c1-4-33-25-17-20(12-13-22(25)27(31)32)18-26(30)28-23(16-19(2)3)21-10-6-7-11-24(21)29-14-8-5-9-15-29/h6-7,10-13,17,19,23H,4-5,8-9,14-16,18H2,1-3H3,(H,28,30)(H,31,32)/t23-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Kir6 (KATP) channel blocker that binds with high affinity for SUR1 when co-expressed with Kir6.2 (Kd = 0.42 nM). Antidiabetic glucose regulator with hypoglycaemic effect in vivo. |

Repaglinide Dilution Calculator

Repaglinide Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.2095 mL | 11.0475 mL | 22.0951 mL | 44.1901 mL | 55.2376 mL |
| 5 mM | 0.4419 mL | 2.2095 mL | 4.419 mL | 8.838 mL | 11.0475 mL |
| 10 mM | 0.221 mL | 1.1048 mL | 2.2095 mL | 4.419 mL | 5.5238 mL |
| 50 mM | 0.0442 mL | 0.221 mL | 0.4419 mL | 0.8838 mL | 1.1048 mL |
| 100 mM | 0.0221 mL | 0.1105 mL | 0.221 mL | 0.4419 mL | 0.5524 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Kir6 (KATP) channel blocker that binds with high affinity for SUR1 when co-expressed with Kir6.2 (Kd = 0.42 nM). Antidiabetic glucose regulator with hypoglycaemic effect in vivo.
- MK-5172
Catalog No.:BCC1762
CAS No.:1350514-68-9
- MK-5172 hydrate
Catalog No.:BCC1763
CAS No.:1350462-55-3
- Clopidogrel bisulfate
Catalog No.:BCC8917
CAS No.:135046-48-9
- Z-Hyp-OH
Catalog No.:BCC3257
CAS No.:13504-85-3
- Pseudotropine
Catalog No.:BCN1932
CAS No.:135-97-7
- 2,2'-Dithiobisbenzanilide
Catalog No.:BCC8497
CAS No.:135-57-9
- Taccalonolide AJ
Catalog No.:BCN2971
CAS No.:1349904-82-0
- SAR245409
Catalog No.:BCC2534
CAS No.:934493-76-2
- 4-P-PDOT
Catalog No.:BCC6900
CAS No.:134865-74-0
- 8-M-PDOT
Catalog No.:BCC6901
CAS No.:134865-70-6
- 6''-O-Acetylglycitin
Catalog No.:BCN3866
CAS No.:134859-96-4
- A 412997 dihydrochloride
Catalog No.:BCC6224
CAS No.:1347744-96-0
- kb-NB77-78
Catalog No.:BCC5462
CAS No.:1350622-33-1
- Camelliaside A
Catalog No.:BCN3871
CAS No.:135095-52-2
- Fmoc-Nva-OH
Catalog No.:BCC3302
CAS No.:135112-28-6
- Z-Tyr-OMe
Catalog No.:BCC2746
CAS No.:13512-31-7
- 2',7-Dihydroxy-5,8-dimethoxyflavanone
Catalog No.:BCN4041
CAS No.:1351338-14-1
- H-Sar-OMe.HCl
Catalog No.:BCC3335
CAS No.:13515-93-0
- H-DL-Ala-OMe.HCl
Catalog No.:BCC2854
CAS No.:13515-97-4
- Sarpogrelate hydrochloride
Catalog No.:BCC5247
CAS No.:135159-51-2
- Amooracetal
Catalog No.:BCN6876
CAS No.:1351617-73-6
- 21,23:24,25-Diepoxy-21,23-dimethoxytirucall-7-en-3-one
Catalog No.:BCN1581
CAS No.:1351617-74-7
- ONO-4059
Catalog No.:BCC6463
CAS No.:1351635-67-0
- HG-10-102-01
Catalog No.:BCC4271
CAS No.:1351758-81-0
Estimation of ellagic acid and/or repaglinide effects on insulin signaling, oxidative stress, and inflammatory mediators of liver, pancreas, adipose tissue, and brain in insulin resistant/type 2 diabetic rats.[Pubmed:28092161]
Appl Physiol Nutr Metab. 2017 Feb;42(2):181-192.
Even though ellagic acid has previously been valued in many models of cancer, so far its full mechanistic effect as a natural antiapoptotic agent in the prevention of type 2 diabetes complications has not been completely elucidated, which was the goal of this study. We fed albino rats a high-fat fructose diet (HFFD) for 2 months to induce insulin resistance/type 2 diabetes and then treated the rats with ellagic acid (10 mg/kg body weight, orally) and/or Repaglinide (0.5 mg/kg body weight, orally) for 2 weeks. At the serum level, ellagic acid challenged the consequences of HFFD, significantly improving the glucose/insulin balance, liver enzymes, lipid profile, inflammatory cytokines, redox level, adipokines, ammonia, and manganese. At the tissue level (liver, pancreas, adipose tissue, and brain), ellagic acid significantly enhanced insulin signaling, autophosphorylation, adiponectin receptors, glucose transporters, inflammatory mediators, and apoptotic markers. Remarkably, combined treatment with both ellagic acid and Repaglinide had a more pronounced effect than treatment with either alone. These outcomes give new insight into the promising molecular mechanisms by which ellagic acid modulates numerous factors induced in the progression of diabetes.
Estimation of the Contribution of CYP2C8 and CYP3A4 in Repaglinide Metabolism by Human Liver Microsomes Under Various Buffer Conditions.[Pubmed:28238899]
J Pharm Sci. 2017 Sep;106(9):2847-2852.
We have previously reported that the microsomal activities of CYP2C8 and CYP3A4 largely depend on the buffer condition used in in vitro metabolic studies, with different patterns observed between the 2 isozymes. In the present study, therefore, the possibility of buffer condition dependence of the fraction metabolized by CYP2C8 (fm2C8) for Repaglinide, a dual substrate of CYP2C8 and CYP3A4, was estimated using human liver microsomes under various buffer conditions. Montelukast and ketoconazole showed a potent and concentration-dependent inhibition of CYP2C8-mediated paclitaxel 6alpha-hydroxylation and CYP3A4-mediated triazolam alpha-hydroxylation, respectively, without dependence on the buffer condition. Repaglinide depletion was inhibited by both inhibitors, but the degree of inhibition depended on buffer conditions. Based on these results, the contribution of CYP2C8 in Repaglinide metabolism was estimated to be larger than that of CYP3A4 under each buffer condition, and the fm2C8 value of 0.760, estimated in 50 mM phosphate buffer, was the closest to the value (0.801) estimated in our previous modeling analysis based on its concentration increase in a clinical drug interaction study. Researchers should be aware of the possibility of buffer condition affecting the estimated contribution of enzyme(s) in drug metabolism processes involving multiple enzymes.
Evaluation of Pharmacokinetic Interactions Between Lesinurad, a New Selective Urate Reabsorption Inhibitor, and CYP Enzyme Substrates Sildenafil, Amlodipine, Tolbutamide, and Repaglinide.[Pubmed:28067999]
Clin Pharmacol Drug Dev. 2017 Jul;6(4):363-376.
Lesinurad is a selective uric acid reabsorption inhibitor approved for the treatment of hyperuricemia associated with gout in combination with xanthine oxidase inhibitors. In vitro assays indicate that lesinurad is an inducer of CYPs in the order CYP3A > CYP2C8 > CYP2C9 > CYP2C19 > CYP2B6 and an inhibitor of CYP2C8 and CYP2C9. To investigate the drug interaction potential of lesinurad, clinical drug interaction studies were conducted. Open-label studies in volunteers investigated the effects of single-/multiple-dose lesinurad on the pharmacokinetics of sildenafil and amlodipine (CYP3A4 induction), tolbutamide (CYP2C9 inhibition/induction), and Repaglinide (CYP2C8 inhibition/induction). There was no apparent induction of CYP2C8 and CYP2C9 following repeated lesinurad administration, although no inhibition of CYP2C9 and modest inhibition of CYP2C8 were observed following single-dose lesinurad. Consistent with in vitro observations, lesinurad (200 mg once daily) was an inducer of CYP3A based on the effects on sildenafil exposure. Sildenafil exposure decreased by approximately 34% for Cmax and AUC when administered with multiple-dose lesinurad 200 mg and allopurinol 300 mg, relative to sildenafil alone. During lesinurad therapy, the possibility of reduced efficacy of concomitant drugs that are CYP3A substrates should be considered and their efficacy monitored because of induction of CYP3A by lesinurad.
Dual crosslinked pectin-alginate network as sustained release hydrophilic matrix for repaglinide.[Pubmed:28115226]
Int J Biol Macromol. 2017 Apr;97:721-732.
Repaglinide, an oral antidiabetic agent, has a rapid onset of action and short half-life of approximately 1h. Developing a controlled and prolonged release delivery system is required to maintain its therapeutic plasma concentration and to eliminate its adverse effects particularly hypoglycemia. The present study aimed to develop controlled release Repaglinide loaded beads using sodium alginate and pectin with dual cross-linking for effective control of drug release. The prepared beads were characterized for size, percentage drug entrapment efficiency, in vitro drug release and the morphological examination using scanning electron microscope. For the comparative study, the release profile of a marketed conventional tablet of Repaglinide (Prandin((R)) tablets 2mg, Novo Nordisk) was determined by the same procedure as followed for beads. The particle size of beads was in the range of 698+/-2.34-769+/-1.43mum. The drug entrapment efficiency varied between 55.24+/-4.61 to 82.29+/-3.42%. The FTIR results suggest that there was no interaction between Repaglinide and excipients. The XRD and DSC results suggest partial molecular dispersion and amorphization of the drug throughout the system. These results suggest that Repaglinide did not dissolve completely in the polymer composition and seems not to be involved in the cross-linking reaction. The percent drug release was decreased with higher polymer concentrations. In conclusion, the developed beads could enhance drug entrapment efficiency, prolong the drug release and enhance bioavailability for better control of diabetes.
Kir6.2-dependent high-affinity repaglinide binding to beta-cell K(ATP) channels.[Pubmed:15678092]
Br J Pharmacol. 2005 Feb;144(4):551-7.
1. The beta-cell K(ATP) channel is composed of two types of subunit - the inward rectifier K(+) channel (Kir6.2) which forms the channel pore, and the sulphonylurea receptor (SUR1), which serves as a regulatory subunit. The N-terminus of Kir6.2 is involved in transduction of sulphonylurea binding into channel closure, and deletion of the N-terminus (Kir6.2DeltaN14) results in functional uncoupling of the two subunits. In this study, we investigate the interaction of the hypoglycaemic agents Repaglinide and glibenclamide with SUR1 and the effect of Kir6.2 on this interaction. We further explore how the binding properties of Repaglinide and glibenclamide are affected by functional uncoupling of SUR1 and Kir6.2 in Kir6.2DeltaN14/SUR1 channels. All binding experiments are performed on membranes in ATP-free buffer at 37 degrees C. 2. Repaglinide was found to bind with low affinity (K(D)=59+/-16 nM) to SUR1 alone, but with high affinity (increased approximately 150-fold) when SUR1 was co-expressed with Kir6.2 (K(D)=0.42+/-0.03 nM). Glibenclamide, tolbutamide and nateglinide all bound with marginally lower affinity to SUR1 than to Kir6.2/SUR1. 3. Repaglinide bound with low affinity (K(D)=51+/-23 nM) to SUR1 co-expressed with Kir6.2DeltaN14. In contrast, the affinity for glibenclamide, tolbutamide and nateglinide was only mildly changed as compared to wild-type channels. 4. In whole-cell patch-clamp experiments inhibition of Kir6.2DeltaN14/SUR1 currents by both Repaglinide and nateglinde is abolished. 5. The results suggest that Kir6.2 causes a conformational change in SUR1 required for high-affinity Repaglinide binding, or that the high-affinity Repaglinide-binding site includes contributions from both SUR1 and Kir6.2. Glibenclamide, tolbutamide and nateglinide binding appear to involve only SUR1.
Hypoglycaemic effects of the novel antidiabetic agent repaglinide in rats and dogs.[Pubmed:9283692]
Br J Pharmacol. 1997 Aug;121(8):1597-604.
1. Repaglinide, a novel compound with a nonsulphonylurea structure, is currently being clinically tested as a therapeutic agent. In the present study, the hypoglycaemic effects of Repaglinide in rats and dogs were investigated. 2. Whereas the R-enantiomer, AG-EE 624 ZW, showed only weak hypoglycaemic activity, the S-enantiomer, Repaglinide, turned out to be a potent hypoglycaemic compound in rats after oral as well as after intravenous administration. Only 50% of the dose of Repaglinide was needed to be equieffective with the racemic mixture AG-EE 388 ZW. The corresponding ED50 values calculated for the effects after 120 min p.a. (intravenous administration) were 3.4 micrograms kg-1 (Repaglinide) and 6 micrograms kg-1 (AG-EE 388 ZW). 3. When compared to glimepiride or glibenclamide, Repaglinide displayed a 18 to 25 times higher potency in fasted rats. The ED50 values calculated for the effects after 120 min p.a. (oral administration) were 10 micrograms kg-1 (Repaglinide), 182 micrograms kg-1 (glimepiride) and 255 micrograms kg-1 (glibenclamide). 4. In glucose loaded rats (0.5, 1.0, 2.0 and 3.0 g kg-1 glucose, p.o.) Repaglinide exerted a very strong antihyperglycaemic activity which was even more pronounced than under normoglycaemic conditions. So for a reduction in blood glucose of 1 mmol l-1, 10.3, 9.3, 7.0 8.4 and 7.2 micrograms kg-1 Repaglinide were needed after glucose loads of 0.0, 0.5, 1.0, 2.0 and 3.0 g kg-1, respectively. 5. In beagle dogs Repaglinide again showed a pronounced hypoglycaemic effect (ED50 28.3 micrograms kg-1) which lasted for up to 24 h. However, insulin levels were only transiently increased. 6. The in vivo data presented are well supported by recently published in vitro findings. From its activity profile, Repaglinide appears to be a promising new therapeutic agent.


